Get your patient on Amphetamine Sulfate - Amphetamine tablet (Amphetamine)
Amphetamine Sulfate - Amphetamine tablet prescribing information
WARNING: ABUSE, MISUSE, AND ADDICTION
Amphetamine sulfate tablets have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including amphetamine sulfate, can result in overdose and death (see OVERDOSAGE ), and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing amphetamine sulfate tablets, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout amphetamine sulfate tablets treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction (see WARNINGS and DRUG ABUSE AND DEPENDENCE ).
INDICATIONS AND USAGE
Amphetamine sulfate tablets, USP 5 mg and 10 mg are indicated for:
1. Narcolepsy
2. Attention Deficit Disorder with Hyperactivity as an integral part of a total treatment program which typically includes other remedial measures (psychological, educational, social) for a stabilizing effect in children with behavioral syndrome characterized by the following group of developmentally inappropriate symptoms: moderate to severe distractibility, short attention span, hyperactivity, emotional lability, and impulsivity. The diagnosis of the syndrome should not be made with finality when these symptoms are only of comparatively recent origin. Nonlocalizing (soft) neurological signs, learning disability, and abnormal EEG may or may not be present, and a diagnosis of central nervous system dysfunction may or not be warranted.
3. Exogenous Obesity as a short term (a few weeks) adjunct in a regimen of weight reduction based on caloric restriction for patients refractory to alternative therapy, e.g., repeated diets, group programs, and other drugs. The limited usefulness of amphetamines (see CLINICAL PHARMACOLOGY ) should be weighed against possible risks inherent in use of the drug, such as those described below.
DOSAGE AND ADMINISTRATION
Regardless of indication, amphetamine should be administered at the lowest effective dosage and dosage should be individually adjusted. Late evening doses should be avoided because of resulting insomnia.
Narcolepsy
Usual dose is 5 to 60 milligrams per day in divided doses depending on the individual patient response.
Narcolepsy seldom occurs in children under 12 years of age; however, when it does, amphetamine sulfate tablets, USP may be used. The suggested initial dose for patients aged 6 to 12 is 5 mg daily; daily dose may be raised in increments of 5 mg at weekly intervals until optimal response obtained. In patients 12 years of age and older, start with 10 mg daily; daily dosage may be raised in increments of 10 mg at weekly intervals until optimal response is obtained. If bothersome adverse reactions appear (e.g., insomnia or anorexia) dosage should be reduced. Give the first dose on awakening; additional doses (5 or 10 mg) at intervals of 4 to 6 hours.
Attention Deficit Disorder with Hyperactivity
Not recommended for children under 3 years of age.
In children from 3 to 5 years of age, start with 2.5 mg daily; daily dosage may be raised in increments of 2.5 mg at weekly intervals until optimal response is obtained.
In children 6 years of age or older, start with 5 mg once or twice daily; daily dosage may be raised in increments of 5 mg at weekly intervals until optimal response is obtained. Only in rare cases will it be necessary to exceed a total of 40 milligrams per day.
With tablets give first dose on awakening; additional doses (1 to 2) at intervals of 4 to 6 hours.
Where possible, drug administration should be interrupted occasionally to determine if there is a recurrence of behavioral symptoms sufficient to require continued therapy.
Prior to treating patients with amphetamine sulfate tablets, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) (see WARNINGS ).
- the family history and clinically evaluate patients for motor or verbal tics or Tourette’s syndrome (see WARNINGS ).
Exogenous Obesity
Usual dosage is up to 30 mg daily, taken in divided doses of 5 mg to 10 mg, 30 to 60 minutes before meals. Not recommended for this use in children under 12 years of age.
CONTRAINDICATIONS
- Known hypersensitivity to amphetamine products.
- During or within 14 days following the administration of monoamine oxidase inhibitors (hypertensive crises may result) (see WARNINGS ).
ADVERSE REACTIONS
Cardiovascular
Palpitations, tachycardia, elevation of blood pressure. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use.
Central Nervous System
Psychotic episodes at recommended doses (rare), overstimulation, restlessness, dizziness, insomnia, euphoria, dyskinesia, dysphoria, tremor, headache, exacerbation of motor and verbal tics and Tourette's syndrome.
Gastrointestinal
Dryness of the mouth, unpleasant taste, diarrhea, constipation, intestinal ischemia and other gastrointestinal disturbances. Anorexia and weight loss may occur as undesirable effects when amphetamines are used for other than the anorectic effect.
Allergic
Urticaria
Endocrine
Impotence, changes in libido, and frequent or prolonged erections.
Musculoskeletal
Rhabdomyolysis
To report SUSPECTED ADVERSE REACTIONS, contact Amneal Pharmaceuticals at 1-877-835-5472 or FDA at 1‑800-FDA-1088 or www.fda.gov/medwatch .Drug Interactions
MAO inhibitors- MAOI antidepressants, as well as a metabolic of furazolidone, slow amphetamine metabolism. This slowing potentiates amphetamines, increasing their effect on the release of norepinephrine and other monoamines from adrenergic nerve endings; this can cause headaches and other signs of hypertensive crisis. A variety of neurological toxic effects and malignant hyperpyrexia can occur, sometimes with fatal results.
Serotonergic Drugs- The concomitant use of amphetamine sulfate tablets and serotonergic drugs increases the risk of serotonin syndrome. Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during amphetamine sulfate tablets initiation or dosage increase. If serotonin syndrome occurs, discontinue amphetamine sulfate tablets and the concomitant serotonergic drug(s) (see WARNINGS and PRECAUTIONS ).
CYP2D6 Inhibitors- The concomitant use of amphetamine sulfate tablets and CYP2D6 inhibitors may increase the exposure of amphetamine sulfate tablets compared to the use of the drug alone and increase the risk of serotonin syndrome. Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during amphetamine sulfate tablets initiation and after a dosage increase. If serotonin syndrome occurs, discontinue amphetamine sulfate tablets and the CYP2D6 inhibitor (see WARNINGS , OVERDOSAGE ). Examples of CYP2D6 inhibitors include paroxetine and fluoxetine (also serotonergic drugs), quinidine, ritonavir.
Acidifying agents - Gastrointestinal acidifying agents (guanethidine, reserpine, glutamic acid HCl, ascorbic acid, fruit juices, etc.) lower absorption of amphetamines. Urinary acidifying agents (ammonium chloride, sodium acid phosphate, etc.) increase concentration of the ionized species of the amphetamine molecule, thereby increasing urinary excretion. Both groups of agents lower blood levels and efficacy of amphetamines.
Adrenergic blockers - Adrenergic blockers are inhibited by amphetamines.
Alkalinizing agents - Gastrointestinal alkalinizing agents (sodium bicarbonate, etc.) increase absorption of amphetamines. Urinary alkalinizing agents (acetazolamide, some thiazides) increase the concentration of the non-ionized species of the amphetamine molecule, thereby decreasing urinary excretion. Both groups of agents increase blood levels and therefore potentiate the action of amphetamines.
Antidepressants tricyclic - Amphetamines may enhance the activity of tricyclic or sympathomimetic agents; d-amphetamine with desipramine or protriptyline and possibly other tricyclics cause striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated.
Antihistamines - Amphetamines may counteract the sedative effect of antihistamines.
Antihypertensives - Amphetamines may antagonize the hypotensive effects of antihypertensives.
Chlorpromazine - Chlorpromazine blocks dopamine and norepinephrine reuptake, thus inhibiting the central stimulant effects of amphetamine, and can be used to treat amphetamine poisoning.
Ethosuximide - Amphetamines may delay intestinal absorption of ethosuximide.
Haloperidol - Haloperidol blocks dopamine and norepinephrine reuptake, thus inhibiting the central stimulant effects of amphetamines.
Lithium carbonate - The antiobesity and stimulatory effects of amphetamines may be inhibited by lithium carbonate.
Meperidine - Amphetamines potentiate the analgesic effect of meperidine.
Methenamine therapy - Urinary excretion of amphetamines is increased, and efficacy is reduced by acidifying agents used in methenamine therapy.
Norepinephrine - Amphetamines enhance the adrenergic effect of norepinephrine.
Phenobarbital - Amphetamines may delay intestinal absorption of phenobarbital. Co-administration of phenobarbital may produce a synergistic anticonvulsant action.
Phenytoin - Amphetamines may delay intestinal absorption of phenytoin; co- administration of phenytoin may produce a synergistic anticonvulsant action.
Propoxyphene - In cases of propoxyphene overdosage, amphetamine CNS stimulation is potentiated and fatal convulsions can occur.
Veratrum alkaloids - Amphetamines inhibit the hypotensive effect of veratrum alkaloids.
DESCRIPTION
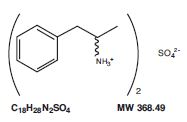
Amphetamine sulfate, USP is a sympathomimetic amino of the amphetamine group. It is a white powder. It has a slightly bitter taste. Its solutions are acid to litmus, having a pH of 5 to 8. It is freely soluble in water, slightly soluble in alcohol and practically insoluble in ether.
Each tablet, for oral administration contains 5 mg or 10 mg of amphetamine sulfate, USP. Each tablet also contains the following inactive ingredients: colloidal silicon dioxide, crospovidone, microcrystalline cellulose, stearic acid, and talc. The 10 mg tablet also contains FD&C Blue #1 aluminum lake.
Structural Formula:
CLINICAL PHARMACOLOGY
Amphetamines are non-catecholamine, sympathomimetic amines with CNS stimulant activity. Peripheral actions include elevations of systolic and diastolic blood pressures, and weak bronchodilator, and respiratory stimulant action.
Amphetamine, as the racemic form, differs from dextroamphetamine in a number of ways. The l-isomer is more potent than the d-isomer in cardiovascular activity, but much less potent in causing CNS excitatory effects. The racemic mixture also is less effective as an appetite suppressant when compared to dextroamphetamine. There is neither specific evidence which clearly establishes the mechanism whereby amphetamines produce mental and behavioral effects in children, nor conclusive evidence regarding how those effects relate to the condition of the central nervous system.
Drugs in this class used in obesity are commonly known as "anorectics" or "anorexigenics." It has not been established, however, that the action of such drugs in treating obesity is primarily one of appetite suppression. Other central nervous system actions or metabolic effects, may be involved, for example. Adult obese subjects instructed in dietary management and treated with "anorectic" drugs lose more weight on the average than those treated with placebo and diet, as determined in relatively short-term clinical trials.
The magnitude of increased weight loss of drug-treated patients over placebo-treated patients is only a fraction of a pound a week. The rate of weight loss is greatest in the first weeks of therapy for both drug and placebo subjects and tends to decrease in succeeding weeks. The origins of the increased weight loss due to the various possible drug effects are not established. The amount of weight loss associated with the use of an "anorectic" drug varies from trial to trial, and the increased weight loss appears to be related in part to variables other than the drug prescribed, such as the physician-investigator, the population treated, and the diet prescribed. Studies do not permit conclusions as to the relative importance of the drug and nondrug factors on weight loss.
The natural history of obesity is measured in years, whereas the studies cited are restricted to few weeks duration; thus, the total impact of drug-induced weight loss over that of diet alone must be considered clinically limited.
HOW SUPPLIED
Amphetamine Sulfate Tablets USP, 5 mg are supplied as white, round biconvex tablets, plain on one side, and ‘5’ with score on the other side.
They are available as follows:
Bottles of 100: NDC 69238-1563-1
Amphetamine Sulfate Tablets USP, 10 mg are supplied as blue, round biconvex tablets, plain on one side, and ‘10’ with double scores on the other side.
They are available as follows:
Bottles of 100: NDC 69238-1564-1
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Dispense in a well-closed container, as defined in the USP.
Manufactured by: Amneal Pharmaceuticals of New York, LLC Brookhaven, NY 11719
Rev. 10-2025-08
Amphetamine Sulfate - Amphetamine tablet PubMed™ news
- Journal Article • 2026 MayStrategies for Substituting ADHD Medications During Stimulant Shortages.
- Journal Article • 2026 MayDrug-induced changes in connectivity to midbrain dopamine cells revealed by rabies monosynaptic tracing.
- Journal Article • 2026 MayModulation of WNT and FGF18 enhances yield and subtype identity of hPSC-derived midbrain dopamine neurons.
- Journal Article • 2026 MaySexualized drug use, anxiety, depression, and suicidal ideation among men who have sex with men in Pakistan: Results of a multi-center cross-sectional survey.
- Journal Article • 2026 MayStimulant Medications and Bone Health.