Benazepril Hydrochloride And Hydrochlorothiazide - Benazepril Hydrochloride And Hydrochlorothiazide tablet, Film Coated prescribing information
WARNING: FETAL TOXICITY
When pregnancy is detected, discontinue benazepril hydrochloride and hydrochlorothiazide as soon as possible.
Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus (see WARNINGS, Fetal Toxicity).
INDICATIONS AND USAGE
Benazepril hydrochloride and hydrochlorothiazide tablets are indicated for the treatment of hypertension.
This fixed combination drug is not indicated for the initial therapy of hypertension (see DOSAGE AND ADMINISTRATION ).
DOSAGE AND ADMINISTRATION
Dose once daily. The dosage may then be increased after 2 to 3 weeks as needed to help achieve blood pressure goals. The maximum recommended dose is 20/25 mg.
Switch Therapy
A patient whose blood pressure is not adequately controlled with benazepril alone or with hydrochlorothiazide alone may be switched to combination therapy with benazepril hydrochloride and hydrochlorothiazide tablets. The usual recommended starting dose is 10/12.5 mg once daily to control blood pressure.
Replacement Therapy
The combination may be substituted for the titrated individual components.
CONTRAINDICATIONS
Benazepril hydrochloride and hydrochlorothiazide is contraindicated in patients who are anuric.
Benazepril hydrochloride and hydrochlorothiazide is also contraindicated in patients who are hypersensitive to benazepril, to any other ACE inhibitor, to hydrochlorothiazide, or to other sulfonamide-derived drugs. Hypersensitivity reactions are more likely to occur in patients with a history of allergy or bronchial asthma.
Benazepril hydrochloride and hydrochlorothiazide is also contraindicated in patients with a history of angioedema with or without previous ACE inhibitor treatment.
Benazepril hydrochloride and hydrochlorothiazide is contraindicated in combination with a neprilysin inhibitor (e.g., sacubitril). Do not administer benazepril hydrochloride and hydrochlorothiazide within 36 hours of switching to or from sacubitril/valsartan, a neprilysin inhibitor (see WARNINGS and PRECAUTIONS ).
Do not coadminister aliskiren with angiotensin receptor blockers, ACE inhibitors, including benazepril hydrochloride and hydrochlorothiazide in patients with diabetes.
ADVERSE REACTIONS
Benazepril hydrochloride and hydrochlorothiazide has been evaluated for safety in over 2500 patients with hypertension; over 500 of these patients were treated for at least 6 months, and over 200 were treated for more than 1 year.
The reported side effects were generally mild and transient, and there was no relationship between side effects and age, sex, race, or duration of therapy. Discontinuation of therapy due to side effects was required in approximately 7% of U.S. patients treated with benazepril hydrochloride and hydrochlorothiazide and in 4% of patients treated with placebo.
The most common reasons for discontinuation of therapy with benazepril hydrochloride and hydrochlorothiazide in U.S. studies were cough (1.0%; see PRECAUTIONS ), “dizziness” (1.0%), headache (0.6%), and fatigue (0.6%).
The side effects considered possibly or probably related to study drug that occurred in U.S. placebo-controlled trials in more than 1% of patients treated with benazepril hydrochloride and hydrochlorothiazide are shown in the table below.
Benazepril Hydrochloride and Hydrochlorothiazide N=665 | Placebo N=235 | |||
N | % | N | % | |
“Dizziness” | 41 | 6.3 | 8 | 3.4 |
Fatigue | 34 | 5.2 | 6 | 2.6 |
Postural Dizziness | 23 | 3.5 | 1 | 0.4 |
Headache | 20 | 3.1 | 10 | 4.3 |
Cough | 14 | 2.1 | 3 | 1.3 |
Hypertonia | 10 | 1.5 | 3 | 1.3 |
Vertigo | 10 | 1.5 | 2 | 0.9 |
Nausea | 9 | 1.4 | 2 | 0.9 |
Impotence | 8 | 1.2 | 0 | 0.0 |
Somnolence | 8 | 1.2 | 1 | 0.4 |
Other side effects considered possibly or probably related to study drug that occurred in U.S. placebo-controlled trials in 0.3% to 1.0% of patients treated with benazepril hydrochloride and hydrochlorothiazide were the following:
Cardiovascular
Palpitations, flushing.
Gastrointestinal
Vomiting, diarrhea, dyspepsia, anorexia, and constipation.
Neurologic and Psychiatric
Insomnia, nervousness, paresthesia, libido decrease, dry mouth, taste perversion, and tinnitus.
Dermatologic
Rash and sweating.
Other
Urinary frequency, arthralgia, myalgia, asthenia, and pain (including chest pain and abdominal pain).
Other adverse experiences reported in 0.3% or more of benazepril hydrochloride and hydrochlorothiazide patients in U.S. controlled clinical trials, and rarer events seen in post-marketing experience, were the following; asterisked entries occurred in more than 1% of patients (in some, a causal relationship to benazepril hydrochloride and hydrochlorothiazide is uncertain):
Cardiovascular
Syncope, peripheral vascular disorder, and tachycardia.
Body as a Whole
Infection, back pain•, flu syndrome•, fever, chills, and neck pain.
Dermatologic
Photosensitivity and pruritus.
Gastrointestinal
Gastroenteritis, flatulence, and tooth disorder.
Neurologic and Psychiatric
Hypesthesia, abnormal vision, abnormal dreams, and retinal disorder.
Respiratory
Upper respiratory infection•, epistaxis, bronchitis, rhinitis•, sinusitis•, and voice alteration.
Other
Conjunctivitis, arthritis, urinary tract infection, alopecia, and urinary frequency•.
Post-Marketing Experience
The following adverse reactions have been identified during post-approval use of either benazepril or hydrochlorothiazide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure:
Non-melanoma Skin Cancer
Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥50,000mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
Benazepril
Stevens-Johnson syndrome, pancreatitis, hemolytic anemia, pemphigus, and thrombocytopenia, eosinophilic pneumonitis.
Hydrochlorothiazide
Digestive
Pancreatitis, small bowel angioedema, jaundice (intrahepatic cholestatic), sialadenitis, vomiting, diarrhea, cramping, nausea, gastric irritation, constipation, and anorexia.
Neurologic
Vertigo, lightheadedness, transient blurred vision, headache, paresthesia, xanthopsia, weakness, and restlessness.
Musculoskeletal
Muscle spasm.
Hematologic
Aplastic anemia, agranulocytosis, leukopenia, neutropenia and thrombocytopenia.
Metabolic
Hyperglycemia, glycosuria, and hyperuricemia, pyrexia, asthenia, parathyroid gland changes with hypercalcemia and hypophosphatemia.
Hypersensitivity
Anaphylactoid reactions, necrotizing angiitis, respiratory distress (including pneumonitis and pulmonary edema), purpura, urticaria, rash, and photosensitivity.
Skin
Erythema multiforme including Stevens-Johnson syndrome, and exfoliative dermatitis including toxic epidermal necrolysis.
Clinical Laboratory Test Findings
Serum Electrolytes
See PRECAUTIONS .
Creatinine and BUN
Minor reversible increases in serum creatinine and BUN were observed in patients with essential hypertension treated with benazepril hydrochloride and hydrochlorothiazide. Such increases occurred most frequently in patients with renal artery stenosis (see PRECAUTIONS ).
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Benazepril hydrochloride, USP is a white to off-white crystalline powder, soluble (>100 mg/mL) in water, in ethanol, and in methanol. Benazepril hydrochloride's chemical name is 3-[[1-(ethoxycarbonyl)-3-phenyl-(1S)-propyl]amino]-2,3,4,5- tetrahydro-2-oxo-1 H -1-(3S)-benzazepine-1-acetic acid monohydrochloride; its structural formula is:
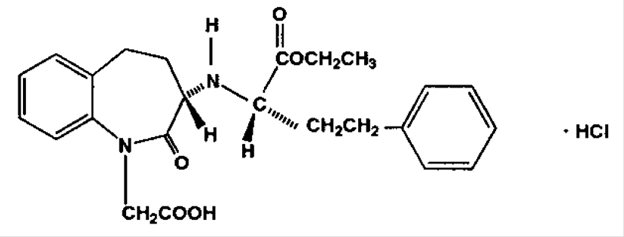
Its molecular formula is C 24 H 28 N 2 O 5 •HCl, and its molecular weight is 460.96.
Benazeprilat, the active metabolite of benazepril, is a nonsulfhydryl angiotensin-converting enzyme inhibitor. Benazepril is converted to benazeprilat by hepatic cleavage of the ester group.
Hydrochlorothiazide, USP is a white, or practically white, practically odorless, crystalline powder. It is slightly soluble in water; freely soluble in sodium hydroxide solution, in n -butylamine, and in dimethylformamide; sparingly soluble in methanol; and insoluble in ether, in chloroform, and in dilute mineral acids. Hydrochlorothiazide's chemical name is 6-chloro-3,4-dihydro-2 H -1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide; its structural formula is:
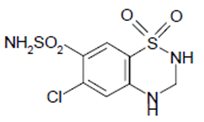
Its molecular formula is C 7 H 8 ClN 3 O 4 S 2, and its molecular weight is 297.73. Hydrochlorothiazide is a thiazide diuretic.
The tablets are a combination of benazepril hydrochloride and hydrochlorothiazide, USP. They are formulated for oral administration with a combination of 5 mg, 10 mg, or 20 mg of benazepril hydrochloride and 6.25 mg, 12.5 mg, or 25 mg of hydrochlorothiazide, USP. The inactive ingredients of the tablets are colloidal silicon dioxide, crospovidone, hydrogenated castor oil, lactose monohydrate, poloxamer, polyethylene glycol, pregelatinized starch (corn), titanium dioxide and zinc stearate. The 5 mg/6.25 mg tablets also contain polyvinyl alcohol - part hydrolyzed and talc. The 10 mg/12.5 mg tablets also contain FD&C red No. 40, FD&C yellow No. 6, polyvinyl alcohol and talc. The 20 mg/12.5 mg tablets also contain FD&C blue No. 2, FD&C red No. 40, hypromellose and polysorbate. The 20 mg/25 mg tablets also contain FD&C red No. 40, hypromellose and polysorbate.
This product meets USP Dissolution Test 2.
CLINICAL PHARMACOLOGY
Mechanism of Action
Benazepril and benazeprilat inhibit angiotensin-converting enzyme (ACE) in human subjects and in animals. ACE is a peptidyl dipeptidase that catalyzes the conversion of angiotensin I to the vasoconstrictor substance, angiotensin II. Angiotensin II also stimulates aldosterone secretion by the adrenal cortex.
Inhibition of ACE results in decreased plasma angiotensin II, which leads to decreased vasopressor activity and to decreased aldosterone secretion. The latter decrease may result in a small increase of serum potassium. Hypertensive patients treated with benazepril alone for up to 52 weeks had elevations of serum potassium of up to 0.2 mEq/L. Similar patients treated with benazepril and hydrochlorothiazide for up to 24 weeks had no consistent changes in their serum potassium (see PRECAUTIONS ).
Removal of angiotensin II negative feedback on renin secretion leads to increased plasma renin activity. In animal studies, benazepril had no inhibitory effect on the vasopressor response to angiotensin II and did not interfere with the hemodynamic effects of the autonomic neurotransmitters acetylcholine, epinephrine, and norepinephrine.
ACE is identical to kininase, an enzyme that degrades bradykinin. Whether increased levels of bradykinin, a potent vasodepressor peptide, play a role in the therapeutic effects of benazepril hydrochloride and hydrochlorothiazide remains to be elucidated.
While the mechanism through which benazepril lowers blood pressure is believed to be primarily suppression of the renin-angiotensin-aldosterone system, benazepril has an antihypertensive effect even in patients with low-renin hypertension.
Hydrochlorothiazide is a thiazide diuretic. Thiazides affect the renal tubular mechanisms of electrolyte reabsorption, directly increasing excretion of sodium and chloride in approximately equivalent amounts. Indirectly, the diuretic action of hydrochlorothiazide reduces plasma volume, with consequent increases in plasma renin activity, increases in aldosterone secretion, increases in urinary potassium loss, and decreases in serum potassium. The renin-aldosterone link is mediated by angiotensin, so coadministration of an ACE inhibitor tends to reverse the potassium loss associated with these diuretics.
The mechanism of the antihypertensive effect of thiazides is unknown.
Pharmacokinetics and Metabolism
Following oral administration of benazepril hydrochloride and hydrochlorothiazide, peak plasma concentrations of benazepril are reached within 0.5 to 1.0 hour. As determined by urinary recovery, the extent of absorption is at least 37%. In fasting subjects, the rate and extent of absorption of benazepril and hydrochlorothiazide from benazepril hydrochloride and hydrochlorothiazide are not different, respectively, from the rate and extent of absorption of benazepril and hydrochlorothiazide from immediate-release monotherapy formulations.
The estimated absolute bioavailability of hydrochlorothiazide after oral administration is about 70%. Peak plasma hydrochlorothiazide concentrations (C max ) are reached within 2 to 5 hours after oral administration. Hydrochlorothiazide binds to albumin (40% to 70%) and distributes into erythrocytes.
The absorption of benazepril from benazepril hydrochloride tablets is not influenced by the presence of food in the gastrointestinal tract. There is no clinically significant effect of food on the bioavailability of hydrochlorothiazide.
Cleavage of the ester group (primarily in the liver) converts benazepril to its active metabolite, benazeprilat. Peak plasma concentrations of benazeprilat are reached 1 to 2 hours after drug intake in the fasting state and 2 to 4 hours after drug intake in the nonfasting state. The serum protein binding of benazepril is about 96.7% and that of benazeprilat about 95.3%, as measured by equilibrium dialysis; on the basis of in vitro studies, the degree of protein binding should be unaffected by age, hepatic dysfunction, or - over the concentration range of 0.24 to 23.6 mcmol/L - concentration.
In studies of rats given 14 C-benazepril, benazepril and its metabolites crossed the blood-brain barrier only to an extremely low extent. Multiple doses of benazepril did not result in accumulation in any tissue except the lung, where, as with other ACE inhibitors in similar studies, there was a slight increase in concentration due to slow elimination in that organ.
Benazepril is almost completely metabolized to benazeprilat, which has much greater ACE inhibitory activity than benazepril, and to the glucuronide conjugates of benazepril and benazeprilat. Only trace amounts of an administered dose of benazepril can be recovered unchanged in the urine; about 20% of the dose is excreted as benazeprilat, 4% as benazepril glucuronide, and 8% as benazeprilat glucuronide.
In patients with hepatic dysfunction due to cirrhosis, levels of benazeprilat are essentially unaltered. Similarly, the pharmacokinetics of benazepril and benazeprilat do not appear to be influenced by age.
The kinetics of benazepril are dose-proportional within the dosage range of 5 mg to 20 mg. Small deviations from dose proportionality were observed when the broader range of 2 mg to 80 mg was studied, possibly due to the saturable binding of the compound to ACE.
The effective half-life of accumulation of benazeprilat following multiple dosing of benazepril is 10 to 11 hours. Thus, steady-state concentrations of benazeprilat should be reached after 2 or 3 doses of benazepril given once daily.
During chronic administration (28 days) of once-daily doses of benazepril between 5 mg and 20 mg, the kinetics did not change, and there was no significant accumulation. Accumulation ratios based on AUC and urinary recovery of benazeprilat were 1.19 and 1.27, respectively.
When dialysis was started 2 hours after ingestion of 10 mg of benazepril, approximately 6% of benazeprilat was removed in 4 hours of dialysis. The parent compound, benazepril, was not detected in the dialysate.
Benazepril and benazeprilat are cleared predominantly by renal excretion in healthy subjects with normal renal function. Nonrenal (i.e., biliary) excretion accounts for approximately 11% to 12% of benazeprilat excretion in healthy subjects. In patients with renal failure, biliary clearance may compensate to an extent for deficient renal clearance.
The disposition of benazepril and benazeprilat in patients with mild-to-moderate renal insufficiency (creatinine clearance >30 mL/min) is similar to that in patients with normal renal function. In patients with creatinine clearance ≤30 mL/min, peak benazeprilat levels and the initial (alpha phase) half-life increase, and time to steady-state may be delayed (see DOSAGE AND ADMINISTRATION ).
Following oral administration, plasma hydrochlorothiazide concentrations decline bi-exponentially, with a mean distribution half-life of about 2 hours and an elimination half-life of about 10 hours. About 70% of an orally administered dose of hydrochlorothiazide is eliminated in the urine as unchanged drug. In a study in individuals with impaired renal function, the mean elimination half-life of hydrochlorothiazide was increased to 2 fold in individuals with mild/moderate renal impairment (30 ˂CLcr ˂90 mL/min) and 3 fold and severe renal impairment (≤30 mL/min), when compared to individuals with normal renal function (CLcr >90 mL/min).
Pharmacodynamics
Benazepril
Single and multiple doses of 10 mg or more of benazepril cause inhibition of plasma ACE activity by at least 80% to 90% for at least 24 hours after dosing. For up to 4 hours after a 10 mg dose, pressor responses to exogenous angiotensin I were inhibited by 60% to 90%.
In normal human volunteers, single doses of benazepril caused an increase in renal blood flow but had no effect on glomerular filtration rate.
Hydrochlorothiazide
After oral administration of hydrochlorothiazide, diuresis begins within 2 hours, peaks in about 4 hours and lasts about 6 to 12 hours.
Drug Interactions
Benazepril hydrochloride and hydrochlorothiazide potentiates the antihypertensive action of other antihypertensive drugs (e.g., curare derivatives, guanethidine, methyldopa, beta-blockers, vasodilators, calcium channel blockers ACE inhibitors and ARBs and DRIs).
CLINICAL STUDIES
In single-dose studies, benazepril lowered blood pressure within 1 hour, with peak reductions achieved 2 to 4 hours after dosing. The antihypertensive effect of a single dose persisted for 24 hours. In multiple-dose studies, once-daily doses of 20 mg to 80 mg decreased seated pressure (systolic/diastolic) 24 hours after dosing by about 6-12/4-7 mmHg. The reductions at trough are about 50% of those seen at peak.
Four dose-response studies of benazepril monotherapy using once-daily dosing were conducted in 470 mild-to-moderate hypertensive patients not using diuretics. The minimal effective once-daily dose of benazepril was 10 mg; further falls in blood pressure, especially at morning trough, were seen with higher doses in the studied dosing range (10 mg to 80 mg). In studies comparing the same daily dose of benazepril given as a single morning dose or as a twice-daily dose, blood pressure reductions at the time of morning trough blood levels were greater with the divided regimen.
The antihypertensive effects of benazepril were not appreciably different in patients receiving high- or low-sodium diets.
Benazepril-Hydrochlorothiazide
In 15 controlled clinical trials, 1453 healthy or hypertensive patients were exposed to benazepril and hydrochlorothiazide of which 459 were exposed for at least 6 months, 214 for at least 12 months and 25 for at least 24 months.
The combination of benazepril-hydrochlorothiazide resulted in mean placebo-adjusted decreases in sitting systolic and diastolic blood pressures of 10/6 mm Hg with 5/6.25 mg and 10/12.5 mg doses, and 20/10 mmHg with 20/25 mg dose.
In clinical trials of benazepril/hydrochlorothiazide using benazepril doses of 5/20 mg and hydrochlorothiazide doses of 6.25/25 mg, the antihypertensive effects were sustained for at least 24 hours, and they increased with increasing dose of either component. Although benazepril monotherapy is somewhat less effective in blacks than in nonblacks, the efficacy of combination therapy appears to be independent of race.
HOW SUPPLIED
Benazepril Hydrochloride and Hydrochlorothiazide Tablets, USP for oral administration, are available as
5 mg/6.25 mg
White to off-white, oblong, film-coated tablets, debossed " E 124" on one side and scored on the other side and supplied as:
NDC 0185-0236-01 bottles of 100
10 mg/12.5 mg
Pink, oblong, film-coated tablets, debossed " E 204" on one side and scored on the other side and supplied as:
NDC 0185-0325-01 bottles of 100
20 mg/12.5 mg
Lavender, oblong, film-coated tablets, debossed " E 211" on one side and scored on the other side and supplied as:
NDC 0185-0211-01 bottles of 100
20 mg/25 mg
Maroon, oblong, film-coated tablets, debossed " E 277" on one side and scored on the other side and supplied as:
NDC 0185-0277-01 bottles of 100
Each strength is supplied in bottles that contain a desiccant.
Dispense in a tight, light-resistant container as defined in the USP with a child-resistant closure, as required.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from light and moisture. Keep tightly closed.
KEEP OUT OF THE REACH OF CHILDREN.
Manufactured in India by
Sandoz Private Ltd., for
Sandoz Inc., Princeton, NJ 08540
Rev. July 2022
46309876