Get your patient on Armlupeg (Pegfilgrastim-Unne)
Armlupeg prescribing information
1 INDICATIONS AND USAGE
Armlupeg is a leukocyte growth factor indicated to
- Decrease the incidence of infection, as manifested by febrile neutropenia, in patients with non-myeloid malignancies receiving myelosuppressive anti-cancer drugs associated with a clinically significant incidence of febrile neutropenia. (1.1)
- Increase survival in patients acutely exposed to myelosuppressive doses of radiation (Hematopoietic Subsyndrome of Acute Radiation Syndrome). (1.2)
Limitations of Use
Armlupeg is not indicated for the mobilization of peripheral blood progenitor cells for hematopoietic stem cell transplantation.
Patients with Cancer Receiving Myelosuppressive Chemotherapy
Armlupeg is indicated in adults and pediatric patients aged newborn and older to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with non-myeloid malignancies receiving myelosuppressive anti-cancer drugs associated with a clinically significant incidence of febrile neutropenia [see Clinical Studies (14.1)].
Limitations of Use
Armlupeg is not indicated for the mobilization of peripheral blood progenitor cells for hematopoietic stem cell transplantation.
Patients with Hematopoietic Subsyndrome of Acute Radiation Syndrome
Armlupeg is indicated to increase survival in adults and pediatric patients aged newborn and older acutely exposed to myelosuppressive doses of radiation [see Dosage and Administration (2.2) and Clinical Studies (14.2)] .
DOSAGE AND ADMINISTRATION
- Patients with cancer receiving myelosuppressive chemotherapy
- For adult patients of any weight and pediatric patients weighing 45 kg and greater, the recommended dosage is 6 mg subcutaneously once per chemotherapy cycle. (2.1)
- Do not administer between 14 days before and 24 hours after administration of chemotherapy. (2.1)
- Patients acutely exposed to myelosuppressive doses of radiation
- For adults of any weight and pediatric patients weighing 45 kg and greater, the recommended dosage is two doses, 6 mg each, subcutaneously one week apart. Administer the first dose as soon as possible after suspected or confirmed exposure to myelosuppressive doses of radiation, and a second dose one week after. (2.2)
Patients with Cancer Receiving Myelosuppressive Chemotherapy
The recommended dosage of Armlupeg for adults of any weight and pediatric patients weighing at least 45 kg with cancer receiving myelosuppressive chemotherapy is a single subcutaneous injection of 6 mg administered once per chemotherapy cycle. Do not administer Armlupeg between 14 days before and 24 hours after administration of chemotherapy.
Patients with Hematopoietic Subsyndrome of Acute Radiation Syndrome
The recommended dosage of Armlupeg for adults of any weight and pediatric patients weighing at least 45 kg with hematopoietic subsyndrome of acute radiation syndrome is two doses, 6 mg each, administered subcutaneously one week apart. Administer the first dose as soon as possible after suspected or confirmed exposure to radiation levels greater than 2 gray (Gy). Administer the second dose one week after the first dose.
Obtain a baseline complete blood count (CBC). Do not delay administration of Armlupeg if a CBC is not readily available. Estimate a patient's absorbed radiation dose (i.e., level of radiation exposure) based on information from public health authorities, biodosimetry if available, or clinical findings such as time to onset of vomiting or lymphocyte depletion kinetics.
Preparation and Administration
Armlupeg is supplied in single-dose prefilled syringes for manual use [see Dosage Forms and Strengths (3)] .
Before using Armlupeg:
Remove the carton from the refrigerator and allow the Armlupeg prefilled syringe to reach room temperature, 20℃ to 25℃ (68℉ to 77℉), for a minimum of 30 minutes. Do not warm in any other way. Discard any prefilled syringe left at room temperature for greater than 48 hours.
Armlupeg is a clear, colorless, preservative-free solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer Armlupeg if discoloration or particulates are observed.
Prefilled Syringe for Manual Use
For adult patients of any weight and pediatric patients weighing 45 kg and greater, the single-dose prefilled syringe for manual use is intended for subcutaneous administration of a single 6 mg/0.6 mL dose of Armlupeg. The syringe does not bear graduation marks and therefore does not allow for direct administration of doses less than 6 mg/0.6 mL. There is no presentation for Armlupeg that allows weight-based dosing for pediatric patients below 45 kg.
The needle cap on the prefilled syringe contains dry natural rubber (derived from latex); persons with latex allergies should not administer this product.
DOSAGE FORMS AND STRENGTHS
Armlupeg is a clear, colorless, preservative-free solution available as:
Injection: 6 mg/0.6 mL in a single-dose prefilled syringe for manual use only.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Although available data with pegfilgrastim product use in pregnant women are insufficient to establish whether there is a drug associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes, there are available data from published studies in pregnant women exposed to filgrastim products. These studies have not established an association of filgrastim product use during pregnancy with major birth defects, miscarriage, or adverse maternal or fetal outcomes.
In animal studies, no evidence of reproductive/developmental toxicity occurred in the offspring of pregnant rats that received cumulative doses of pegfilgrastim approximately 10 times the recommended human dose (based on body surface area). In pregnant rabbits, increased embryolethality and spontaneous abortions occurred at 4 times the maximum recommended human dose simultaneously with signs of maternal toxicity (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal data
Pregnant rabbits were dosed with pegfilgrastim subcutaneously every other day during the period of organogenesis. At cumulative doses ranging from the approximate human dose to approximately 4 times the recommended human dose (based on body surface area), the treated rabbits exhibited decreased maternal food consumption, maternal weight loss, as well as reduced fetal body weights and delayed ossification of the fetal skull; however, no structural anomalies were observed in the offspring from either study. Increased incidences of post-implantation losses and spontaneous abortions (more than half the pregnancies) were observed at cumulative doses approximately 4 times the recommended human dose, which were not seen when pregnant rabbits were exposed to the recommended human dose.
Three studies were conducted in pregnant rats dosed with pegfilgrastim at cumulative doses up to approximately 10 times the recommended human dose at the following stages of gestation: during the period of organogenesis, from mating through the first half of pregnancy, and from the first trimester through delivery and lactation. No evidence of fetal loss or structural malformations was observed in any study. Cumulative doses equivalent to approximately 3 and 10 times the recommended human dose resulted in transient evidence of wavy ribs in fetuses of treated mothers (detected at the end of gestation but no longer present in pups evaluated at the end of lactation).
Lactation
Risk Summary
There are no data on the presence of pegfilgrastim products in human milk, the effects on the breastfed child, or the effects on milk production. Other filgrastim products are secreted poorly into breast milk, and filgrastim products are not absorbed orally by neonates. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Armlupeg and any potential adverse effects on the breastfed child from Armlupeg or from the underlying maternal condition.
Pediatric Use
The safety and effectiveness of Armlupeg for chemotherapy-induced neutropenia and hematopoietic subsyndrome of acute radiation syndrome have been established in pediatric patients aged newborn and older.
Use of Armlupeg in pediatric patients for chemotherapy-induced neutropenia is based on adequate and well controlled studies of pegfilgrastim in adults with additional pharmacokinetic and safety data of pegfilgrastim in pediatric patients aged 8 months and older with sarcoma [see Clinical Pharmacology (12.3) and Clinical Studies (14.1)].
The use of Armlupeg to increase survival in pediatric patients acutely exposed to myelosuppressive doses of radiation is based on efficacy studies of pegfilgrastim conducted in animals and clinical data supporting the use of pegfilgrastim in patients with cancer receiving myelosuppressive chemotherapy. Efficacy studies of pegfilgrastim products could not be conducted in humans with acute radiation syndrome for ethical and feasibility reasons. Results from population modeling and simulation indicate that two doses of pegfilgrastim, administered one week apart provide pediatric patients with exposures comparable to that in adults receiving two 6 mg doses one week apart [ see Dosage and Administration (2.1, 2.2), Clinical Pharmacology (12.3) and Clinical Studies (14.2)].
Geriatric Use
Of the 932 patients with cancer who received pegfilgrastim in clinical studies, 139 (15%) were aged 65 and over, and 18 (2%) were aged 75 and over. No overall differences in safety or effectiveness were observed between patients aged 65 and older and younger patients.
CONTRAINDICATIONS
Armlupeg is contraindicated in patients with a history of a serious hypersensitivity reaction to pegfilgrastim products or filgrastim products. Reactions have included anaphylaxis [see Warnings and Precautions (5.3)].
WARNINGS AND PRECAUTIONS
- Fatal splenic rupture: Evaluate patients who report left upper abdominal or shoulder pain for an enlarged spleen or splenic rupture. (5.1)
- Acute respiratory distress syndrome (ARDS): Evaluate patients who develop fever, lung infiltrates, or respiratory distress. Discontinue Armlupeg in patients with ARDS. (5.2)
- Serious hypersensitivity reactions, including anaphylaxis: Permanently discontinue Armlupeg in patients with serious hypersensitivity reactions. (5.3)
- Fatal sickle cell crises: Discontinue Armlupeg if sickle cell crisis occurs (5.4)
- Glomerulonephritis: Evaluate and consider dose-reduction or interruption of Armlupeg if causality is likely. (5.5)
- Thrombocytopenia: Monitor platelet counts. (5.7)
- Myelodysplastic Syndrome (MDS) and Acute Myeloid Leukemia (AML): Monitor patients with breast and lung cancer using Armlupeg in conjunction with chemotherapy and/or radiotherapy for signs and symptoms of MDS/AML. (5.10)
- Aortitis: Discontinue Armlupeg if aortitis is suspected (5.11)
Splenic Rupture
Splenic rupture, including fatal cases, can occur following the administration of pegfilgrastim products. Evaluate for an enlarged spleen or splenic rupture in patients who report left upper abdominal or shoulder pain after receiving Armlupeg.
Acute Respiratory Distress Syndrome
Acute respiratory distress syndrome (ARDS) can occur in patients receiving pegfilgrastim products. Evaluate patients who develop fever and lung infiltrates or respiratory distress after receiving Armlupeg, for ARDS. Discontinue Armlupeg in patients with ARDS.
Serious Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, can occur in patients receiving pegfilgrastim products. The majority of reported events occurred upon initial exposure. Hypersensitivity reactions, including anaphylaxis, can recur within days after the discontinuation of initial therapies to manage the reaction. Permanently discontinue Armlupeg in patients with serious hypersensitivity reactions. Armlupeg is contraindicated in patients with a history of a serious hypersensitivity reaction to pegfilgrastim products or filgrastim products.
Use in Patients with Sickle Cell Disorders
Severe and sometimes fatal sickle cell crises can occur in patients with sickle cell disorders receiving pegfilgrastim products. Discontinue Armlupeg if sickle cell crisis occurs.
Glomerulonephritis
Glomerulonephritis has occurred in patients receiving pegfilgrastim products. The diagnoses were based upon azotemia, hematuria (microscopic and macroscopic), proteinuria, and renal biopsy. Generally, events of glomerulonephritis resolved after dose‑reduction or discontinuation of pegfilgrastim products. If glomerulonephritis is suspected, evaluate for cause. If causality is likely, consider dose-reduction or interruption of Armlupeg.
Leukocytosis
White blood cell (WBC) counts of 100×10 9 /L or greater have been observed in patients receiving pegfilgrastim products. Monitoring of complete blood count (CBC) during Armlupeg therapy is recommended.
Thrombocytopenia
Pegfilgrastim products can cause thrombocytopenia. Monitor platelet counts during Armlupeg therapy.
Capillary Leak Syndrome
Capillary leak syndrome has been reported after G-CSF administration, including pegfilgrastim products, and is characterized by hypotension, hypoalbuminemia, edema and hemoconcentration. Episodes vary in frequency, severity and may be life-threatening if treatment is delayed. Patients who develop symptoms of capillary leak syndrome should be closely monitored and receive standard symptomatic treatment, which may include a need for intensive care.
Potential for Tumor Growth Stimulatory Effects on Malignant Cells
The granulocyte colony-stimulating factor (G-CSF) receptor through which pegfilgrastim products and filgrastim products act has been found on tumor cell lines. The possibility that pegfilgrastim products acts as a growth factor for any tumor type, including myeloid malignancies and myelodysplasia, diseases for which pegfilgrastim products is not approved, cannot be excluded.
Myelodysplastic Syndrome (MDS) and Acute Myeloid Leukemia (AML) in Patients with Breast and Lung Cancer
MDS and AML have been associated with the use of pegfilgrastim products in conjunction with chemotherapy and/or radiotherapy in patients with breast and lung cancer. Monitor patients for signs and symptoms of MDS/AML in these settings.
Aortitis
Aortitis has been reported in patients receiving pegfilgrastim products. It may occur as early as the first week after start of therapy. Manifestations may include generalized signs and symptoms such as fever, abdominal pain, malaise, back pain, and increased inflammatory markers (e.g., c-reactive protein and white blood cell count). Consider aortitis in patients who develop these signs and symptoms without known etiology. Discontinue Armlupeg if aortitis is suspected.
Nuclear Imaging
Increased hematopoietic activity of the bone marrow in response to growth factor therapy, including pegfilgrastim products, has been associated with transient positive bone imaging changes. This should be considered when interpreting bone imaging results.
ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Splenic Rupture [see Warnings and Precautions (5.1)]
- Acute Respiratory Distress Syndrome [see Warnings and Precautions (5.2)]
- Serious Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Use in Patients with Sickle Cell Disorders [see Warnings and Precautions (5.4)]
- Glomerulonephritis [see Warnings and Precautions (5.5)]
- Leukocytosis [see Warnings and Precautions (5.6)]
- Thrombocytopenia [see Warnings and Precautions (5.7)]
- Capillary Leak Syndrome [see Warnings and Precautions (5.8)]
- Potential for Tumor Growth Stimulatory Effects on Malignant Cells [see Warnings and Precautions (5.9)]
- Myelodysplastic syndrome [see Warnings and Precautions ( 5.10 )]
- Acute myeloid leukemia [see Warnings and Precautions ( 5.10 )]
- Aortitis [see Warnings and Precautions (5.11)]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Pegfilgrastim clinical trials safety data are based upon 932 patients receiving pegfilgrastim in seven randomized clinical trials. The population was 21 to 88 years of age and 92% female. The ethnicity was 75% Caucasian, 18% Hispanic, 5% Black, and 1% Asian. Patients with breast (n=823), lung and thoracic tumors (n=53) and lymphoma (n=56) received pegfilgrastim after nonmyeloablative cytotoxic chemotherapy. Most patients received a single 100 mcg/kg (n=259) or a single 6 mg (n= 546) dose per chemotherapy cycle over 4 cycles.
The following adverse reaction data in Table 1 are from a randomized, double-blind, placebo- controlled study in patients with metastatic or non-metastatic breast cancer receiving docetaxel 100 mg/m2 every 21 days (Study 3). A total of 928 patients were randomized to receive either 6 mg pegfilgrastim (n=467) or placebo (n=461). The patients were 21 to 88 years of age and 99% female. The ethnicity was 66% Caucasian, 31% Hispanic, 2% Black, and < 1% Asian, Native American, or other.
The most common adverse reactions occurring in ≥ 5% of patients and with a between-group difference of ≥ 5% higher in the pegfilgrastim arm in placebo-controlled clinical trials are bone pain and pain in extremity.
| Table 1. Adverse Reactions with ≥ 5% Higher Incidence in Pegfilgrastim Patients Compared to Placebo in Study 3 | ||
| Body System Adverse Reaction | Placebo (N = 461) | Pegfilgrastim 6 mg SC on Day 2 (N = 467) |
| Musculoskeletal and connective tissue disorders | ||
| Bone pain | 26% | 31% |
| Pain in extremity | 4% | 9% |
Leukocytosis
In clinical studies, leukocytosis (WBC counts >100x10 9 /L) was observed in less than 1% of 932 patients with non-myeloid malignancies receiving Pegfilgrastim. No complications attributable to leukocytosis were reported in clinical studies.
Postmarketing Experience
The following adverse reactions have been identified during post approval use of Pegfilgrastim products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Splenic rupture and splenomegaly (enlarged spleen) [see Warnings and Precautions (5.1)]
- Acute respiratory distress syndrome (ARDS ) [see Warnings and Precautions (5.2)]
- Allergic reactions/hypersensitivity, including anaphylaxis, skin rash, urticaria, generalized erythema, and flushing [see Warnings and Precautions (5.3)]
- Sickle cell crisis [see Warnings and Precautions (5.4)]
- Glomerulonephritis [see Warnings and Precautions (5.5)]
- Leukocytosis [see Warnings and Precautions (5.6)]
- Thrombocytopenia [see Warnings and Precautions (5.7)]
- Capillary Leak Syndrome [see Warnings and Precautions (5.8)]
- Injection site reactions
- Sweet's syndrome (acute febrile neutrophilic dermatosis), cutaneous vasculitis
- Myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML) in patients with breast and lung cancer receiving chemotherapy and/or radiotherapy [see Warnings and Precautions (5.10)]
- Aortitis [see Warnings and Precautions (5.11)]
- Alveolar hemorrhage
DESCRIPTION
Pegfilgrastim-unne is a covalent conjugate of recombinant methionyl human G-CSF and monomethoxypolyethylene glycol. Recombinant methionyl human G-CSF is a water-soluble 175 amino acid protein with a molecular weight of approximately 19 kilodaltons (kD). Recombinant methionyl human G-CSF is obtained from the bacterial fermentation of a strain of E coli transformed with a genetically engineered plasmid containing the human G-CSF gene. To produce pegfilgrastim-unne, a 20 kD monomethoxypolyethylene glycol molecule is covalently bound to the N-terminal methionyl residue of recombinant methionyl human G-CSF. The average molecular weight of pegfilgrastim-unne is approximately 39 kD.
Armlupeg (pegfilgrastim-unne) injection is a sterile, clear, colorless, preservative-free solution intended for subcutaneous use only and is supplied in a single-dose prefilled syringe with a 27‑gauge ½" needle and rigid needle shield, with UltraSafe Plus Passive TM Needle Guard. The prefilled syringe does not bear graduation marks and is designed to deliver the entire contents of the syringe (6 mg/0.6 mL).
Each prefilled syringe contains 0.6 mL solution of 6 mg pegfilgrastim-unne (based on protein weight), glacial acetic acid (0.77 mg), polysorbate 20 (0.02 mg), sodium acetate (0.066 mg) and sorbitol (30 mg) in Water for Injection, USP. The pH is 4.
CLINICAL PHARMACOLOGY
Mechanism of Action
Pegfilgrastim products are colony-stimulating factor that acts on hematopoietic cells by binding to specific cell surface receptors, thereby stimulating proliferation, differentiation, commitment, and end cell functional activation.
Pharmacodynamics
Animal data and clinical data in humans suggest a correlation between pegfilgrastim products' exposure and the duration of severe neutropenia as a predictor of efficacy. Selection of the dosing regimen of Armlupeg is based on reducing the duration of severe neutropenia.
Pharmacokinetics
The pharmacokinetics of pegfilgrastim was studied in 379 patients with cancer. The pharmacokinetics of pegfilgrastim was nonlinear, and clearance decreased with increases in dose. Neutrophil receptor binding is an important component of the clearance of pegfilgrastim, and serum clearance is directly related to the number of neutrophils. In addition to numbers of neutrophils, body weight appeared to be a factor. Patients with higher body weights experienced higher systemic exposure to pegfilgrastim after receiving a dose normalized for body weight. A large variability in the pharmacokinetics of pegfilgrastim was observed. The half‑life of pegfilgrastim ranged from 15 to 80 hours after subcutaneous injection.
Specific Populations
No gender-related differences were observed in the pharmacokinetics of pegfilgrastim, and no differences were observed in the pharmacokinetics of geriatric patients (≥ 65 years of age) compared with younger patients (< 65 years of age) [see Use in Specific Populations (8.5)].
Renal Impairment
In a study of 30 subjects with varying degrees of renal dysfunction, including end stage renal disease, renal dysfunction had no effect on the pharmacokinetics of pegfilgrastim.
Pediatric Patients with Cancer Receiving Myelosuppressive Chemotherapy
The pharmacokinetics and safety of pegfilgrastim were studied in 37 pediatric patients with sarcoma in Study 4 [see Clinical Studies (14.1)] . The mean (± standard deviation [SD]) systemic exposure (AUC 0-inf ) of pegfilgrastim after subcutaneous administration at 100 mcg/kg was 47.9 (± 22.5) mcg·hr/mL in the youngest age group (0 to 5 years, n = 11), 22.0 (± 13.1) mcg·hr/mL in the 6 to 11 years age group (n=10), and 29.3 (± 23.2) mcg·hr/mL in the 12 to 21 years age group (n=13). The terminal elimination half-lives of the corresponding age groups were 30.1 (±38.2) hours, 20.2 (±11.3) hours, and 21.2 (±16.0) hours, respectively.
Patients Acutely Exposed to Myelosuppressive Doses of Radiation
The pharmacokinetics of pegfilgrastim products is not available in patients acutely exposed to myelosuppressive doses of radiation. Based on limited pharmacokinetic data in irradiated non-human primates, the area under the concentration-time curve (AUC), reflecting the exposure to pegfilgrastim in non-human primates following a 300 mcg/kg dose of pegfilgrastim, appears to be greater than in humans receiving a 6 mg dose. Results from population modeling and simulation indicate that two 6 mg doses of pegfilgrastim administered one week apart in adults result in clinically relevant effects on duration of grade 3 and 4 neutropenia. In addition, weight based dosing in pediatric patients weighing less than 45 kg provides exposures comparable to those in adults receiving two 6 mg doses one week apart.
Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of pegfilgrastim or of other pegfilgrastim products.
Binding antibodies to pegfilgrastim were detected using a BIAcore assay. The approximate limit of detection for this assay is 500 ng/mL. Pre-existing binding antibodies were detected in approximately 6% (51/849) of patients with metastatic breast cancer. Four of 521 pegfilgrastim -treated subjects who were negative at baseline developed binding antibodies to pegfilgrastim following treatment. None of these 4 patients had evidence of neutralizing antibodies detected using a cell-based bioassay.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity or mutagenesis studies have been performed with pegfilgrastim products.
Pegfilgrastim did not affect reproductive performance or fertility in male or female rats at cumulative weekly doses approximately 6 to 9 times higher than the recommended human dose (based on body surface area).
CLINICAL STUDIES
Patients with Cancer Receiving Myelosuppressive Chemotherapy
Pegfilgrastim was evaluated in three randomized, double-blind, controlled studies. Studies 1 and 2 were active‑controlled studies that employed doxorubicin 60 mg/m 2 and docetaxel 75 mg/m 2 administered every 21 days for up to 4 cycles for the treatment of metastatic breast cancer. Study 1 investigated the utility of a fixed dose of pegfilgrastim. Study 2 employed a weight-adjusted dose. In the absence of growth factor support, similar chemotherapy regimens have been reported to result in a 100% incidence of severe neutropenia (ANC < 0.5x10 9 /L) with a mean duration of 5 to 7 days and a 30% to 40% incidence of febrile neutropenia. Based on the correlation between the duration of severe neutropenia and the incidence of febrile neutropenia found in studies with filgrastim, duration of severe neutropenia was chosen as the primary endpoint in both studies, and the efficacy of pegfilgrastim was demonstrated by establishing comparability to filgrastim-treated patients in the mean days of severe neutropenia.
In study 1, 157 patients were randomized to receive a single subcutaneous injection of pegfilgrastim (6 mg) on day 2 of each chemotherapy cycle or daily subcutaneous filgrastim (5 mcg/kg/day) beginning on day 2 of each chemotherapy cycle. In Study 2, 310 patients were randomized to receive a single subcutaneous injection of pegfilgrastim (100 mcg/kg) on day 2 or daily subcutaneous filgrastim (5 mcg/kg/day) beginning on day 2 of each chemotherapy cycle.
Both studies met the major efficacy outcome measure of demonstrating that the mean days of severe neutropenia of pegfilgrastim‑treated patients did not exceed that of filgrastim-treated patients by more than 1 day in cycle 1 of chemotherapy. The mean days of cycle 1 severe neutropenia in Study 1 were 1.8 days in the pegfilgrastim arm compared to 1.6 days in the filgrastim arm [difference in means 0.2 (95% CI - 0.2, 0.6)] and in Study 2 were 1.7 days in the pegfilgrastim arm compared to 1.6 days in the filgrastim arm [difference in means 0.1 (95% CI - 0.2, 0.4)].
A secondary endpoint in both studies was days of severe neutropenia in cycles 2 through 4 with results similar to those for cycle 1.
Study 3 was a randomized, double-blind, placebo-controlled study that employed docetaxel 100 mg/m 2 administered every 21 days for up to 4 cycles for the treatment of metastatic or non-metastatic breast cancer. In this study, 928 patients were randomized to receive a single subcutaneous injection of pegfilgrastim (6 mg) or placebo on day 2 of each chemotherapy cycle. Study 3 met the major trial outcome measure of demonstrating that the incidence of febrile neutropenia (defined as temperature ≥ 38.2°C and ANC ≤ 0.5 x 10 9 /L) was lower for pegfilgrastim‑treated patients as compared to placebo-treated patients (1% versus 17%, respectively, p< 0.001). The incidence of hospitalizations (1% versus 14%) and IV anti-infective use (2% versus 10%) for the treatment of febrile neutropenia was also lower in the pegfilgrastim-treated patients compared to the placebo‑treated patients.
Study 4 was a multicenter, randomized, open-label study to evaluate the efficacy, safety, and pharmacokinetics [see Clinical Pharmacology (12.3)] of pegfilgrastim in pediatric and young adult patients with sarcoma. Patients with sarcoma receiving chemotherapy age 0 to 21 years were eligible. Patients were randomized to receive subcutaneous pegfilgrastim as a single dose of 100 mcg/kg (n = 37) or subcutaneous filgrastim at a dose 5 mcg/kg/day (n = 6) following myelosuppressive chemotherapy. Recovery of neutrophil counts was similar in the pegfilgrastim and filgrastim groups. The most common adverse reaction reported was bone pain.
Patients with Hematopoietic Subsyndrome of Acute Radiation Syndrome
Efficacy studies of pegfilgrastim products could not be conducted in humans with acute radiation syndrome for ethical and feasibility reasons. Approval of this indication was based on efficacy studies conducted in animals and data supporting pegfilgrastim's effect on severe neutropenia in patients with cancer receiving myelosuppressive chemotherapy [see Dosage and Administration (2.1)] .
The recommended dose of Armlupeg is two doses, 6 mg each, administered one week apart for humans exposed to myelosuppressive doses of radiation. For pediatric patients weighing less than 45 kg, dosing of Armlupeg is weight based. This dosing regimen is based on population modeling and simulation analyses. The exposure associated with this dosing regimen is expected to provide sufficient pharmacodynamic activity to treat humans exposed to myelosuppressive doses of radiation [see Clinical Pharmacology (12.3)] . The safety of pegfilgrastim at a dose of 6 mg has been assessed on the basis of clinical experience in patients with cancer receiving myelosuppressive chemotherapy.
The efficacy of pegfilgrastim for the acute radiation syndrome setting was studied in a randomized, placebo-controlled non-human primate model of radiation injury. Rhesus macaques were randomized to either a control (n = 23) or treated (n = 23) cohort. On study day 0, animals (n = 6 to 8 per irradiation day) were exposed to total body irradiation (TBI) of 7.50 ± 0.15 Gy delivered at 0.8 ± 0.03 Gy/min, representing a dose that would be lethal in 50% of animals by 60 days of follow-up (LD50/60). Animals were administered subcutaneous injections of a blinded treatment (control article [5% dextrose in water] or pegfilgrastim [300-319 mcg/kg/day]) on study day 1 and on study day 8. The primary endpoint was survival. Animals received medical management consisting of intravenous fluids, antibiotics, blood transfusions, and other support as required.
Pegfilgrastim significantly (at 0.0014 level of significance) increased 60-day survival in irradiated non-human primates: 91% survival (21/23) in the pegfilgrastim group compared to 48% survival (11/23) in the control group.
HOW SUPPLIED/STORAGE AND HANDLING
Armlupeg (pegfilgrastim-unne) injection is a clear, colorless solution supplied in a prefilled single-dose syringe for manual use containing 6 mg pefilgrastim-unne, supplied with a 27-gauge, 1/2-inch needle with an UltraSafe® Plus Passive TM Needle Guard.
The needle cap of the prefilled syringe contains dry natural rubber (a derivative of latex).
Armlupeg is provided in a carton containing one sterile 6 mg/0.6 mL prefilled syringe (NDC 70748-273-01).
Armlupeg prefilled syringe does not bear graduation marks and is intended only to deliver the entire contents of the syringe (6 mg/0.6 mL) for direct administration.
Store refrigerated between 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not shake. Discard syringes stored at room temperature for more than 48 hours. Avoid freezing; if frozen, thaw in the refrigerator before administration. Discard syringe if frozen more than once.
INSTRUCTIONS FOR USE SECTION
Instructions for Use
ARMLUPEG ® (arm loo peg)
(pegfilgrastim-unne)
injection, for subcutaneous use
Single-Dose Prefilled Syringe
Guide to parts
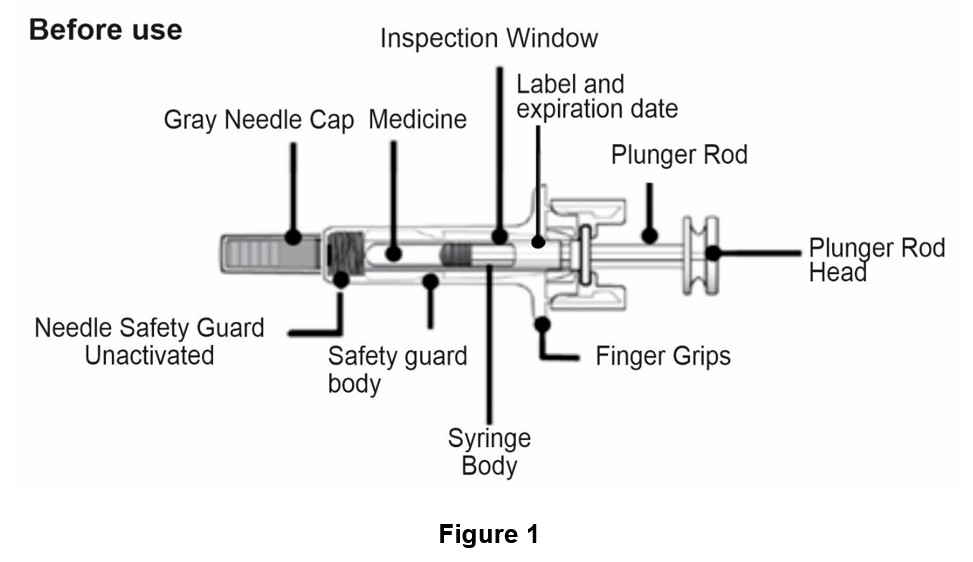
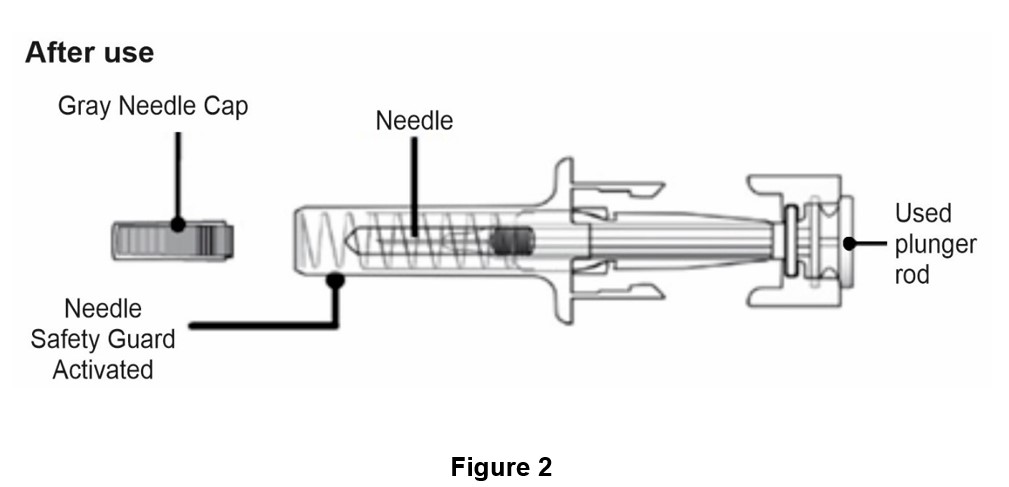
Important: The needle is covered by the gray needle cap before use.
Important
Read the Patient Information for important information you need to know about Armlupeg before using these Instructions for Use.
Before you use an Armlupeg prefilled syringe, read this important information.
Storing the prefilled syringe
Store Armlupeg in the refrigerator between 36°F to 46° F (2°C to 8°C).
Do not freeze. If Armlupeg is accidentally frozen, allow the prefilled syringe to thaw in the refrigerator before injecting.
Do not use an Armlupeg prefilled syringe that has been frozen more than 1 time. Throw it away and use a new Armlupeg prefilled syringe.
Keep the prefilled syringe in the original carton to protect from light or physical damage.
Take the prefilled syringe out of the refrigerator 30 minutes before use and allow it to reach room temperature, 68°F to 77°F (20°C to 25°C), before preparing an injection.
Throw away (dispose of) any Armlupeg that has been left at room temperature, 68°F to 77°F (20°C to 25°C), for more than 48 hours.
Keep Armlupeg and all medicines out of the reach of children .
Using the prefilled syringe
It is important that you do not try to give the injection unless you or your caregiver has received training from your healthcare provider.
Make sure the name Armlupeg appears on the carton and prefilled syringe label.
Check the carton and prefilled syringe label to make sure the dose strength is 6 mg.
You should not inject a dose of Armlupeg to children weighing less than 99 pounds (45 kg) from an Armlupeg prefilled syringe.
Do not use a prefilled syringe after the expiration date on the label.
Do not shake the prefilled syringe.
Do not remove the gray needle cap from the prefilled syringe until you are ready to inject.
Do not use the prefilled syringe if the carton is open or damaged.
Do not use a prefilled syringe if it has been dropped on a hard surface. The prefilled syringe may be broken even if you cannot see the break. Use a new prefilled syringe.
Do not attempt to activate the needle safety guard prior to injection. Do not use the prefilled syringe if the needle safety guard has been activated. Use another prefilled syringe that has not been activated and is ready to use.
The gray needle cap on the prefilled syringe contains dry natural rubber (made from latex). Tell your healthcare provider if you are allergic to latex. You should not give Armlupeg using the prefilled syringe if you have latex allergies.
Call your healthcare provider if you have any questions.
STEP 1: Prepare
A. Remove the prefilled syringe carton from refrigerator.
Check the expiration date printed on the carton.
Important: Do not use if the expiration date has passed.
Remove the syringe tray from the carton. On a clean, well-lit surface, place the syringe tray at room temperature for 30 minutes before you give an injection.
Do not remove the prefilled syringe from the original carton until you are ready to inject.
Do not use the prefilled syringe if the carton is damaged.
Do not try to warm the prefilled syringe by using a heat source such as hot water or microwave.
Do not leave the prefilled syringe in direct sunlight.
Do not shake the prefilled syringe.
Open the tray by peeling away the cover. Grab the safety guard to remove the prefilled syringe from the tray. (See Figure 3)
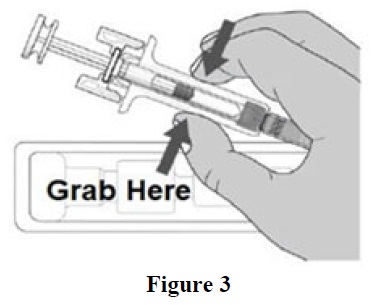
For Safety reasons:
B. Inspect the medicine and prefilled syringe (See Figure 4).
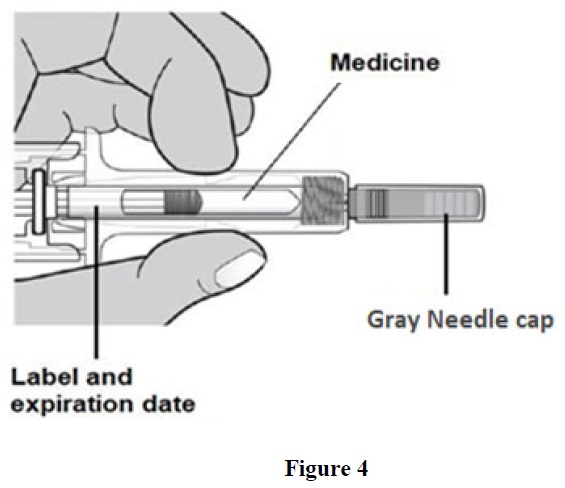
Make sure the medicine in prefilled syringe is clear and colorless (See Figure 4). It is normal to see one or more air bubbles in the syringe.
Do not use the prefilled syringe if:
The medicine is cloudy or discolored or contains flakes or particles.
Any part appears cracked or broken.
The prefilled syringe has been dropped.
The gray needle cap is missing or not securely attached.
The expiration date printed on the label has passed.
In all cases, use a new prefilled syringe and call your healthcare provider.
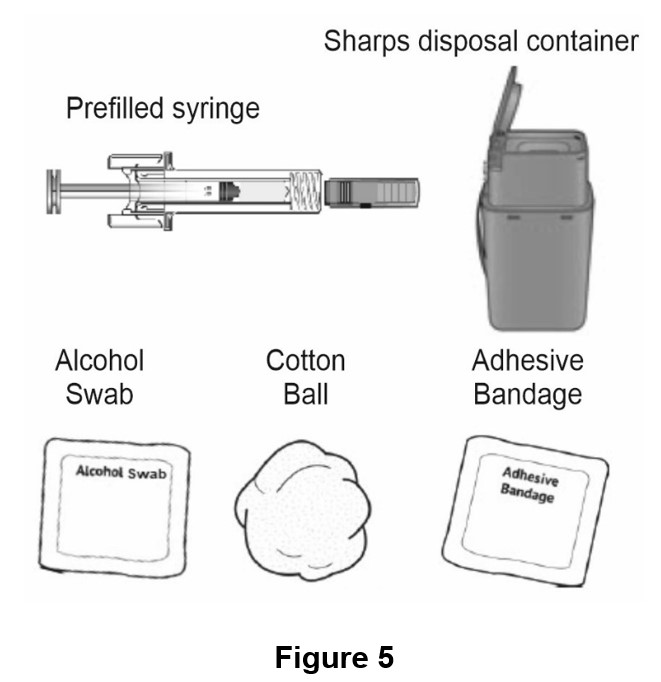
C. Gather all materials needed for injection (See Figure 5)
Wash your hands thoroughly with soap and water
On a clean, well-lit work surface, place the:
Prefilled syringe
Alcohol swab
Cotton ball or gauze pad
Adhesive bandage
Sharps disposal container
STEP 2: Get ready
D. Select and clean the injection site (See Figures 6 and 7).
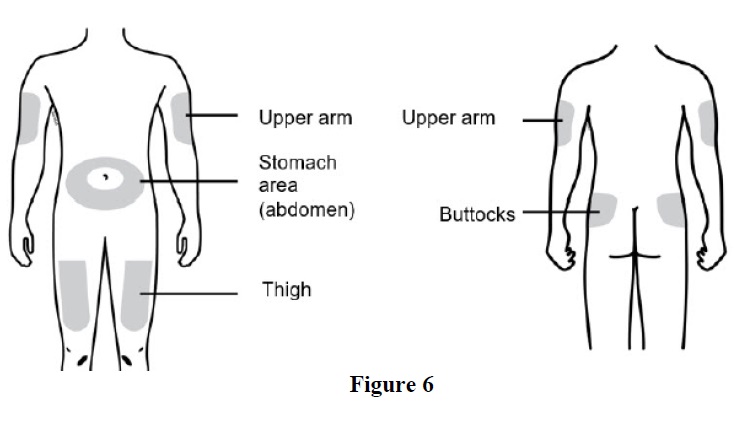
You can use:
Clean the injection site with an alcohol swab (See Figure 7). Let the skin dry.
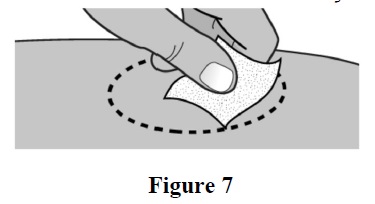
Do not touch this area again before injecting.
If you want to use the same injection site, make sure it is not the same spot on the injection site you used for a previous injection.
Do not inject into areas where the skin is tender, bruised, red, or hard. Avoid injecting into areas with scars or stretch marks.
E. Hold the prefilled syringe by the syringe body. Carefully pull the gray needle cap straight off and away from the body (See Figure 8).
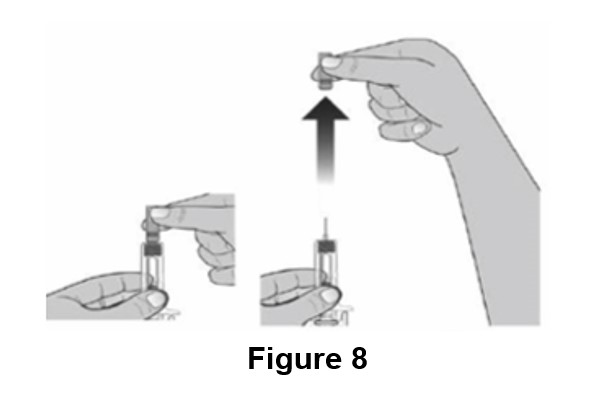
Do not remove the gray needle cap from the prefilled syringe until you are ready to inject.
Do not twist or bend the needle cap.
Do not hold the prefilled syringe by the plunger rod
Do not put the gray needle cap back onto the syringe or never recap. If the needle cap is removed accidently and you are not ready for the injection, discard (throw away) the opened syringe and use a new syringe when ready.
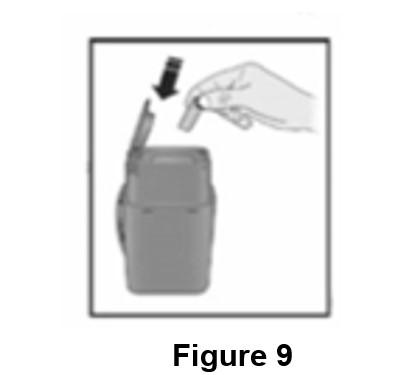
Important: Throw the gray needle cap into the sharps disposal container (See Figure 9).
Step 3: Subcutaneous (under the skin) injection
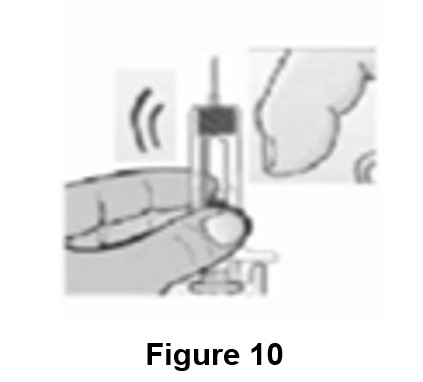
F. Point the needle up and gently tap the syringe body with your fingers until the air bubble rises to the top of the syringe (See Figure 10).
G. Pinch the injection site to create a firm surface (See Figure 11).
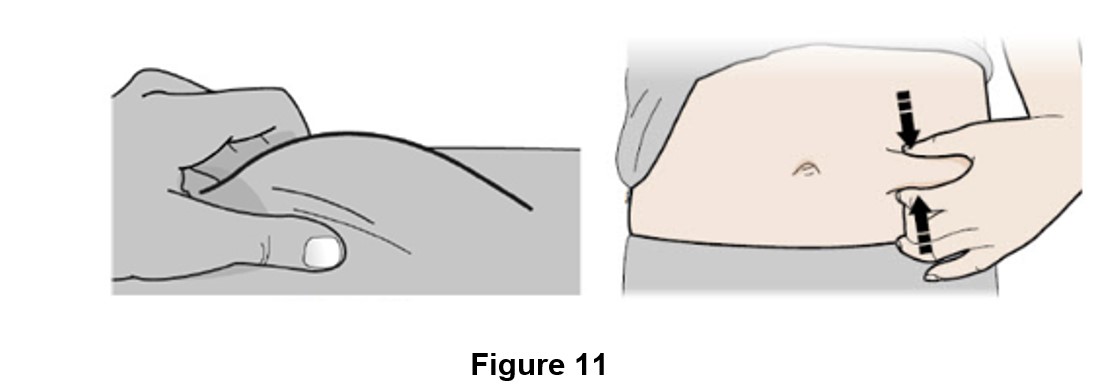
Important: Keep skin pinched while injecting
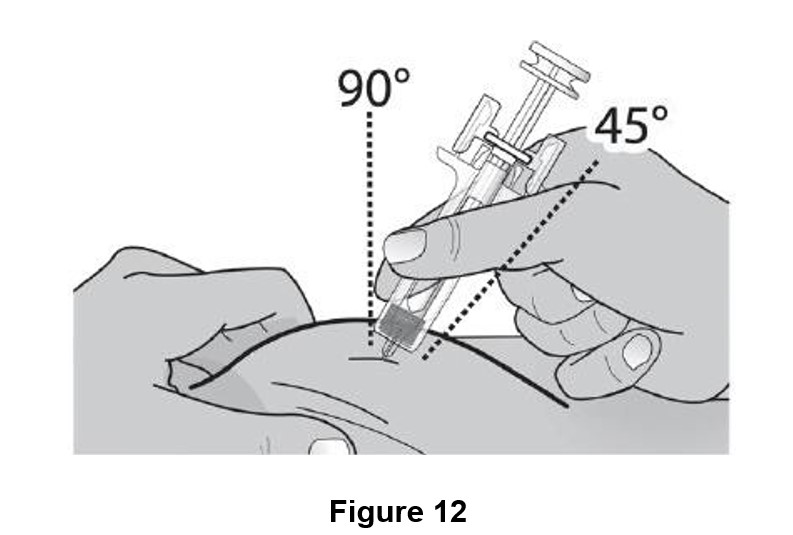
H. Hold the pinch. Insert the needle into the skin at 45 to 90 degrees. (See Figure 12)
Do not touch the plunger rod or grasp the syringe above the finger grips.
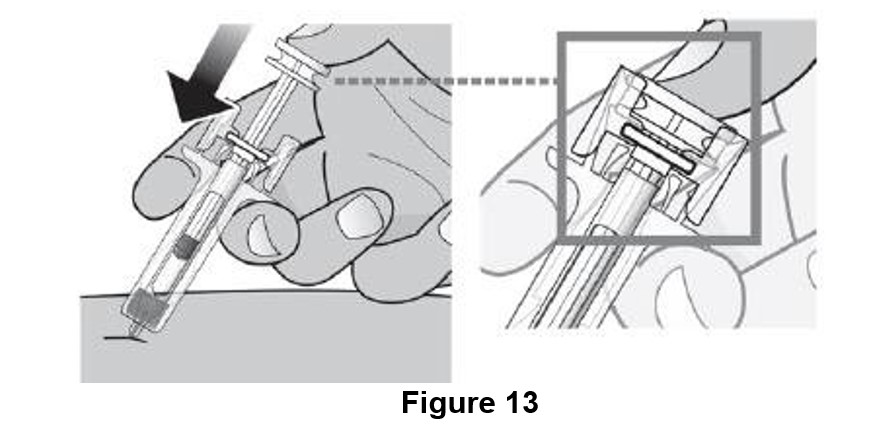
I. Using slow and constant pressure, push the plunger head until it reaches the bottom. (See Figure 13)
While the needle is still inserted in the skin, slowly move your thumb back, allowing the plunger rod to rise. This will release the needle safety guard to safely cover the needle. Then remove the syringe from the injection site. (See Figure 14)
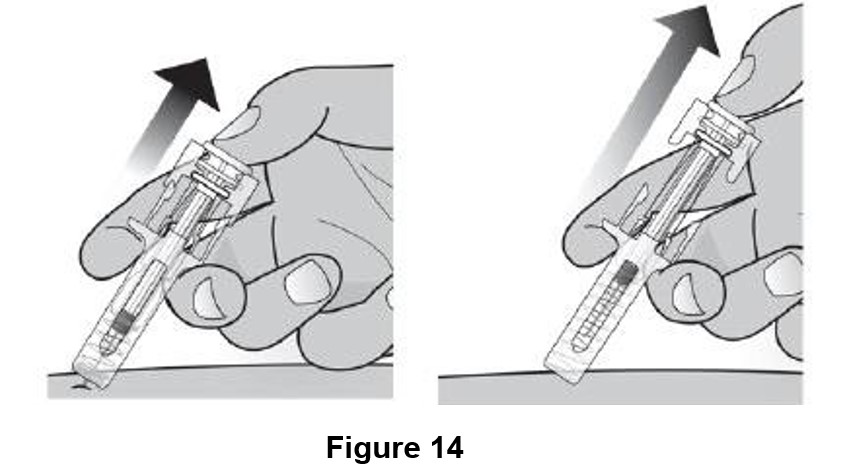
Important: When you remove the syringe, if it looks like the needle safety guard is not activated or some medicine is still left in the syringe, this means you have not received a full dose. Call your healthcare provider right away.
Step 4: Finish

J. For your safety, make sure that the needle safety guard is activated and fully covers the needle.
Once the needle safety guard is extended, it will lock into position and will not slide back over the needle.
Important : Keep your hands away from the needle at all times.
K. Discard (throw away) the used prefilled syringe.

Put the used prefilled syringe into an FDA-cleared sharps disposal container right away after use (See Figure 15). Do not throw away (dispose of) the syringe in the household trash.
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
o made of a heavy-duty plastic,
o can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
o upright and stable during use,
o leak-resistant, and
o properly labeled to warn of hazardous waste inside the container.
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
Do not reuse the prefilled syringe.
Do not recycle prefilled syringes or sharps disposal container or throw them into household trash.
Important: Always keep the sharps disposal container out of the reach of children.
L. Examine the injection site.
If there is blood, press a cotton ball or gauze pad on the injection site. Do not rub the injection site. Apply an adhesive bandage if needed.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Lupin Pharmaceuticals, Inc.
Naples, FL 34108 United States.
Manufactured by:
Lupin Limited (Biotech Division)
Gat No. 1156, 1157, 1158, 1159 and 1160,
Village-Ghotawade, Taluka-Mulshi,
Pune – 412115, Maharashtra, India
Distributed by:
Valorum Biologics, LLC
555 Madison Ave. Suite 11D
New York, NY USA 10022
U.S. License No. 2052
Approved: November 2025
Mechanism of Action
Pegfilgrastim products are colony-stimulating factor that acts on hematopoietic cells by binding to specific cell surface receptors, thereby stimulating proliferation, differentiation, commitment, and end cell functional activation.