Get your patient on Alcaine - Proparacaine Hydrochloride solution/ Drops (Proparacaine Hydrochloride)
Alcaine - Proparacaine Hydrochloride solution/ Drops prescribing information
INDICATIONS AND USAGE:
ALCAINE™ ophthalmic solution is indicated for procedures in which a topical ophthalmic anesthetic is indicated: corneal anesthesia of short duration, e.g. tonometry, gonioscopy, removal of corneal foreign bodies, and for short corneal and conjunctival procedures.
DOSAGE AND ADMINISTRATION:
Usual Dosage: Removal of foreign bodies and sutures, and for tonometry: 1 to 2 drops (in single instillations) in each eye before operating.
Short Corneal and Conjunctival Procedures: 1 drop in each eye every 5 to 10 minutes for 5 to 7 doses.
NOTE: ALCAINE™ (proparacaine hydrochloride ophthalmic solution, USP) 0.5% should be clear to straw-color. If the solution becomes darker, discard the solution.
CONTRAINDICATIONS:
ALCAINE™ ophthalmic solution should be considered contraindicated in patients with known hypersensitivity to any of the ingredients of this preparation.
ADVERSE REACTIONS:
Occasional temporary stinging, burning and conjunctival redness may occur with the use of proparacaine. Corneal edema has been reported following the use of proparacaine. A rare, severe, immediate-type, apparently hyperallergic corneal reaction characterized by acute, intense and diffuse epithelial keratitis, a gray, ground glass appearance, sloughing of large areas of necrotic epithelium, corneal filaments and, sometimes, iritis with descemetitis has been reported.
Allergic contact dermatitis from proparacaine with drying and fissuring of the fingertips has also been reported.
DESCRIPTION:
ALCAINE™ (proparacaine hydrochloride ophthalmic solution, USP) 0.5% is a topical local anesthetic for ophthalmic use. The active ingredient is represented by the structural formula:
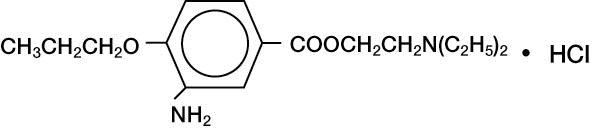
Established Name: Proparacaine Hydrochloride Chemical Name: Benzoic acid, 3-amino-4-propoxy-,2-(diethylamino) ethyl ester, monohydrochloride. Molecular Weight: 330.85 g/mol
Each mL contains of ALCAINE™ (proparacaine hydrochloride ophthalmic solution, USP) 0.5%: Active: proparacaine hydrochloride 5 mg 0.5%. Preservative: benzalkonium chloride (0.01%). Inactives: glycerin and purified water. The pH may be adjusted with hydrochloric acid and/or sodium hydroxide.
CLINICAL PHARMACOLOGY:
ALCAINE™ ophthalmic solution is a rapidly-acting topical anesthetic, with induced anesthesia lasting approximately 10-20 minutes.
HOW SUPPLIED:
ALCAINE™ (proparacaine hydrochloride ophthalmic solution, USP) 0.5% is supplied in DROP-TAINER® dispensers as follows:
15 mL NDC 0998-0016-15
Storage:
Bottle must be stored in unit carton to protect contents from light. Store bottles under refrigeration at 2°C to 8°C (36°F to 46°F).
After opening, ALCAINE™ can be used until the expiration date on the bottle.
NOTE: ALCAINE™ (proparacaine hydrochloride ophthalmic solution, USP) 0.5% should be clear to straw-color. If the solution becomes darker, discard the solution.
Distributed by: ALCON LABORATORIES, INC. Fort Worth, Texas 76134
Revised: 06/2024 Alcon Alcon Laboratories, Inc. 6201 South Freeway Fort Worth, Texas 76134 USA © 2024 Alcon Inc. Printed in USA alcon.medinfo@alcon.com 1-800-757-9195 300063110-0723