Get your patient on Albuterol Sulfate - Albuterol Sulfate solution (Albuterol Sulfate)
Albuterol Sulfate - Albuterol Sulfate solution prescribing information
INDICATIONS AND USAGE
Albuterol sulfate inhalation solution is indicated for the relief of bronchospasm in patients 2 to 12 years of age with asthma (reversible obstructive airway disease).
DOSAGE AND ADMINISTRATION
The usual starting dosage for patients 2 to 12 years of age is 1.25 mg or 0.63 mg of albuterol sulfate inhalation solution administered 3 or 4 times daily, as needed, by nebulization. More frequent administration is not recommended.
To administer 1.25 mg or 0.63 mg of albuterol, use the entire contents of one unit-dose vial (3 mL of 1.25 mg or 0.63 mg inhalation solution) by nebulization. Adjust nebulizer flow rate to deliver albuterol sulfate inhalation solution over 5 to 15 minutes.
The use of albuterol sulfate inhalation solution can be continued as medically indicated to control recurring bouts of bronchospasm. During this time most patients gain optimum benefit from regular use of the inhalation solution.
Patients 6 to 12 years of age with more severe asthma (baseline FEV 1 less than 60% predicted), weight > 40 kg, or patients 11 to 12 years of age may achieve a better initial response with the 1.25 mg dose.
Albuterol sulfate inhalation solution has not been studied in the setting of acute attacks of bronchospasm. A 2.5 mg dose of albuterol provided by a higher concentration product (2.5 mg albuterol per 3 mL) may be more appropriate for treating acute exacerbations, particularly in children 6 years old and above.
If a previously effective dosage regimen fails to provide the usual relief, medical advice should be sought immediately, as this is often a sign of seriously worsening asthma which would require reassessment of therapy.
The drug compatibility (physical and chemical), clinical efficacy and safety of albuterol sulfate inhalation solution, when mixed with other drugs in a nebulizer have not been established.
The safety and efficacy of albuterol sulfate inhalation solution have been established in clinical trials when administered using the Pari LC Plus™ nebulizer and Pari PRONEB™ compressor. The safety and efficacy of albuterol sulfate inhalation solution when administered with other nebulizer systems have not been established.
Albuterol sulfate inhalation solution should be administered via jet nebulizer connected to an air compressor with adequate air flow, equipped with a mouthpiece or suitable face mask.
CONTRAINDICATIONS
Albuterol sulfate inhalation solution is contraindicated in patients with a history of hypersensitivity to any of its components.
ADVERSE REACTIONS
Clinical Trial Experience: Adverse events reported in >1% of patients receiving albuterol sulfate inhalation solution and more frequently than in patients receiving placebo in a four-week double-blind study are listed in the following table.
| 1.25 mg Albuterol Sulfate Inhalation Solution (N=115) | 0.63 mg Albuterol Sulfate Inhalation Solution (N=117) | Placebo (N=117) | |
| Asthma Exacerbation | 13 | 11.1 | 8.5 |
| Otitis Media | 4.3 | 0.9 | 0 |
| Allergic Reaction | 0.9 | 3.4 | 1.7 |
| Gastroenteritis | 0.9 | 3.4 | 0.9 |
| Cold Symptoms | 0 | 3.4 | 1.7 |
| Flu Syndrome | 2.6 | 2.6 | 1.7 |
| Lymphadenopathy | 2.6 | 0.9 | 1.7 |
| Skin/Appendage Infection | 1.7 | 0 | 0 |
| Urticaria | 1.7 | 0.9 | 0 |
| Migraine | 0.9 | 1.7 | 0 |
| Chest Pain | 0.9 | 1.7 | 0 |
| Bronchitis | 0.9 | 1.7 | 0.9 |
| Nausea | 1.7 | 0.9 | 0.9 |
There was one case of ST segment depression in the 1.25 mg albuterol sulfate inhalation solution treatment group.
No clinically relevant laboratory abnormalities related to albuterol sulfate inhalation solution administration were seen in this study.
Postmarketing Experience: Metabolic acidosis has been reported after the use of albuterol sulfate inhalation solution. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate its frequency or establish a causal relationship to drug exposure.
To report SUSPECTED ADVERSE REACTIONS, contact Ingenus Pharmaceuticals, LLC at 1-877-748-1970 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
DESCRIPTION
Albuterol sulfate inhalation solution is a sterile, clear, colorless solution of the sulfate salt of racemic albuterol, albuterol sulfate. Albuterol sulfate is a relatively selective beta 2 -adrenergic bronchodilator (see CLINICAL PHARMACOLOGY ). The chemical name for albuterol sulfate is α 1 [(tert-butylamino) methyl]-4-hydroxy-m-xylene-α, α'-diol sulfate (2:1) (salt), and its established chemical structure is as follows:
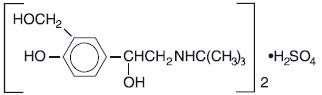
The molecular weight of albuterol sulfate is 576.7 and the empirical formula is (C 13 H 21 NO 3 ) 2 • H 2 SO 4 . Albuterol sulfate is a white crystalline powder, soluble in water and slightly soluble in ethanol. The World Health Organization recommended name for albuterol is salbutamol.
Albuterol sulfate inhalation solution is supplied in two strengths in unit dose vials. Each unit dose vial contains either 0.63 mg of albuterol equivalent to 0.75 mg of albuterol sulfate or 1.25 mg of albuterol equivalent to 1.5 mg of albuterol sulfate with sodium chloride and sulfuric acid in a 3-mL isotonic, sterile, aqueous solution. Sodium chloride is added to adjust isotonicity of the solution and sulfuric acid is added to adjust pH of the solution to between 3.0 and 5.0 (see HOW SUPPLIED ).
Albuterol sulfate inhalation solution does not require dilution prior to administration by nebulization. For albuterol sulfate inhalation solution, like all other nebulized treatments, the amount delivered to the lungs will depend on patient factors, the jet nebulizer utilized, and compressor performance. Using the Pari LC Plus™ nebulizer (with face mask or mouthpiece) connected to a Pari PRONEB™ compressor, under in vitro conditions, the mean delivered dose from the mouth piece (% nominal dose) was approximately 43% of albuterol (1.25 mg strength) and 39% of albuterol (0.63 mg strength) at a mean flow rate of 3.6 L/min. The mean nebulization time was 15 minutes or less. Albuterol sulfate inhalation solution should be administered from a jet nebulizer at an adequate flow rate, via a mouthpiece or face mask (see DOSAGE AND ADMINISTRATION ).
CLINICAL PHARMACOLOGY
The prime action of beta-adrenergic drugs is to stimulate adenyl cyclase, the enzyme which catalyzes the formation of cyclic-3',5'-adenosine monophosphate (cyclic AMP) from adenosine triphosphate (ATP). The cyclic AMP thus formed mediates the cellular responses. In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta 2 –adrenergic receptors compared with isoproterenol. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors in bronchial smooth muscle, recent data indicate that 10% to 50% of the beta-receptors in the human heart may be beta 2 -receptors. The precise function of these receptors, however, is not yet established. Controlled clinical studies and other clinical experience have shown that inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes. Albuterol is longer acting than isoproterenol in most patients by any route of administration because it is not a substrate for the cellular uptake processes for catecholamines nor for catechol-O-methyl transferase.
Pharmacokinetics: Studies in asthmatic patients have shown that less than 20% of a single albuterol dose was absorbed following either intermittent positive-pressure breathing (IPPB) or nebulizer administration; the remaining amount was recovered from the nebulizer and apparatus, and expired air. Most of the absorbed dose was recovered in urine collected during the 24 hours after drug administration. Following oral administration of 4 mg albuterol, the elimination half-life was five to six hours. Following a 3 mg dose of nebulized albuterol in adults, the mean maximum albuterol plasma level at 0.5 hours was 2.1 ng/mL (range, 1.4 to 3.2 ng/mL). The pharmacokinetics of albuterol following administration of 0.63 mg or 1.25 mg albuterol sulfate inhalation solution by nebulization have not been determined in children 2 to 12 years old.
Animal Pharmacology/Toxicology: Intravenous studies in rats with albuterol sulfate have demonstrated that albuterol crosses the blood-brain barrier and reaches brain concentrations amounting to approximately 5% of plasma concentrations. In structures outside the blood-brain barrier (pineal and pituitary glands), albuterol concentrations were found to be 100 times those found in whole brain.
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta-agonists and methylxanthines are administered concurrently. The clinical significance of these findings is unknown.
Clinical Trials: The safety and efficacy of albuterol sulfate inhalation solution was evaluated in a 4-week, multi-center, randomized, double-blind, placebo-controlled, parallel group study in 349 children 6 to 12 years of age with mild-to-moderate asthma (mean baseline FEV 1 60% to 70% of predicted). Approximately half of the patients were also receiving inhaled corticosteroids. Patients were randomized to receive albuterol sulfate inhalation solution 0.63 mg, albuterol sulfate inhalation solution 1.25 mg, or placebo three times a day administered via a Pari LC Plus™ nebulizer and a Pari PRONEB™ compressor. Racemic albuterol, delivered by a chlorofluorocarbon (CFC) metered dose inhaler (MDI) or nebulized, was used on an as-needed basis as the rescue medication.
Efficacy, as measured by the mean percent change from baseline in the area under the 6-hour curve for FEV 1 , was demonstrated for both active treatment regimens (n=112 [1.25 mg group] and n=110 [0.63 mg group]) compared with placebo (n=110) on day 1 and day 28. Figures 1 and 2 illustrate the mean percentage change from pre-dose FEV 1 on day 1 and day 28, respectively. The mean baseline FEV 1 for all patients was 1.49 L.
Figure 1 % Change from Pre-Dose FEV 1 Intent-to-Treat Population Day 1
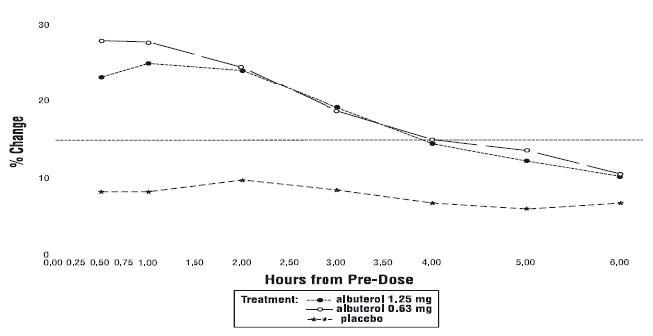
Figure 2 % Change from Pre-Dose FEV 1 Intent-to-Treat Population Day 28
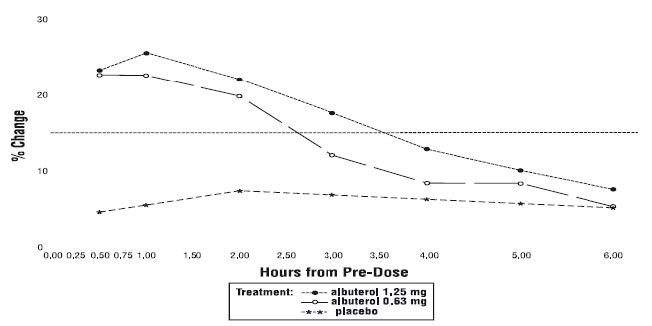
The onset of a 15% increase in FEV 1 over baseline for both doses of albuterol sulfate inhalation solution was seen at 30 minutes (the first post-dose assessment). The mean time to peak effect was approximately 30 to 60 minutes for both doses on day 1 and after 4 weeks of treatment. The mean duration of effect, as measured by a >15% increase from baseline in FEV 1 , was approximately 2.5 hours for both doses on day 1 and approximately 2 hours for both doses after 4 weeks of treatment. In some patients, the duration of effect was as long as 6 hours.
HOW SUPPLIED
Albuterol Sulfate Inhalation Solution is supplied as a 3 mL, clear, colorless, sterile, preservative-free, aqueous solution in two different strengths, 0.63 mg/3 mL and 1.25 mg/3 mL, of albuterol (equivalent to 0.75 mg of albuterol sulfate or 1.5 mg of albuterol sulfate per 3 mL) in unit-dose low-density polyethylene (LDPE) vials. Each unit-dose LDPE vial is protected in a foil pouch, and each foil pouch contains 5 unit-dose LDPE vials. Each strength of albuterol sulfate inhalation solution is available in a shelf carton containing multiple foil pouches.
Albuterol Sulfate Inhalation Solution, 0.63 mg/3 mL (potency expressed as albuterol) contains 0.75 mg albuterol sulfate per 3 mL in unit-dose vials and is available in the following packaging configuration.
NDC 50742-392-25 5 foil pouches, each containing 5 vials, total 25 vials per carton
Albuterol Sulfate Inhalation Solution, 1.25 mg/3 mL (potency expressed as albuterol) contains 1.5 mg albuterol sulfate per 3 mL in unit-dose vials and is available in the following packaging configuration.
NDC 50742-393-25 5 foil pouches, each containing 5 vials, total 25 vials per carton
Rx only
STORAGE
Store between 2°C to 25°C (36°F to 77°F). Protect from light and excessive heat.
Store unit-dose vials in protective foil pouch at all times. Once removed from the foil pouch, use vial(s) within one week. Discard the vial if the solution is not colorless.
Keep out of the reach of children.
Manufactured for:
Ingenus Pharmaceuticals, LLC
Orlando, FL 32811-7193
Made in China
Revised: 08/2024

LXK0042/01
INSTRUCTIONS FOR USE SECTION
| Albuterol Sulfate Inhalation Solution 0.63 mg•/3 mL and 1.25 mg•/3 mL (•Equivalent to 0.75 mg of albuterol sulfate or 1.5 mg of albuterol sulfate per 3 mL) |
| PATIENT'S INSTRUCTIONS FOR USE |
| Read this patient information completely every time your prescription is filled as information may have changed. Keep these instructions with your medication, as you may want to read them again. |
| Albuterol sulfate inhalation solution should only be used under the direction of a physician. Your physician and pharmacist have more information about albuterol sulfate inhalation solution and the condition for which it has been prescribed. Contact them if you have additional questions. |
| Storing your Medicine Store albuterol sulfate inhalation solution between 2°C to 25°C (36°F to 77°F). Vials should be protected from light before use, therefore, keep unused vials in the foil pouch. Do not use after the expiration (EXP) date printed on the vial. Dose Albuterol sulfate inhalation solution is supplied as a single-dose, ready-to-use vial containing 3 mL of solution. No mixing or dilution is needed. Use one new vial with each nebulizer treatment. |
| Instructions for Use 1. Remove one vial from the foil pouch. Place remaining vials back into foil pouch for storage. 2. Twist the cap completely off the vial and squeeze the contents into the nebulizer reservoir (Figure 1). |
 |
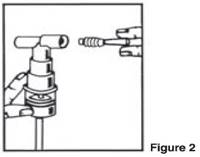
| 3. Connect the nebulizer to the mouthpiece or face mask (Figure 2). |
 |
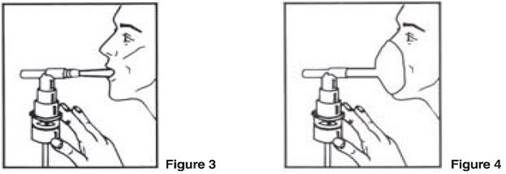
| 4. Connect the nebulizer to the compressor. 5. Sit in a comfortable, upright position; place the mouthpiece in your mouth (Figure 3) or put on the face mask (Figure 4); and turn on the compressor. |
 |
| 6. Breathe as calmly, deeply and evenly as possible through your mouth until no more mist is formed in the nebulizer chamber (about 5-15 minutes). At this point, the treatment is finished. 7. Clean the nebulizer (see manufacturer's instructions). |
Manufactured for: Ingenus Pharmaceuticals, LLC Orlando, FL 32811-7193 Made in China Revised: 08/2024
LXK0042/01 |