Get your patient on Acetylcysteine - Acetylcysteine solution (Acetylcysteine)
Acetylcysteine - Acetylcysteine solution prescribing information
INDICATIONS AND USAGE:
Acetylcysteine Solution, USP is indicated as adjuvant therapy for patients with abnormal, viscid, or inspissated mucous secretions in such conditions as:
Chronic bronchopulmonary disease (chronic emphysema, emphysema with bronchitis, chronic asthmatic bronchitis, tuberculosis, bronchiectasis and primary amyloidosis of the lung)
Acute bronchopulmonary disease (pneumonia, bronchitis, tracheobronchitis)
Pulmonary complications of cystic fibrosis
Tracheostomy care
Pulmonary complications associated with surgery
Use during anesthesia
Post-traumatic chest conditions
Atelectasis due to mucous obstruction
Diagnostic bronchial studies (bronchograms, bronchospirometry, and bronchial wedge catheterization)
DOSAGE AND ADMINISTRATION:
General
Acetylcysteine is available in rubber stoppered glass vials containing 4, 10, or 30 mL. The 20% solution may be diluted to a lesser concentration with either Sodium Chloride Injection, Sodium Chloride Inhalation Solution, Sterile Water for Injection, or Sterile Water for Inhalation. The 10% solution may be used undiluted.
Acetylcysteine does not contain an antimicrobial agent, and care must be taken to minimize contamination of the sterile solution. If only a portion of the solution in a vial is used for inhalation, store the remainder in a refrigerator and use within 96 hours.
Nebulization-face mask, mouth piece, tracheostomy
When nebulized into a face mask, mouth piece, or tracheostomy, 1 to 10 mL of the 20% solution or 2 to 20 mL of the 10% solution may be given every 2 to 6 hours; the recommended dose for most patients is 3 to 5 mL of the 20% solution or 6 to 10 mL of the 10% solution 3 to 4 times a day.
Nebulization tent, Croupette
In special circumstances it may be necessary to nebulize into a tent or Croupette, and this method of use must be individualized to take into account the available equipment and the patient's particular needs. This form of administration requires very large volumes of the solution, occasionally as much as 300 mL during a single treatment period.
If a tent or Croupette must be used, the recommended dose is the volume of acetylcysteine (using 10% or 20%) that will maintain a very heavy mist in the tent or Croupette for the desired period. Administration for intermittent or continuous prolonged periods, including overnight, may be desirable.
Direct Instillation
When used by direct instillation, 1 to 2 mL of a 10% to 20% solution may be given as often as every hour. When used for the routine nursing care of patients with tracheostomy, 1 to 2 mL of a 10% to 20% solution may be given every 1 to 4 hours by instillation into the tracheostomy.
Acetylcysteine may be introduced directly into a particular segment of the bronchopulmonary tree by inserting (under local anesthesia and direct vision) a small plastic catheter into the trachea. Two to 5 mL of the 20% solution may then be instilled by means of a syringe connected to the catheter.
Acetylcysteine may also be given through a percutaneous intratracheal catheter. One to 2 mL of the 20% or 2 to 4 mL of the 10% solution every 1 to 4 hours may then be given by a syringe attached to the catheter.
Diagnostic Bronchograms
For diagnostic bronchial studies, two or three administrations of 1 to 2 mL of the 20% solution or 2 to 4 mL of the 10% solution should be given by nebulization or by instillation intratracheally, prior to the procedure.
Administration of Aerosol
Materials
Acetylcysteine may be administered using conventional nebulizers made of plastic or glass. Certain materials used in nebulization equipment react with acetylcysteine. The most reactive of these are certain metals (notably iron and copper) and rubber. Where materials may come into contact with acetylcysteine solution, parts made of the following acceptable materials should be used: glass, plastic, aluminum, anodized aluminum, chromed metal, tantalum, sterling silver, or stainless steel. Silver may become tarnished after exposure, but this is not harmful to the drug action or to the patient.
Nebulizing Gases
Compressed tank gas (air) or an air compressor should be used to provide pressure for nebulizing the solution. Oxygen may also be used but should be used with the usual precautions in patients with severe respiratory disease and CO 2 retention.
Apparatus
Acetylcysteine is usually administered as fine nebulae and the nebulizer used should be capable of providing optimal quantities of a suitable range of particle sizes.
Commercially available nebulizers will produce nebulae of acetylcysteine satisfactory for retention in the respiratory tract. Most of the nebulizers tested will supply a high proportion of the drug solution as particles of less than 10 microns in diameter. Mitchell 2 has shown that particles less than 10 microns should be retained in the respiratory tract satisfactorily.
Various intermittent positive pressure breathing devices nebulized acetylcysteine with a satisfactory efficiency including: No. 40 De Vilbiss (The De Vilbiss Co., Somerset, Pennsylvania), and the Bennett Twin-Jet Nebulizer (Puritan Bennett Corp., Oak at 13th., Kansas City, Missouri).
The nebulized solution may be inhaled directly from the nebulizer. Nebulizers may also be attached to the plastic face masks or plastic mouthpieces. Suitable nebulizers may also be fitted for use with the various intermittent positive pressure breathing (IPPB) machines. The nebulizing equipment should be cleaned immediately after use because the residues may clog the smaller orifices or corrode metal parts.
Hand bulbs are not recommended for routine use in nebulizing acetylcysteine because their output is generally too small. Also, some hand-operated nebulizers deliver particles that are larger than optimum for inhalation therapy.
Acetylcysteine should not be placed directly into the chamber of a heated (hot pot) nebulizer. A heated nebulizer may be part of the nebulization assembly to provide a warm saturated atmosphere if the acetylcysteine aerosol is introduced by means of a separate unheated nebulizer. Usual precautions for administration of warm saturated nebulae should be observed.
The nebulized solution may be breathed directly from the nebulizer. Nebulizers may also be attached to plastic face masks, plastic face tents, plastic mouth pieces, conventional plastic oxygen tents, or head tents. Suitable nebulizers may also be fitted for use with the various intermittent positive pressure breathing (IPPB) machines.
The nebulizing equipment should be cleaned immediately after use, otherwise the residues may occlude the fine orifices or corrode metal parts.
Prolonged Nebulization
When three fourths of the initial volume of acetylcysteine solution have been nebulized, a quantity of Sterile Water for Injection, USP (approximately equal to the volume of solution remaining) should be added to the nebulizer. This obviates any concentration of the agent in the residual solvent remaining after prolonged nebulization.
Compatibility
The physical and chemical compatibility of acetylcysteine solutions with certain other drugs that might be concomitantly administered by nebulization, direct instillation, or topical application has been studied.
Acetylcysteine should not be mixed with certain antibiotics. For example, the antibiotics, tetracycline hydrochloride, oxytetracycline hydrochloride, and erythromycin lactobionate, were found to be incompatible when mixed in the same solution. These agents may be administered from separate solutions if administration of these agents is desirable.
The supplying of these data should not be interpreted as a recommendation for combining acetylcysteine with other drugs. The table is not presented as positive assurance that no incompatibility will be present, since these data are based only on short-term compatibility studies done in the Mead Johnson Research Center. Manufacturers may change their formulations, and this could alter compatibilities. These data are intended to serve only as a guide for predicting compounding problems.
If it is deemed advisable to prepare an admixture, it should be administered as soon as possible after preparation. Do not store unused mixtures.
IN VITRO COMPATIBILITY 1 TESTS OF ACETYLCYSTEINE | |||
PRODUCT AND/OR AGENT | COMPATIBILITY RATING | RATIO TESTED 6 | |
ACETYLCYSTEINE | PRODUCT OR AGENT | ||
ANESTHETIC GAS | |||
Halothane | Compatible | 20% | Infinite |
Nitrous Oxide | Compatible | 20% | Infinite |
ANESTHETIC LOCAL | |||
Cocaine HCl | Compatible | 10% | 5% |
Lidocaine HCl | Compatible | 10% | 2% |
Tetracaine HCl | Compatible | 10% | 1% |
ANTIBACTERIALS (A parenteral form of each antibiotic was used) | |||
Bacitracin 2,3 (mix and use at once) | Compatible | 10% | 5,000 U/mL |
Chloramphenicol Sodium Succinate | Compatible | 20% | 20 mg/mL |
Carbenicillin Disodium 2 (mix and use at once) | Compatible | 10% | 125 mg/mL |
Gentamicin Sulfate 2 | Compatible | 10% | 20 mg/mL |
Kanamycin Sulfate 2 (mix and use at once) | Compatible | 10% | 167 mg/mL |
Compatible | 17% | 85 mg/mL | |
Lincomycin HCl 2 | Compatible | 10% | 150 mg/mL |
Neomycin Sulfate 2 | Compatible | 10% | 100 mg/mL |
Novobiocin Sodium 2 | Compatible | 10% | 25 mg/mL |
Penicillin G Potassium 2 (mix and use at once) | Compatible | 10% | 25,000 U/mL |
Compatible | 10% | 100,000 U/mL | |
Polymyxin B Sulfate 2 | Compatible | 10% | 50,000 U/mL |
Cephalothin Sodium | Compatible | 10% | 110 mg/mL |
Colistimethate Sodium 2 (mix and use at once) | Compatible | 10% | 37.5 mg/mL |
Vancomycin HCl 2 | Compatible | 10% | 25 mg/mL |
Amphotericin B | Incompatible | 4% to 15% | 1 to 4 mg/mL |
Chlortetracycline HCl 2 | Incompatible | 10% | 12.5 mg/mL |
Erythromycin Lactobionate | Incompatible | 10% | 15 mg/mL |
Oxytetracycline HCl | Incompatible | 10% | 12.5 mg/mL |
Ampicillin Sodium | Incompatible | 10% | 50 mg/mL |
Tetracycline HCl | Incompatible | 10% | 12.5 mg/mL |
BRONCHODILATORS | |||
Isoproterenol HCl 2 | Compatible | 3.0% | 0.5% |
Isoproterenol HCl 2 | Compatible | 10% | 0.05% |
Isoproterenol HCl 2 | Compatible | 20% | 0.05% |
Isoproterenol HCl | Compatible | 13.3% (2 parts) | 0.33% (1 part) |
Isoetharine HCl | Compatible | 13.3% (2 parts) | (1 part) |
Epinephrine HCl | Compatible | 13.3% (2 parts) | 0.33% (1 part) |
CONTRAST MEDIA | |||
Iodized Oil | Incompatible | 20%/20 mL | 40%/10 mL |
DECONGESTANTS | |||
Phenylephrine HCl 2 | Compatible | 3.0% | 0.25% |
Phenylephrine HCl | Compatible | 13.3% (2 parts) | 0.17% (1 part) |
ENZYMES | |||
Chymotrypsin | Incompatible | 5% | 400 γ/mL |
Trypsin | Incompatible | 5% | 400 γ/mL |
SOLVENTS | |||
Alcohol | Compatible | 12% | 10% to 20% |
Propylene Glycol | Compatible | 3% | 10% |
STEROIDS | |||
Dexamethasone Sodium Phosphate | Compatible | 16% | 0.8 mg/mL |
Prednisolone Sodium Phosphate 5 | Compatible | 16.7% | 3.3 mg/mL |
OTHER AGENTS | |||
Hydrogen Peroxide | Incompatible | (All ratios) | |
Sodium Bicarbonate | Compatible | 20% (1 part) | 4.2% (1 part) |
| |||
CONTRAINDICATIONS:
Acetylcysteine is contraindicated in those patients who are sensitive to it.
ADVERSE REACTIONS:
Adverse effects have included stomatitis, nausea, vomiting, fever, rhinorrhea, drowsiness, clamminess, chest tightness, and bronchoconstriction. Clinically overt acetylcysteine induced bronchospasm occurs infrequently and unpredictably even in patients with asthmatic bronchitis or bronchitis complicating bronchial asthma.
Acquired sensitization to acetylcysteine has been reported rarely. Reports of sensitization in patients have not been confirmed by patch testing. Sensitization has been confirmed in several inhalation therapists who reported a history of dermal eruptions after frequent and extended exposure to acetylcysteine.
Reports of irritation to the tracheal and bronchial tracts have been received and although hemoptysis has occurred in patients receiving acetylcysteine such findings are not uncommon in patients with bronchopulmonary disease and a causal relationship has not been established.
Drug Interactions
Drug stability and safety of acetylcysteine when mixed with other drugs in a nebulizer have not been established.
DESCRIPTION:
Acetylcysteine Solution, USP is for inhalation (mucolytic agent) or oral administration (acetaminophen antidote), available as a sterile, unpreserved solution (NOT FOR INJECTION). The solutions contain 20% (200 mg/mL) or 10% (100 mg/mL) acetylcysteine, with disodium edetate in purified water. Sodium hydroxide and/or hydrochloric acid is added to adjust pH (range 6.0 to 7.5). Acetylcysteine is the N-acetyl derivative of the naturally-occurring amino acid, cysteine. The compound is a white crystalline powder with the molecular formula C 5 H 9 NO 3 S, a molecular weight of 163.2, and chemical name of N-acetyl-L-cysteine. Acetylcysteine has the following structural formula:
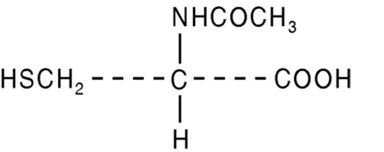
This product contains the following inactive ingredients: disodium edetate, sodium hydroxide, purified water, and hydrochloric acid, if necessary.
ACETYLCYSTEINE AS A MUCOLYTIC AGENT CLINICAL PHARMACOLOGY:
The viscosity of pulmonary mucous secretions depends on the concentrations of mucoprotein and, to a lesser extent, deoxyribonucleic acid (DNA). The latter increases with increasing purulence owing to the presence of cellular debris. The mucolytic action of acetylcysteine is related to the sulfhydryl group in the molecule. This group probably “opens” disulfide linkages in mucus thereby lowering the viscosity. The mucolytic activity of acetylcysteine is unaltered by the presence of DNA, and increases with increasing pH. Significant mucolysis occurs between pH 7 and 9.
Acetylcysteine undergoes rapid deacetylation in vivo to yield cysteine or oxidation to yield diacetylcystine.
Occasionally, patients exposed to the inhalation of an acetylcysteine aerosol respond with the development of increased airways obstruction of varying and unpredictable severity. Those patients who are reactors cannot be identified a priori from a random patient population. Even when patients are known to have reacted previously to the inhalation of an acetylcysteine aerosol, they may not react during a subsequent treatment. The converse is also true; patients who have had inhalation treatments of acetylcysteine without incident may still react to subsequent inhalation with increased airways obstruction. Most patients with bronchospasm are quickly relieved by the use of a bronchodilator given by nebulization. If bronchospasm progresses, the medication should be discontinued immediately.
HOW SUPPLIED
Acetylcysteine Solution, USP is available in rubber stoppered glass vials containing 4, or 30 mL. The 20% solution may be diluted to a lesser concentration with either Sodium Chloride for Injection, Sodium Chloride for Inhalation, Sterile Water for Injection, or Sterile Water for Inhalation.
Acetylcysteine is sterile, not for injection and can be used for inhalation (mucolytic agent) or oral administration (acetaminophin antidote). It is available as follows:
Acetylcysteine 20% solution (200 mg acetylcysteine per mL). Sterile, not for injection.
| Product Code | Unit of Sale | Strength | Each |
| PRX694104 | NDC 63323-694-44 Unit of 25 | 800 mg per 4 mL 20% (200 mg/mL) | NDC 63323-694-41 4 mL fill in a 5 mL vial |
| PRX690130 | NDC 63323-690-44 Unit of 3 | 6 g per 30 mL 20% (200 mg/mL) | NDC 63323-690-41 30 mL fill in a 30 mL vial |
STORAGE:
Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature]. Store in refrigerator 2° - 8°C (36° - 46°F) after opening.
Acetylcysteine does not contain an antimicrobial agent, and care must be taken to minimize contamination of the sterile solution. Dilutions of acetylcysteine should be used freshly prepared and utilized within one hour. If only a portion of the solution in a vial is used, store the remaining undiluted portion in a refrigerator and use within 96 hours.