Get your patient on Amiloride Hydrochloride - Amiloride Hydrochloride tablet (Amiloride Hydrochloride)
Amiloride Hydrochloride - Amiloride Hydrochloride tablet prescribing information
Like other potassium-conserving agents, amiloride may cause hyperkalemia (serum potassium levels greater than 5.5 mEq per liter) which, if uncorrected, is potentially fatal. Hyperkalemia occurs commonly (about 10%) when amiloride is used without a kaliuretic diuretic. This incidence is greater in patients with renal impairment, diabetes mellitus (with or without recognized renal insufficiency), and in the elderly. When amiloride HCl is used concomitantly with a thiazide diuretic in patients without these complications, the risk of hyperkalemia is reduced to about 1-2 percent. It is thus essential to monitor serum potassium levels carefully in any patient receiving amiloride, particularly when it is first introduced, at the time of diuretic dosage adjustments, and during any illness that could affect renal function.
INDICATIONS AND USAGE
Amiloride HCl is indicated as adjunctive treatment with thiazide diuretics or other kaliuretic-diuretic agents in congestive heart failure or hypertension to:
- help restore normal serum potassium levels in patients who develop hypokalemia on the kaliuretic diuretic
- prevent development of hypokalemia in patients who would be exposed to particular risk if hypokalemia were to develop, e.g., digitalized patients or patients with significant cardiac arrhythmias.
The use of potassium-conserving agents is often unnecessary in patients receiving diuretics for uncomplicated essential hypertension when such patients have a normal diet. Amiloride HCl has little additive diuretic or anti-hypertensive effect when added to a thiazide diuretic.
Amiloride HCl should rarely be used alone. It has weak (compared with thiazides) diuretic and antihypertensive effects. Used as single agents, potassium sparing diuretics, including amiloride HCl, result in an increased risk of hyperkalemia (approximately 10% with amiloride). Amiloride HCl should be used alone only when persistent hypokalemia has been documented and only with careful titration of the dose and close monitoring of serum electrolytes.
DOSAGE AND ADMINISTRATION
Amiloride HCl should be administered with food.
Amiloride HCl, one 5 mg tablet daily, should be added to the usual antihypertensive or diuretic dosage of a kaliuretic diuretic. The dosage may be increased to 10 mg per day, if necessary. More than two 5 mg tablets of amiloride HCl daily usually are not needed, and there is little controlled experience with such doses. If persistent hypokalemia is documented with 10 mg, the dose can be increased to 15 mg, then 20 mg, with careful monitoring of electrolytes.
In treating patients with congestive heart failure after an initial diuresis has been achieved, potassium loss may also decrease and the need for amiloride HCl should be re-evaluated. Dosage adjustment may be necessary. Maintenance therapy may be on an intermittent basis.
If it is necessary to use amiloride HCl alone (See INDICATIONS ), the starting dosage should be one 5 mg tablet daily. This dosage may be increased to 10 mg per day, if necessary. More than two 5 mg tablets usually are not needed, and there is little controlled experience with such doses. If persistent hypokalemia is documented with 10 mg, the dose can be increased to 15 mg, then 20 mg, with careful monitoring of electrolytes.
CONTRAINDICATIONS
Hyperkalemia
Amiloride HCl should not be used in the presence of elevated serum potassium levels (greater than 5.5 mEq per liter).
Antikaliuretic Therapy or Potassium Supplementation
Amiloride HCl should not be given to patients receiving other potassium-conserving agents, such as spironolactone or triamterene. Potassium supplementation in the form of medication, potassium-containing salt substitutes or a potassium-rich diet should not be used with amiloride HCl except in severe and/or refractory cases of hypokalemia. Such concomitant therapy can be associated with rapid increases in serum potassium levels. If potassium supplementation is used, careful monitoring of the serum potassium level is necessary.
Impaired Renal Function
Anuria, acute or chronic renal insufficiency, and evidence of diabetic nephropathy are contraindications to the use of amiloride HCl. Patients with evidence of renal functional impairment (blood urea nitrogen [BUN] levels over 30 mg per 100 mL or serum creatinine levels over 1.5 mg per 100 mL) or diabetes mellitus should not receive the drug without careful, frequent and continuing monitoring of serum electrolytes, creatinine, and BUN levels. Potassium retention associated with the use of an anti-kaliuretic agent is accentuated in the presence of renal impairment and may result in the rapid development of hyperkalemia.
Hypersensitivity
Amiloride HCl is contraindicated in patients who are hypersensitive to this product.
ADVERSE REACTIONS
Amiloride HCl is usually well tolerated and, except for hyperkalemia (serum potassium levels greater than 5.5 mEq per liter — see WARNINGS ), significant adverse effects have been reported infrequently. Minor adverse reactions were reported relatively frequently (about 20%) but the relationship of many of the reports to amiloride HCl is uncertain and the overall frequency was similar in hydrochlorothiazide treated groups. Nausea/anorexia, abdominal pain, flatulence, and mild skin rash have been reported and probably are related to amiloride. Other adverse experiences that have been reported with amiloride are generally those known to be associated with diuresis, or with the underlying disease being treated.
The adverse reactions for amiloride HCl listed in the following table have been arranged into two groups: (1) incidence greater than one percent; and (2) incidence one percent or less. The incidence for group (1) was determined from clinical studies conducted in the United States (837 patients treated with amiloride HCl). The adverse effects listed in group (2) include reports from the same clinical studies and voluntary reports since marketing. The probability of a causal relationship exists between amiloride HCl and these adverse reactions, some of which have been reported only rarely.
Incidence > 1% | Incidence ≤ 1% |
Body as a Whole | |
Weakness Fatigability | Back pain Chest pain Neck/shoulder ache Pain, extremities |
Cardiovascular | |
| Angina pectoris Orthostatic hypotension Arrhythmia Palpitation |
Digestive | |
Diarrhea• Vomiting• Abdominal pain Gas pain Appetite changes Constipation | Jaundice GI bleeding Abdominal fullness GI disturbance Thirst Heartburn Flatulence Dyspepsia |
Metabolic | |
| None |
Skin | |
| Skin rash Itching Dryness of mouth Pruritus Alopecia |
Musculoskeletal | |
| Joint pain Leg ache |
Nervous | |
Encephalopathy | Paresthesia Tremors Vertigo |
Psychiatric | |
| Nervousness Mental confusion Insomnia Decreased libido Depression Somnolence |
Respiratory | |
Dyspnea | Shortness of breath |
Special Senses | |
| Visual disturbances Nasal congestion Tinnitus Increased intraocular pressure |
Urogenital | |
| Polyuria Dysuria Urinary frequency Bladder spasms Gynecomastia |
- •Reactions occurring in 3% to 8% of patients treated with amiloride HCl. (Those reactions occurring in less than 3% of the patients are unmarked.)
- ••See WARNINGS .
Causal Relationship Unknown
Other reactions have been reported but occurred under circumstances where a causal relationship could not be established. However, in these rarely reported events, that possibility cannot be excluded. Therefore, these observations are listed to serve as alerting information to physicians.
Activation of probable pre-existing peptic ulcer
Aplastic anemia
Neutropenia
Abnormal liver function
Drug Interactions
When amiloride HCl is administered concomitantly with an angiotensin-converting enzyme inhibitor, an angiotensin II receptor antagonist, cyclosporine or tacrolimus, the risk of hyperkalemia may be increased. Therefore, if concomitant use of these agents is indicated because of demonstrated hypokalemia, they should be used with caution and with frequent monitoring of serum potassium. (See WARNINGS .)
Lithium generally should not be given with diuretics because they reduce its renal clearance and add a high risk of lithium toxicity. Read circulars for lithium preparations before use of such concomitant therapy.
In some patients, the administration of a non-steroidal anti-inflammatory agent can reduce the diuretic, natriuretic, and antihypertensive effects of loop, potassium-sparing and thiazide diuretics. Therefore, when amiloride HCl and non-steroidal anti-inflammatory agents are used concomitantly, the patient should be observed closely to determine if the desired effect of the diuretic is obtained. Since indomethacin and potassium-sparing diuretics, including amiloride HCl, may each be associated with increased serum potassium levels, the potential effects on potassium kinetics and renal function should be considered when these agents are administered concurrently.
DESCRIPTION
Amiloride HCl, an antikaliuretic-diuretic agent, is a pyrazine-carbonyl-guanidine that is unrelated chemically to other known antikaliuretic or diuretic agents. It is the salt of a moderately strong base (pKa 8.7). It is designated chemically as 3,5-diamino-6-chloro- N -(diaminomethylene) pyrazinecarboxamide monohydrochloride, dihydrate and has a molecular weight of 302.12. Its empirical formula is C 6 H 8 ClN 7 O•HCl•2H 2 O and its structural formula is:
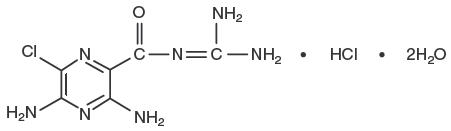
Amiloride HCl is available for oral use as tablets containing 5 mg of anhydrous amiloride HCl. Each tablet contains the following inactive ingredients: calcium phosphate, lactose, magnesium stearate and starch.
CLINICAL PHARMACOLOGY
Amiloride HCl is a potassium-conserving (antikaliuretic) drug that possesses weak (compared with thiazide diuretics) natriuretic, diuretic, and antihypertensive activity. These effects have been partially additive to the effects of thiazide diuretics in some clinical studies. When administered with a thiazide or loop diuretic, amiloride HCl has been shown to decrease the enhanced urinary excretion of magnesium which occurs when a thiazide or loop diuretic is used alone. Amiloride HCl has potassium-conserving activity in patients receiving kaliuretic-diuretic agents.
Amiloride HCl is not an aldosterone antagonist and its effects are seen even in the absence of aldosterone.
Amiloride HCl exerts its potassium sparing effect through the inhibition of sodium reabsorption at the distal convoluted tubule, cortical collecting tubule and collecting duct; this decreases the net negative potential of the tubular lumen and reduces both potassium and hydrogen secretion and their subsequent excretion. This mechanism accounts in large part for the potassium sparing action of amiloride.
Amiloride HCl usually begins to act within 2 hours after an oral dose. Its effect on electrolyte excretion reaches a peak between 6 and 10 hours and lasts about 24 hours. Peak plasma levels are obtained in 3 to 4 hours and the plasma half-life varies from 6 to 9 hours. Effects on electrolytes increase with single doses of amiloride HCl up to approximately 15 mg.
Amiloride HCl is not metabolized by the liver but is excreted unchanged by the kidneys. About 50 percent of a 20 mg dose of amiloride HCl is excreted in the urine and 40 percent in the stool within 72 hours. Amiloride HCl has little effect on glomerular filtration rate or renal blood flow. Because amiloride HCl is not metabolized by the liver, drug accumulation is not anticipated in patients with hepatic dysfunction, but accumulation can occur if the hepatorenal syndrome develops.
HOW SUPPLIED
Amiloride HCl Tablets, 5 mg, are off-white to light yellow, diamond-shaped, compressed tablets, embossed with "P291". They are supplied in bottles of 100 (NDC 0574-0292-01).
STORAGE
Protect from moisture, freezing and excessive heat. Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Amiloride Hydrochloride - Amiloride Hydrochloride tablet PubMed™ news
- Journal Article • 2026 MayAcid Sensing Ion Channel 2a assembles with Epithelial Na(+) Channel β and γ subunits to form mechanosensitive ion channels.
- Journal Article • 2026 MayGenome wide identification, evolution and expression analysis of NHX gene family in cultivated barley (Hordeum vulgare. L) grown under salinity.
- Journal Article • 2026 MayIn silico characterisation of a novel SARS-CoV-2 envelope protein inhibitor and in vitro validation against murine coronavirus.
- Journal Article • 2026 AprMacropinocytosis inhibition attenuates pro-fibrotic responses in lung fibroblasts and pulmonary fibrosis models.
- Journal Article • 2026 AprThe pharmacological rationale for amiloride nasal spray: potentially a portable and rapid acting treatment for panic disorder: commentary, Fagan.