Get your patient on Azelastine Hydrochloride - Azelastine Hydrochloride solution/ Drops (Azelastine Hydrochloride)
Azelastine Hydrochloride - Azelastine Hydrochloride solution/ Drops prescribing information
INDICATIONS AND USAGE
Azelastine hydrochloride is indicated for the treatment of itching of the eye associated with allergic conjunctivitis.
DOSAGE AND ADMINISTRATION
The recommended dose is one drop instilled into each affected eye twice a day.
CONTRAINDICATIONS
Azelastine hydrochloride is contraindicated in persons with known or suspected hypersensitivity to any of its components.
ADVERSE REACTIONS
In controlled multiple-dose studies where patients were treated for up to 56 days, the most frequently reported adverse reactions were transient eye burning/stinging (approximately 30%), headaches (approximately 15%) and bitter taste (approximately 10%). The occurrence of these events was generally mild.
The following events were reported in 1–10% of patients: asthma, conjunctivitis, dyspnea, eye pain, fatigue, influenza-like symptoms, pharyngitis, pruritus, rhinitis and temporary blurring. Some of these events were similar to the underlying disease being studied.
DESCRIPTION
Azelastine hydrochloride ophthalmic solution, 0.05% is a sterile ophthalmic solution containing azelastine hydrochloride, a relatively selective H 1 -receptor antagonist for topical administration to the eyes. Azelastine hydrochloride is a white crystalline powder with a molecular weight of 418.37. Azelastine hydrochloride is sparingly soluble in water, methanol and propylene glycol, and slightly soluble in ethanol, octanol, and glycerine. Azelastine hydrochloride is a racemic mixture with a melting point of 225°C. The chemical name for azelastine hydrochloride is (±)-1-(2H)-phthalazinone,4-[(4-chlorophenyl) methyl]-2-(hexahydro-1-methyl-1H-azepin-4-yl)-, mono hydrochloride and is represented by the following chemical structure:
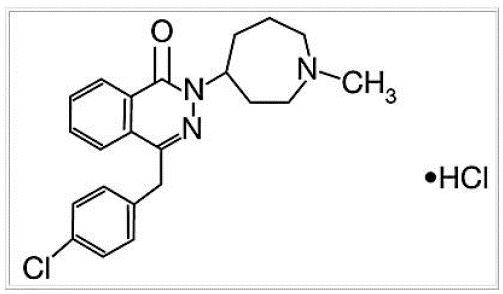
Empirical chemical structure: C 22 H 24 ClN 3 O•HCl
Each mL of Azelastine hydrochloride ophthalmic solution contains: Active: 0.5 mg azelastine hydrochloride, equivalent to 0.457 mg of azelastine base; Preservative: 0.125 mg benzalkonium chloride; Inactives: disodium edetate dihydrate, hypromellose, sorbitol solution, sodium hydroxide and water for injection. It has a pH of approximately 5.0 to 6.5 and an osmolarity of approximately 271 to 312 mOsmol/L.
CLINICAL PHARMACOLOGY
Azelastine hydrochloride is a relatively selective histamine H 1 antagonist and an inhibitor of the release of histamine and other mediators from cells (e.g. mast cells) involved in the allergic response. Based on in-vitro studies using human cell lines, inhibition of other mediators involved in allergic reactions (e.g. leukotrienes and PAF) has been demonstrated with azelastine hydrochloride. Decreased chemotaxis and activation of eosinophils has also been demonstrated.
Pharmacokinetics and Metabolism
Absorption of azelastine following ocular administration was relatively low. A study in symptomatic patients receiving one drop of azelastine hydrochloride in each eye two to four times a day (0.06 to 0.12 mg azelastine hydrochloride) demonstrated plasma concentrations of azelastine hydrochloride to generally be between 0.02 and 0.25 ng/mL after 56 days of treatment. Three of nineteen patients had quantifiable amounts of N-desmethyl azelastine that ranged from 0.25–0.87 ng/mL at Day 56.
Based on intravenous and oral administration, the elimination half-life, steady-state volume of distribution and plasma clearance were 22 hours, 14.5 L/kg and 0.5 L/h/kg, respectively. Approximately 75% of an oral dose of radiolabeled azelastine hydrochloride was excreted in the feces with less than 10% as unchanged azelastine. Azelastine hydrochloride is oxidatively metabolized to the principal metabolite, N-desmethyl azelastine, by the cytochrome P450 enzyme system. In-vitro studies in human plasma indicate that the plasma protein binding of azelastine and N-desmethyl azelastine are approximately 88% and 97%, respectively.
Clinical Trials
In a conjunctival antigen challenge study, azelastine hydrochloride was more effective than its vehicle in preventing itching associated with allergic conjunctivitis. Azelastine hydrochloride had a rapid (within 3 minutes) onset of effect and a duration of effect of approximately 8 hours for the prevention of itching.
In environmental studies, adult and pediatric patients with seasonal allergic conjunctivitis were treated with azelastine hydrochloride for two to eight weeks. In these studies, azelastine hydrochloride was more effective than its vehicle in relieving itching associated with allergic conjunctivitis.
HOW SUPPLIED
Azelastine hydrochloride ophthalmic solution, 0.05% is supplied as follows:
6 mL (NDC # 70069-091-01) solution in 10 mL natural HDPE bottle plugged with natural LDPE nozzle and white HDPE screw cap.
Storage
Store UPRIGHT between 2° and 25°C (36° and 77°F).
Rx only
For Product Inquiry call +1-800-417-9175
Manufactured for:
Somerset Therapeutics, LLC
Hollywood, FL 33024
Made in India
Code No.:KR/DRUGS/KTK/28/289/97
Issued: December, 2019
ST-AZL11/P/02
PSSO0480