Get your patient on Acyclovir - Acyclovir ointment (Acyclovir)
Acyclovir - Acyclovir ointment prescribing information
INDICATIONS AND USAGE
Acyclovir ointment USP, 5% is indicated in the management of initial genital herpes and in limited non-life-threatening mucocutaneous Herpes simplex virus infections in immunocompromised patients.
DOSAGE AND ADMINISTRATION
Apply sufficient quantity to adequately cover all lesions every 3 hours, 6 times per day for 7 days. The dose size per application will vary depending upon the total lesion area but should approximate a one-half inch ribbon of ointment per 4 square inches of surface area. A finger cot or rubber glove should be used when applying acyclovir ointment 5% to prevent autoinoculation of other body sites and transmission of infection to other persons. Therapy should be initiated as early as possible following onset of signs and symptoms.
CONTRAINDICATIONS
Acyclovir ointment 5% is contraindicated in patients who develop hypersensitivity to the components of the formulation.
ADVERSE REACTIONS
In the controlled clinical trials, mild pain (including transient burning and stinging) was reported by about 30% of patients in both the active and placebo arms; treatment was discontinued in 2 of these patients. Local pruritus occurred in 4% of these patients. In all studies, there was no significant difference between the drug and placebo group in the rate or type of reported adverse reactions nor were there any differences in abnormal clinical laboratory findings.
Observed During Clinical Practice: Based on clinical practice experience in patients treated with acyclovir ointment 5% in the US, spontaneously reported adverse events are uncommon. Data are insufficient to support an estimate of their incidence or to establish causation. These events may also occur as part of the underlying disease process. Voluntary reports of adverse events that have been received since market introduction include:
General: Edema and/or pain at the application site.
Skin: Pruritus, rash.
Drug Interactions:
Clinical experience has identified no interactions resulting from topical or systemic administration of other drugs concomitantly with acyclovir ointment 5%.
DESCRIPTION
Acyclovir, USP, is a synthetic nucleoside analogue active against herpes viruses. Acyclovir ointment USP, 5% is a formulation for topical administration. Each gram acyclovir ointment USP, 5% contains 50 mg of acyclovir in a polyethylene glycol (PEG) base.
Acyclovir, USP is a white, crystalline powder with the molecular formula C 8 H 11 N 5 O 3 and a molecular weight of 225. The maximum solubility in water at 37°C is 2.5 mg/mL. The pka's of acyclovir are 2.27 and 9.25.
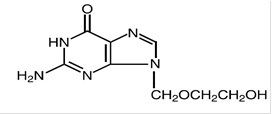
The chemical name of acyclovir is 2-amino-1, 9-dihydro-9- [(2-hydroxyethoxy)methyl]-6 H - purin-6-one; it has the following structural formula:
CLINICAL PHARMACOLOGY
Two clinical pharmacology studies were performed with acylovir ointment 5% in immunocompromised adults at risk of developing mucocutaneous Herpes simplex virus infections or with localized varicella-zoster infections. These studies were designed to evaluate the dermal tolerance, systemic toxicity, and percutaneous absorption of acyclovir.
In 1 of these studies, which included 16 inpatients, the complete ointment or its vehicle were randomly administered in a dose of 1-cm strips (25 mg acyclovir) 4 times a day for 7 days to an intact skin surface area of 4.5 square inches. No local intolerance, systemic toxicity, or contact dermatitis were observed. In addition, no drug was detected in blood and urine by radioimmunoassay (sensitivity, 0.01 mcg/mL).
The other study included 11 patients with localized varicella-zoster infections. In this uncontrolled study, acyclovir was detected in the blood of 9 patients and in the urine of all patients tested. Acyclovir levels in plasma ranged from <0.01 to 0.28 mcg/mL in 8 patients with normal renal function, and from <0.01 to 0.78 mcg/mL in 1 patient with impaired renal function. Acyclovir excreted in the urine ranged from <0.02% to 9.4% of the daily dose. Therefore, systemic absorption of acyclovir after topical application is minimal.
CLINICAL TRIALS
In clinical trials of initial genital herpes infections, acyclovir ointment 5% has shown a decrease in healing time and, in some cases, a decrease in duration of viral shedding and duration of pain. In studies in immunocompromised patients mainly with herpes labialis, there was a decrease in duration of viral shedding and a slight decrease in duration of pain.
In studies of recurrent genital herpes and of herpes labialis in nonimmunocompromised patients, there was no evidence of clinical benefit; there was some decrease in duration of viral shedding.
HOW SUPPLIED
Acyclovir Ointment USP, 5% is supplied as a white to off-white color ointment. Each gram of acyclovir ointment USP, 5% contains 50 mg acyclovir, USP in a polyethylene glycol base.
It is supplied as follows:
5-g tubes NDC 62332-248-05
15-g tubes NDC 62332-248-15
30-g tubes NDC 62332-248-30
Store at 15° to 25°C (59° to 77°F). [See USP Controlled Room Temperature]. Store in a dry place
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
For more information call Alembic Pharmaceuticals Limited at 1-866-210-9797.
Manufactured for:
Alembic Pharmaceuticals, Inc.
Bedminster, NJ 07921, USA
Manufactured by:
Alembic Pharmaceuticals Limited
(Derma Division),
Karakhadi, Vadodara 391450, India.
Mfg. License No.: G/25/2216
Manufactured by:
Encube Ethicals Pvt. Ltd.,
Madkaim, Ponda, Goa, India – 403404
Mfg. License No: 361
Revised: 03/2025