Get your patient on Adapalene - Adapalene cream (Adapalene)
Adapalene - Adapalene cream prescribing information
INDICATIONS AND USAGE
Adapalene Cream, 0.1% is indicated for the topical treatment of acne vulgaris.
DOSAGE AND ADMINISTRATION
Adapalene Cream, 0.1% should be applied to affected areas of the skin, once daily at nighttime. A thin film of the cream should be applied to the skin areas where acne lesions appear, using enough to cover the entire affected areas lightly. A mild transitory sensation of warmth or slight stinging may occur shortly after the application of Adapalene Cream, 0.1%.
CONTRAINDICATIONS
Adapalene Cream, 0.1% should not be administered to individuals who are hypersensitive to adapalene or any of the components in the cream vehicle.
ADVERSE REACTIONS
In controlled clinical trials, local cutaneous irritation was monitored in 285 acne patients who used Adapalene Cream, 0.1% once daily for 12 weeks. The frequency and severity of erythema, scaling, dryness, pruritus and burning were assessed during these studies. The incidence of local cutaneous irritation with Adapalene Cream, 0.1% from the controlled clinical studies is provided in the following table:
| None | Mild | Moderate | Severe | |
| Erythema | 52% (148) | 38% (108) | 10% (28) | ‹1% (1) |
| Scaling | 58% (166) | 35% (100) | 6% (18) | ‹1% (1) |
| Dryness | 48% (136) | 42% (121) | 9% (26) | ‹1% (2) |
| Pruritus (persistent) | 74% (211) | 21% (61) | 4% (12) | ‹1% (1) |
| Burning/Stinging (persistent) | 71% (202) | 24% (69) | 4% (12) | ‹1% (2) |
Other reported local cutaneous adverse events in patients who used Adapalene Cream, 0.1% once daily included: sunburn (2%), skin discomfort-burning and stinging (1%) and skin irritation (1%). Events occurring in less than 1% of patients treated with Adapalene Cream, 0.1% included: acne flare, dermatitis and contact dermatitis, eyelid edema, conjunctivitis, erythema, pruritus, skin discoloration, rash, and eczema.
Drug Interactions
As Adapalene Cream, 0.1% has the potential to produce local irritation in some patients, concomitant use of other potentially irritating topical products (medicated or abrasive soaps and cleansers, soaps and cosmetics that have a strong drying effect, and products with high concentrations of alcohol, astringents, spices or lime rind) should be approached with caution. Particular caution should be exercised in using preparations containing sulfur, resorcinol, or salicylic acid in combination with Adapalene Cream, 0.1%. If these preparations have been used, it is advisable not to start therapy with Adapalene Cream, 0.1% until the effects of such preparations in the skin have subsided.
DESCRIPTION
Adapalene Cream, 0.1%, contains adapalene 0.1% in an aqueous cream emulsion consisting of carbomer 934P, cyclomethicone, edetate disodium, glycerin, methyl glucose sesquistearate, methylparaben, PEG-20 methyl glucose sesquistearate, phenoxyethanol, propylparaben, purified water, squalane, and trolamine.
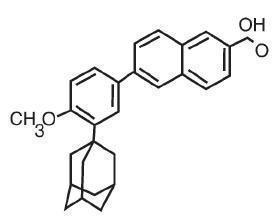
The chemical name of adapalene is 6-[3-(1-adamantyl)-4-methoxyphenyl]-2-naphthoic acid. It is a white to off-white powder which is soluble in tetrahydrofuran, sparingly soluble in ethanol, and practically insoluble in water. The molecular formula is C 28 H 28 O 3 and molecular weight is 412.53. Adapalene is represented by the following structural formula.
CLINICAL PHARMACOLOGY
Mechanism of Action
Adapalene acts on retinoid receptors. Biochemical and pharmacological profile studies have demonstrated that adapalene is a modulator of cellular differentiation, keratinization, and inflammatory processes all of which represent important features in the pathology of acne vulgaris.
Mechanistically, adapalene binds to specific retinoic acid nuclear receptors but does not bind to the cytosolic receptor protein. Although the exact mode of action of adapalene is unknown, it is suggested that topical adapalene normalizes the differentiation of follicular epithelial cells resulting in decreased microcomedone formation.
Pharmacokinetics
Absorption of adapalene from Adapalene Cream, 0.1% through human skin is low. In a pharmacokinetic study with six acne patients treated once daily for 5 days with 2 grams of Adapalene Cream, 0.1% applied to 1000 cm 2 of acne involved skin, there were no quantifiable amounts (limit of quantification = 0.35 ng/mL) of adapalene in the plasma samples from any patient. Excretion appears to be primarily by the biliary route.
CLINICAL STUDIES
Two vehicle-controlled clinical studies were conducted in patients 12 to 30 years of age with mild to moderate acne vulgaris, in which Adapalene Cream, 0.1% was compared with its vehicle. Patients were instructed to apply their treatment medication once daily at bedtime for 12 weeks. In one study patients were provided with a soapless cleanser and were encouraged to refrain from using moisturizers. No other topical medications, other than Adapalene Cream, 0.1%, were to be applied to the face during the studies. Adapalene Cream, 0.1% was significantly more effective than its vehicle in the reduction of acne lesion counts. The mean percent reduction in lesion counts from baseline after treatment for 12 weeks are presented in the following table:
| Efficacy Variable | Study No. 1 | Study No. 2 | ||
Adapalene Cream, 0.1% N=119 | Cream Vehicle N=118 | Adapalene Cream, 0.1% N=175 | Cream Vehicle N=175 | |
| Non-inflammatory lesions | 34% | 18% | 35% | 15% |
| Inflammatory lesions | 32% | 17% | 14% | 6% |
| Total lesions | 34% | 18% | 30% | 15% |
The trend in the Investigator’s global assessment of severity supported the efficacy of Adapalene Cream, 0.1% when compared to the cream vehicle.
HOW SUPPLIED
Adapalene Cream, 0.1% is available as follows:
45 g tube (NDC 45802- 453 -84)
Mechanism of Action
Adapalene acts on retinoid receptors. Biochemical and pharmacological profile studies have demonstrated that adapalene is a modulator of cellular differentiation, keratinization, and inflammatory processes all of which represent important features in the pathology of acne vulgaris.
Mechanistically, adapalene binds to specific retinoic acid nuclear receptors but does not bind to the cytosolic receptor protein. Although the exact mode of action of adapalene is unknown, it is suggested that topical adapalene normalizes the differentiation of follicular epithelial cells resulting in decreased microcomedone formation.