Get your patient on Aptensio Xr - Methylphenidate Hydrochloride capsule, Extended Release (Methylphenidate Hydrochloride)
Aptensio Xr - Methylphenidate Hydrochloride capsule, Extended Release prescribing information
WARNING: ABUSE, MISUSE, AND ADDICTION
APTENSIO XR has a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including APTENSIO XR, can result in overdose and death [see Overdosage (10) ] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing APTENSIO XR, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout APTENSIO XR treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1) and Drug Abuse and Dependence (9.2) ] .
INDICATIONS AND USAGE
APTENSIO XR is indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD ) in patients 6 years and older [see Clinical Studies (14) ].
Limitations of Use
The use of Aptensio XR is not recommended in pediatric patients younger than 6 years of age because they had higher plasma exposure and a higher incidence of adverse reactions(e.g., weight loss) than patients 6 years and older at the same dosage [see Warnings and Precautions (5.7) , Use in Specific Populations (8.4) ].
DOSAGE AND ADMINISTRATION
- Recommended starting dose for patients 6 years and older: 10 mg once daily with or without food in the morning. Dosage may be increased weekly in increments of 10 mg per day. Daily dosage above 60 mg is not recommended. (2.1 )
- Capsules may be swallowed whole or opened and the entire contents sprinkled onto applesauce. (2.1 )
Pretreatment Screening
Prior to treating patients with APTENSIO XR, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions 5.2 ].
- the family history and clinically evaluate patients for motor or verbal tics or Tourette's syndrome before initiating APTENSIO XR [see Warnings and Precautions (5.10) ] .
Recommended Dosage
The recommended starting dose of APTENSIO XR in patients 6 years and older is 10 mg once daily orally in the morning with or without food. Advise patients to establish a routine pattern with regard to meals. The dose should be individualized according to the needs and response of the patient.
The dose may be titrated weekly in increments of 10 mg. Daily doses above 60 mg have not been studied and are not recommended.
Administration Instructions
APTENSIO XR may be taken whole or the capsule may be opened and the entire contents sprinkled onto applesauce. If the patient is using the sprinkled administration method, the sprinkled applesauce should be consumed immediately; it should not be stored. Patients should take the applesauce with sprinkled beads in its entirety without chewing. The dose of a single capsule should not be divided. The contents of the entire capsule should be taken, and patients should not take anything less than one capsule per day.
Dosage Reduction and Discontinuation
If paradoxical aggravation of symptoms or other adverse reactions occur; the dosage should be reduced, or, if necessary, discontinue APTENSIO XR.
If improvement is not observed after appropriate dosage adjustment over a one-month period, discontinue APTENSIO XR.
DOSAGE FORMS AND STRENGTHS
- 10 mg Extended-Release Capsules – light turquoise blue cap/white body (imprinted with "APTENSIO XR" on cap and "10 mg" on the body)
- 15 mg Extended-Release Capsules – orange cap/white body (imprinted with "APTENSIO XR" on cap and "15 mg" on the body)
- 20 mg Extended-Release Capsules – yellow cap/white body (imprinted with "APTENSIO XR" on cap and "20 mg" on the body)
- 30 mg Extended-Release Capsules – blue violet cap/white body (imprinted with "APTENSIO XR" on cap and "30 mg" on the body)
- 40 mg Extended-Release Capsules – pink cap/white body (imprinted with "APTENSIO XR" on cap and "40 mg" on the body)
- 50 mg Extended-Release Capsules – green cap/white body (imprinted with "APTENSIO XR" on cap and "50 mg" on the body)
- 60 mg Extended-Release Capsules – gray cap/white body (imprinted with "APTENSIO XR" on cap and "60 mg" on the body)
USE IN SPECIFIC POPULATIONS
Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to APTENSIO XR during pregnancy. Healthcare providers are encouraged to register patients by calling the National Pregnancy Registry for Psychostimulants at 1-866-961-2388.
Risk Summary
Limited published studies report on the use of methylphenidate in pregnant women; however, the data are insufficient to inform any drug-associated risks. No effects on morphological development were observed in embryo-fetal development studies with oral administration of methylphenidate to pregnant rats and rabbits during organogenesis at doses up to 10 and 15 times, respectively, the maximum recommended human dose (MRHD) of 60 mg/day given to adolescents on a mg/m 2 basis. However, spina bifida was observed in rabbits at a dose 52 times the MRHD given to adolescents. A decrease in pup body weight was observed in a pre- and post-natal development study with oral administration of methylphenidate to rats throughout pregnancy and lactation at the highest dose of 60 mg/kg/day (6 times the MRHD given to adolescents) [see Data ]. The background risk of major birth defects and miscarriage for the indicated population are unknown. However, the background risk in the U.S. general population of major birth defects is 2% to 4% and of miscarriage is 15% to 20% of clinically recognized pregnancies.
Clinical Considerations
Fetal/Neonatal adverse reactions
CNS stimulants, such as APTENSIO XR, can cause vasoconstriction and thereby decrease placental perfusion. No fetal and/or neonatal adverse reactions have been reported with the use of therapeutic doses of methylphenidate during pregnancy; however, premature delivery and low birth weight infants have been reported in amphetamine-dependent mothers.
Data
Animal Data
In embryo-fetal development studies conducted in rats and rabbits, methylphenidate was administered orally at doses of up to 75 and 200 mg/kg/day, respectively, during the period of organogenesis. Malformations (increased incidence of fetal spina bifida) were observed in rabbits at the highest dose, which is approximately 52 times the maximum recommended human dose (MRHD) of 60 mg/day given to adolescents on a mg/m 2 basis. The no effect level for embryo-fetal development in rabbits was 60 mg/kg/day (15 times the MRHD given to adolescents on a mg/m 2 basis). There was no evidence of morphological development effects in rats, although increased incidences of fetal skeletal variations were seen at the highest dose level (10 times the MRHD of 60 mg/day given to adolescents on a mg/m 2 basis), which was also maternally toxic. The no effect level for embryo-fetal development in rats was 25 mg/kg/day (2 times the MRHD on a mg/m 2 basis). When methylphenidate was administered to rats throughout pregnancy and lactation at doses of up to 45 mg/kg/day, offspring body weight gain was decreased at the highest dose (6 times the MRHD of 60 mg/day given to adolescents on a mg/m 2 basis), but no other effects on postnatal development were observed. The no effect level for pre- and postnatal development in rats was 15 mg/kg/day (1.5 times the MRHD given to adolescents on a mg/m 2 basis).
Lactation
Risk Summary
Limited published literature, based on breast milk sampling from five mothers, reports that methylphenidate is present in human milk, which resulted in infant doses of 0.16% to 0.7% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 1.1 and 2.7. There are no reports of adverse effects on the breastfed infant and no effects on milk production. However, long-term neurodevelopmental effects on infants from stimulant exposure are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for APTENSIO XR and any potential adverse effects on the breastfed infant from APTENSIO XR or from the underlying maternal condition.
Clinical Considerations
Monitor breastfeeding infants for adverse reactions, such as agitation, anorexia, and reduced weight gain.
Pediatric Use
The safety and effectiveness of APTENSIO XR have not been established in pediatric patients below the age of 6 years.
Safety and efficacy of APTENSIO XR were evaluated in a multicenter, placebo-controlled, double-blind, parallel group study in 119 children 4 to <6 years of age with ADHD followed by a 12-month open-label extension in 44 of these children. In these studies, patients experienced high rates of adverse reactions, most notably weight loss. Comparing weights prior to initiation of APTENSIO XR (in the safety and efficacy study) to weights after 12 months of treatment (in the open-label extension), 20 of 39 patients with data (50%) had lost enough weight to decrease 10 or more percentiles on a Centers for Disease Control growth chart for weight. In addition, systemic drug exposures in patients 4 to <6 years of age were higher than those observed in older children and adolescents at the same dose (2 to 3 fold higher C max and AUC). Therefore, the benefits of APTENSIO XR do not outweigh the risks in pediatric patients 4 to <6 years of age.
The safety and effectiveness of APTENSIO XR have been established in pediatric patients ages 6 to 17 years in two adequate and well-controlled clinical trials [see Clinical Studies (14) ] .
Long Term Suppression of Growth
Growth should be monitored during treatment with stimulants, including APTENSIO XR. Pediatric patients who are not growing or gaining weight as expected may need to have their treatment interrupted [see Warnings and Precautions (5.7) ] .
Juvenile Animal Toxicity Data
Rats treated with methylphenidate early in the postnatal period through sexual maturation demonstrated a decrease in spontaneous locomotor activity in adulthood. A deficit in acquisition of a specific learning task was observed in females only. The doses at which these findings were observed are at least 6 times the maximum recommended human dose (MRHD) of 60 mg/day given to children on a mg/m 2 basis.
In the study conducted in young rats, methylphenidate was administered orally at doses of up to 100 mg/kg/day for 9 weeks, starting early in the postnatal period (postnatal day 7) and continuing through sexual maturity (postnatal week 10). When these animals were tested as adults (postnatal weeks 13-14), decreased spontaneous locomotor activity was observed in males and females previously treated with 50 mg/kg/day (approximately 6 times the MRHD of 60 mg/day given to children on a mg/m 2 basis) or greater, and a deficit in the acquisition of a specific learning task was observed in females exposed to the highest dose (8 times the MRHD given to children on a mg/m 2 basis). The no effect level for juvenile neurobehavioral development in rats was 5 mg/kg/day (approximately 0.5 times the MRHD given to children on a mg/m 2 basis). The clinical significance of the long-term behavioral effects observed in rats is unknown.
Geriatric Use
Clinical trials of APTENSIO XR did not include any patients aged 65 years and over. In general, dose selection for an elderly patient start at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
CONTRAINDICATIONS
- Hypersensitivity to methylphenidate or other components of the product. Hypersensitivity reactions such as angioedema and anaphylactic reactions have been reported in patients treated with methylphenidate products [see Adverse Reactions (6.1) ].
- Concomitant treatment with monoamine oxidase inhibitors, and also within 14 days following discontinuation of treatment with a monoamine oxidase inhibitor, because of the risk of hypertensive crisis [see Drug Interactions (7.1) ] .
WARNINGS AND PRECAUTIONS
- Risks to Patients with Serious Cardiac Disease: Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmias, coronary artery disease, or other serious cardiac disease. (5.2 )
- Increased Blood Pressure and Heart Rate: Monitor blood pressure and pulse. (5.3 )
- Psychiatric Adverse Reactions: Prior to initiating APTENSIO XR, screen patients for risk factors for developing a manic episode. If new psychotic or manic symptoms occur, consider discontinuing APTENSIO XR. (5.4 )
- Priapism: If abnormally sustained or frequent and painful erections occur, patients should seek immediate medical attention. (5.5 )
- Peripheral Vasculopathy, including Raynaud's Phenomenon: Careful observation for digital changes is necessary during APTENSION XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for patients who develop signs or symptoms of peripheral vasculopathy. (5.6 )
- Long-Term Suppression of Growth in Pediatric Patients: Closely monitor growth (height and weight) in pediatric patients. Pediatric patients not growing or gaining height or weight as expected may need to have their treatment interrupted. (5.7 )
- Acute Angle Closure Glaucoma: APTENSIO XR-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist. (5.8 )
- Increased Intraocular Pressure (IOP) and Glaucoma: Prescribe APTENSIO XR to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor patients with a history of increased IOP or open angle glaucoma. (5.9 )
- Motor and Verbal Tics, and Worsening of Tourette's Syndrome: Before initiating APTENSIO XR, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor patients for the emergence or worsening of tics or Tourette's syndrome. Discontinue treatment if clinically appropriate. (5.10 )
Abuse, Misuse, and Addiction
APTENSIO XR has a high potential for abuse and misuse. The use of APTENSIO XR exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. APTENSIO XR can be diverted for non-medical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2) ]. Misuse and abuse of CNS stimulants, including APTENSIO XR, can result in overdose and death [see Overdosage (10) ] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing APTENSIO XR, assess each patient's risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store APTENSIO XR in a safe place, preferably locked, and instruct patients to not give APTENSIO XR to anyone else. Throughout APTENSIO XR treatment, reassess each patient's risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
Risks to Patients with Serious Cardiac Disease
Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosage.
Avoid APTENSIO XR use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart arrhythmia, coronary artery disease, or other serious cardiac disease.
Increased Blood Pressure and Heart Rate
CNS stimulants cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 bpm). Some patients may have larger increases.
Monitor all APTENSIO XR-treated patients for hypertension and tachycardia.
Psychiatric Adverse Reactions
Exacerbation of Pre-Existing Psychosis
CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a pre-existing psychotic disorder.
Induction of a Manic Episode in Patients with Bipolar Disorder
CNS stimulants may induce a manic or mixed episode in patients. Prior to initiating APTENSIO XR treatment, screen patients for risk factors for developing a manic episode (e.g., comorbid or history of depressive symptoms or a family history of suicide, bipolar disorder, or depression).
New Psychotic or Manic Symptoms
CNS stimulants, at the recommended dosage, may cause psychotic or manic symptoms (e.g., hallucinations, delusional thinking, or mania) in patients without a prior history of psychotic illness or mania. In a pooled analysis of multiple short-term, placebo-controlled studies of CNS stimulants, psychotic or manic symptoms occurred in approximately 0.1% of CNS stimulant-treated patients, compared to 0% of placebo-treated patients. If such symptoms occur, consider discontinuing APTENSIO XR.
Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate use, in both adult and pediatric male patients. Although priapism was not reported with methylphenidate initiation, it developed after some time on methylphenidate, often subsequent to an increase in dosage. Priapism also occured during methylphenidate withdrawal (drug holidays or during discontinuation).
APTENSIO XR patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
Peripheral Vasculopathy, including Raynaud's Phenomenon
CNS stimulants, including APTENSIO XR, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports and at the therapeutic dosages of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during APTENSIO XR treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for APTENSIO XR-treated patients who develop signs or symptoms of peripheral vasculopathy.
Long-Term Suppression of Growth in Pediatric Patients
APTENSIO XR is not approved for use and is not recommended in pediatric patients below 6 years of age [see Use in Specific Populations (8.4) ] .
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in pediatric patients ages 7 to 10 years who were randomized to either methylphenidate or non-medication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and non-medication treated pediatric patients over 36 months (to the ages of 10 to 13 years), suggests that pediatric patients who received methylphenidate for 7 days per week throughout the year had a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this development period.
Closely monitor growth (weight and height) in APTENSIO XR-treated pediatric patients. Pediatric patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
Acute Angle Closure Glaucoma
There have been reports of angle closure glaucoma associated with methylphenidate treatment.
Although the mechanism is not clear, APTENSIO XR-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
Increased Intraocular Pressure and Glaucoma
There have been reports of an elevation of intraocular pressure (IOP) associated with methylphenidate treatment [see Adverse Reactions (6.2) ] .
Prescribe APTENSIO XR to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor APTENSIO XR-treated patients with a history of abnormally increased IOP or open angle glaucoma.
Motor and Verbal Tics, and Worsening of Tourette's Syndrome
CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics. Worsening of Tourette's syndrome has also been reported [see Adverse Reactions (6.2) ] .
Before initiating APTENSIO XR, assess the family history and clinically evaluate patients for tics or Tourette's syndrome. Regularly monitor APTENSIO XR-treated patients for the emergence or worsening of tics or Tourette's syndrome, and discontinue treatment if clinically appropriate.
ADVERSE REACTIONS
The following are discussed in more detail in other sections of the labeling:
- Abuse, Misuse, and Addiction [see Boxed Warning , Warnings and Precautions (5.1) , and Drug Abuse and Dependence (9.2 , 9.3) ]
- Hypersensitivity to Methylphenidate [see Contraindications (4) ]
- Hypertensive Crisis with Concomitant Use of Monoamine Oxidase Inhibitors [see Contraindications (4) and Drug Interactions (7.1) ]
- Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2) ]
- Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3) ]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4) ]
- Priapism [see Warnings and Precautions (5.5) ]
- Peripheral Vasculopathy, including Raynaud's Phenomenon [see Warnings and Precautions (5.6) ]
- Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.7) ]
- Acute Angle Closure Glaucoma [see Warnings and Precautions (5.8) ]
- Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.9) ]
- Motor and Verbal Tics, and Worsening of Tourette's Syndrome [see Warnings and Precautions (5.10) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Adverse Reactions in Studies with Other Methylphenidate Products in Children, Adolescents, and Adults with ADHD
Commonly reported (≥2% of the methylphenidate group and at least twice the rate of the placebo group) adverse reactions from placebo-controlled trials of methylphenidate products include: decreased appetite, decreased weight, nausea, abdominal pain, dyspepsia, dry mouth, vomiting, insomnia, anxiety, nervousness, restlessness, affect lability, agitation, irritability, dizziness, vertigo, tremor, blurred vision, increased blood pressure, increased heart rate, tachycardia, palpitations, hyperhidrosis, and pyrexia.
Adverse Reactions in Studies with APTENSIO XR in Pediatric Patients with ADHD
The safety data in this section is based on data from two one-week controlled clinical studies of APTENSIO XR in pediatric patients with ADHD, one in children ages 6 to 12 years (RP-BP-EF001, hereafter "Study 1"), and one in children and adolescents ages 6 to 17 years (RP-BP-EF002, hereafter "Study 2").
Two APTENSIO XR clinical studies evaluated a total of 256 patients with ADHD. Two hundred and forty-three (243) patients participated in the double-blind phase of these two clinical studies.
Study 1 was a randomized, double-blind, single center, placebo-controlled, flexible-dose, cross-over study to evaluate the time of onset, duration of efficacy, tolerability and safety of APTENSIO XR 15 mg, 20 mg, 30 mg, or 40 mg administered for one week in 26 pediatric patients aged 6 to 12 years who met DSM-IV criteria for ADHD [see Clinical Studies (14) ] .
Most Common Adverse Reactions (incidence of ≥ 5% and at a rate at least twice placebo): abdominal pain, pyrexia and headache.
Adverse Reactions Leading to Discontinuation: No subjects discontinued due to adverse reactions during the double-blind phase of this study.
Study 2 was a randomized, double-blind, multicenter, placebo-controlled, parallel group, fixed-dose study of 10 mg, 15 mg, 20 mg, and 40 mg of APTENSIO XR administered for one week in 221 pediatric patients (6 to 17 years of age) who met DSM-IV criteria for ADHD [see Clinical Studies (14) ] .
Most Common Adverse Reactions (incidence of ≥ 5% and at a rate of at least twice placebo): abdominal pain, decreased appetite, headache and insomnia.
Adverse Reactions Leading to Discontinuation: Two patients (4.4%) in the APTENSIO XR 40 mg group discontinued due to insomnia, nausea and rapid heart rate, respectively during the double-blind phase of the study.
| System Organ Class Adverse Reaction | Aptensio XR (n=183) | Placebo (n=47) |
|---|---|---|
| Nervous System Disorders | ||
| Headache | 10.9% | 8.5% |
| Insomnia | 9.8% | 2.1% |
| Dizziness | 2.2% | 2.1% |
| Gastrointestinal Disorders | ||
| Abdominal pain upper | 8.2% | 0% |
| Nausea | 3.8% | 2.1% |
| Vomiting | 3.8% | 0% |
| Metabolism and Nutritional | ||
| Decreased Appetite | 4.9% | 0% |
Post-Marketing Experience
The following adverse reactions have been identified during post approval use of methylphenidate products. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These adverse reactions are as follows:
Blood and Lymphatic System Disorders : Pancytopenia, Thrombocytopenia, Thrombocytopenic purpura
Cardiac Disorders : Angina pectoris, Bradycardia, Extrasystole, Supraventricular tachycardia, Ventricular extrasystole
Eye Disorders : Diplopia, Increased intraocular pressure, Mydriasis, Visual impairment
General Disorders : Chest pain, Chest discomfort, Hyperpyrexia
Immune System Disorders : Hypersensitivity reactions such as Angioedema, Anaphylactic reactions, Auricular swelling, Bullous conditions, Exfoliative conditions, Urticarias, Pruritus NEC, Rashes, Eruptions, and Exanthems NEC
Investigations : Alkaline phosphatase increased, Bilirubin increased, Hepatic enzyme increased, Platelet count decreased, White blood cell count abnormal, severe hepatic injury
Musculoskeletal, Connective Tissue and Bone Disorders : Arthralgia, Myalgia, Muscle twitching, Rhabdomyolysis
Nervous System : Convulsion, Grand mal convulsion, Dyskinesia, serotonin syndrome in combination with serotonergic drugs, Motor and Verbal Tics
Psychiatric Disorders : Disorientation, Libido changes
Skin and Subcutaneous Tissue Disorders : Alopecia, Erythema
DRUG INTERACTIONS
Antihypertensive Drugs: Monitor blood pressure. Adjust dosage of antihypertensive drug as needed. (7 )
Clinically Important Interactions with APTENSIO XR
Monoamine Oxidase Inhibitors (MAOIs)
Do not administer APTENSIO XR concomitantly or within 14 days after discontinuing MAOI treatment. Concomitant use of MAOIs and CNS stimulants can cause hypertensive crisis. Potential outcomes include death, stroke, myocardial infarction, aortic dissection, ophthalmological complications, eclampsia, pulmonary edema, and renal failure [see Contraindications (4) ].
Antihypertensive Drugs
Aptensio XR may decrease the effectiveness of drugs used to treat hypertension. Monitor blood pressure and adjust the dosage of the antihypertensive drug as needed [see Warnings and Precautions (5.3) ] .
Halogenated Anesthetics
Concomitant use of halogenated anesthetics and APTENSIO XR may increase the risk of sudden blood pressure and heart rate increase during surgery. Avoid use of APTENSIO XR in patients being treated with anesthetics on the day of surgery.
Risperidone
Combined use of methylphenidate with risperidone when there is a change, whether an increase or decrease, in dosage of either or both medications, may increase the risk of extrapyramidal symptoms (EPS). Monitor for signs of EPS.
DESCRIPTION
APTENSIO XR contains methylphenidate hydrochloride, a central nervous system (CNS) stimulant. APTENSIO XR capsules contain multi layered beads, which are composed of an immediate-release layer which contains approximately 40% of the methylphenidate dose, and a controlled release layer which contains approximately 60% of the methylphenidate dose. APTENSIO XR is available in seven capsule strengths. Each extended-release capsule for once-a-day oral administration contains 10 mg, 15 mg, 20 mg, 30 mg, 40 mg, 50 mg, or 60 mg of methylphenidate HCl USP, which is equivalent to 8.6 mg, 13.0 mg, 17.3 mg, 25.9 mg, 34.6 mg, 43.2 mg, or 51.9 mg of methylphenidate free base, respectively. Chemically, methylphenidate HCl is d,l (racemic) methyl α-phenyl-2-piperidineacetate hydrochloride. Its molecular formula is C 14 H 19 NO 2 ∙HCl. Its structural formula is:
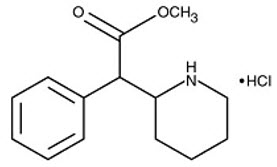
Methylphenidate hydrochloride USP is a white to off-white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone. Its molecular weight is 269.77.
Inactive Ingredients:ammonio methacrylate copolymer, type B; colloidal silicon dioxide (added if necessary); gelatin; hypromelloses; methacrylic acid copolymer, type C; polyethylene glycol; sugar spheres; talc; titanium oxide; and triethyl citrate.
Each strength capsule also contains colorant ingredients in the capsule shell as follows:
10 mg: FD&C Blue No. 1
15 mg: D&C Red No. 28, D&C Yellow No. 10, FD&C Red No. 40
20 mg: D&C Red No. 33, D&C Yellow No. 10
30 mg: FD&C Blue No. 1, FD&C Red No. 3
40 mg: D&C Red No. 28, FD&C Blue No. 1, FD&C Red No. 40
50 mg: D&C Yellow No. 10, FD&C Green No. 3
60 mg: Black Iron Oxide
CLINICAL PHARMACOLOGY
Mechanism of Action
Methylphenidate HCl is a central nervous system (CNS) stimulant. The mode of therapeutic action in ADHD is not known.
Pharmacodynamics
Methylphenidate is a racemic mixture comprised of the d- and l- isomers. The d- isomer is more pharmacologically active than the l- isomer. Methylphenidate blocks the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
Pharmacokinetics
Absorption
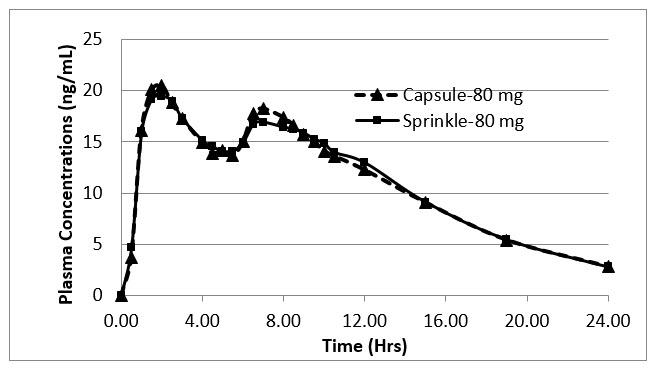
Following oral administration of APTENSIO XR in adults, plasma methylphenidate concentrations increase rapidly, reaching an initial maximum at about 2 hours, followed by gradual descending concentrations over the next 4 to 6 hours, after which a gradual increase begins, reaching a second peak at approximately 8 hours (Figure 1). The relative bioavailability of APTENSIO XR given once daily as compared to a methylphenidate immediate-release oral product given three times daily in adults is comparable. The relative bioavailability is 102%.
The pharmacokinetic profiles and parameters of methylphenidate are similar when APTENSIO XR is administered either as a whole capsule or sprinkled onto applesauce in subjects under fasting conditions (see Table 2 and Figure 1 ).
| Pharmacokinetic Parameters | Capsule | Sprinkle |
|---|---|---|
| C max C max , AUC (0-t) AUC (0-inf) presented as mean ± SD (ng/mL) | 23.47 ± 11.4 | 21.78 ± 9.5 |
| AUC (0-t) (ng.hr/mL) | 262.7 ± 135 | 262.9 ± 128 |
| AUC (0-inf ) (ng.hr/mL) | 258.1 ± 94.2 | 258.0 ± 84.4 |
| T max (hr) data presented as median (range) | 2.0 | 2.0 |
| Half-life (hr) | 5.09 | 5.43 |
| Relative bioavailability | 102% | 101% |
Figure 1: Mean d,l -Methylphenidate Plasma Concentration-Time Profiles following 80 mg Administered as Capsule and Sprinkle Dose in Healthy Adults
Metabolism and Excretion
In humans, methylphenidate is metabolized primarily via deesterification to alpha-phenyl-piperidine acetic acid (PPAA). The metabolite has little or no pharmacologic activity.
After oral dosing of radiolabeled methylphenidate in humans, about 90% of the radioactivity was recovered in urine. The main urinary metabolite was PPAA, accounting for approximately 80% of the dose.
Food Effects
Administration of APTENSIO XR with high fat meal showed a decreased or diminished second peak. A high-fat meal also increased the average C max of methylphenidate by about 28% and the AUC by about 19%. In the clinical trials of APTENSIO XR, it was administered without regard to meals.
Alcohol Effect
At an alcohol concentration up to 40%, there was 96% release of methylphenidate from APTENSIO XR 80 mg capsule within two hours. The results with the 80 mg capsule are considered to be representative of the other available capsules strengths.
Studies in Specific Populations
Gender
There is insufficient experience with the use of APTENSIO XR to detect gender variations in pharmacokinetics.
Race
There is insufficient experience with the use of APTENSIO XR to detect ethnic variations in pharmacokinetics.
Age
The pharmacokinetics of methylphenidate after APTENSIO XR administration was studied in pediatric patients with ADHD between 6 and 12 years of age. Following administration of APTENSIO XR, the bi-phasic plasma methylphenidate concentration profile was qualitatively similar in healthy adult volunteers and pediatric patients with ADHD. The bi-phasic profile in both groups is characterized by an early peak due to rapid absorption of the immediate-release component followed by a delayed, secondary peak due to the controlled-release component of APTENSIO XR.
Renal Insufficiency
There is no experience with the use of APTENSIO XR in patients with renal insufficiency. After oral administration of radiolabeled methylphenidate in humans, methylphenidate was extensively metabolized and approximately 80% of the radioactivity was excreted in the urine in the form of ritalinic acid metabolite. Since renal clearance is not an important route of methylphenidate clearance, renal insufficiency is expected to have little effect on the pharmacokinetics of APTENSIO XR.
Hepatic Insufficiency
There is no experience with the use of APTENSIO XR in patients with hepatic insufficiency.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a lifetime carcinogenicity study carried out in B6C3F1 mice, methylphenidate caused an increase in hepatocellular adenomas and, in males only, an increase in hepatoblastomas, at a daily dose of approximately 60 mg/kg/day. This dose is approximately 2 times the maximum recommended human dose (MRHD) of 60 mg/day given to children on a mg/m 2 basis. Hepatoblastoma is a relatively rare rodent malignant tumor type. There was no increase in total malignant hepatic tumors. The mouse strain used is sensitive to the development of hepatic tumors, and the significance of these results to humans is unknown.
Methylphenidate did not cause any increase in tumors in a lifetime carcinogenicity study carried out in F344 rats; the highest dose used was approximately 45 mg/kg/day, which is approximately 4 times the MRHD (children) on a mg/m 2 basis.
Mutagenesis
Methylphenidate was not mutagenic in the in vitro Ames reverse mutation assay or in the in vitro mouse lymphoma cell forward mutation assay. Sister chromatid exchanges and chromosome aberrations were increased, indicative of a weak clastogenic response, in an in vitro assay in cultured Chinese Hamster Ovary (CHO) cells. Methylphenidate was negative in vivo in males and females in the mouse bone marrow micronucleus assay.
Impairment of Fertility
Methylphenidate did not impair fertility in male or female mice that were fed diets containing the drug in an 18-week Continuous Breeding study. The study was conducted at doses of up to 160 mg/kg/day, approximately 10 times the maximum recommended human dose of 60 mg/day given to adolescents on a mg/m 2 basis.
CLINICAL STUDIES
The efficacy of APTENSIO XR for the treatment of ADHD was established in a randomized, double-blind, single center, placebo-controlled, flexible-dose, cross-over trial in pediatric patients aged 6 to 12 years and a second randomized, double-blind, multicenter, placebo-controlled, fixed–dose trial in pediatric patients 6 to 17 years.
Pediatric Patients
A randomized, double-blind, placebo-controlled, flexible-dose, cross-over, analog classroom study (Study 1) was conducted in pediatric patients ages 6 to 12 years (N=26) who met DSM-IV-TR criteria for ADHD inattentive, hyperactive-impulsive or combined inattentive/hyperactive-impulsive subtypes.
Following a 2 to 4 week open-label dose optimization phase in which patients received flexible-dose APTENSIO XR 15 mg, 20 mg, 30 mg, or 40 mg administered once daily in the morning, patients were randomly assigned to APTENSIO XR (dose from open-label phase) or placebo. After 1-week of treatment, patients were evaluated over a period of 12 hours. Subsequently, patients were given the opposite treatment for 1-week and returned for the second evaluation. Patients could then enter an open-label extension phase for up to 21 months.
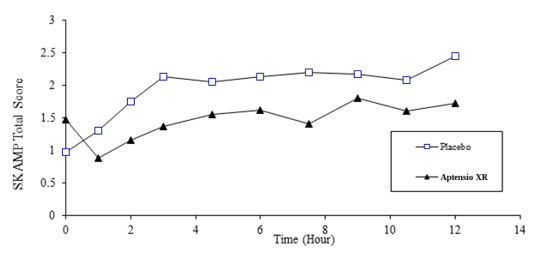
Efficacy assessments were conducted at 1, 2, 3, 4.5, 6, 7.5, 9, 10.5 and 12 hours post-dose using the Swanson, Kotkin, Agler, M. Flynn, and Pelham Total score (SKAMP). The primary efficacy endpoint was the average SKAMP Total Score, comparing APTENSIO XR to placebo. SKAMP is a validated 13-item teacher-rated scale that assesses manifestations of ADHD in a classroom setting.
The SKAMP Total Scores were statistically significantly better (lower) for APTENSIO XR than for placebo at the test day average and at all time points (1, 2, 3, 4.5, 6, 7.5, 9, 10.5 and 12 hours) post-dosing (see Figure 2 ).
Figure 2: Absolute SKAMP- Total Score after treatment with APTENSIO XR or Placebo (Study 1).
A randomized, double-blind, multicenter, placebo-controlled, parallel-group, fixed-dose study (Study 2) was conducted in pediatric patients age 6 to 17 years (N=230) who met DSM-IV-TR criteria for ADHD inattentive, hyperactive-impulsive or combined inattentive/hyperactive-impulsive subtypes.
The ADHD-RS-IV is an 18-item questionnaire with a score range of 0 to 54 points that measures the core symptoms of ADHD and includes both hyperactive/impulsive and inattentive subscales.
Patients were randomized to a daily morning dose of APTENSIO XR 10 mg, 15 mg, 20 mg, or 40 mg, or placebo for 1 week. An 11-week open label phase followed the double-blind phase. Patients could then enter another open-label phase for up to 21 months.
The primary efficacy endpoint was the mean decrease from baseline to the end of Week 1 in the ADHD-RS-IV Total Score. Each of the four APTENSIO XR doses (10 mg, 15 mg, 20 mg, and 40 mg/day) was compared to placebo at the end of week 1. For both the 20 mg/day and the 40 mg/day doses, APTENSIO XR was superior to placebo in reduction of the ADHD-RS-IV Total Score, but not for the 10 mg/day or the 15 mg/day doses.
A total of 221 patients completed the 1-week double-blind phase. Among those, 200 (90.5%) completed the 11-week open label phase and 173 (86.5%) patients continued into the 21-month open-label extension phase.
| Study Number | Treatment Group | Primary Efficacy Measure: ADHD-RS-IV Total Score | ||
|---|---|---|---|---|
| Mean Baseline Score (SD) | LS Mean Reduction from Baseline (SE) | Placebo-subtracted Difference Difference (placebo minus drug) in least-squares mean change from baseline. Positive numbers indicate reduction (improvement). (95% CI) | ||
| Note: SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: confidence interval, not adjusted for multiple comparisons. | ||||
| Study 2 (Pediatric) | APTENSIO XR 10 mg/day | 37.6 (8.32) | 9.1 (1.40) | 3.7 (-0.31, 7.66) |
| APTENSIO XR 15 mg/day | 38.0 (8.64) | 10.3 (1.59) | 4.9 (0.63, 9.07) | |
| APTENSIO XR 20 mg/day Doses that are demonstrated to be effective. | 36.2 (8.46) | 11.4 (1.49) | 6.0 (1.92, 10.02) | |
| APTENSIO XR 40 mg/day | 35.6 (9.16) | 12.8 (1.49) | 7.4 (3.38, 11.45) | |
| Placebo | 33.4 (11.01) | 5.4 (1.48) | -- | |
HOW SUPPLIED/STORAGE AND HANDLING
APTENSIO XR (methylphenidate hydrochloride extended-release) capsules are available as follows:
10 mg Capsules – light turquoise blue cap/white body, (imprinted with "APTENSIO XR" on cap and "10 mg" on the body)
| Bottles of 90 | NDC 42858-401-45 |
15 mg Capsules – orange cap/white body, (imprinted with "APTENSIO XR" on cap and "15 mg" on the body)
| Bottles of 90 | NDC 42858-402-45 |
20 mg Capsules – yellow cap/white body, (imprinted with "APTENSIO XR" on cap and "20 mg" on the body)
| Bottles of 90 | NDC 42858-403-45 |
30 mg Capsules – blue violet cap/white body, (imprinted with "APTENSIO XR" on cap and "30 mg" on the body)
| Bottles of 90 | NDC 42858-404-45 |
40 mg Capsules – pink cap/white body, (imprinted with "APTENSIO XR" on cap and "40 mg" on the body)
| Bottles of 90 | NDC 42858-405-45 |
50 mg Capsules – green cap/white body, (imprinted with "APTENSIO XR" on cap and "50 mg" on the body)
| Bottles of 90 | NDC 42858-406-45 |
60 mg Capsules – gray cap/white body, (imprinted with "APTENSIO XR" on cap and "60 mg" on the body)
| Bottles of 90 | NDC 42858-407-45 |
Storage and Handling
APTENSIO XR (methylphenidate hydrochloride extended-release) capsules should be stored at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Dispense in tight container (USP).
Mechanism of Action
Methylphenidate HCl is a central nervous system (CNS) stimulant. The mode of therapeutic action in ADHD is not known.
Aptensio Xr - Methylphenidate Hydrochloride capsule, Extended Release PubMed™ news
- Journal Article • 2026 MayStrategies for Substituting ADHD Medications During Stimulant Shortages.
- Journal Article • 2026 MayMethodological considerations in the combined methylphenidate and physical activity trial for cancer-related fatigue in immunotherapy patients.
- Journal Article • 2026 MayMethylphenidate and manic relapse in bipolar disorders: A replication study in a nationwide claims database.
- Journal Article • 2026 MayPrescribing medications for attention-deficit/hyperactivity disorder in children: a retrospective analysis of a Japanese health insurance claims database.
- Journal Article • 2026 MayStimulant Medications and Bone Health.