Get your patient on Benztropine Mesylate - Benztropine Mesylate tablet (Benztropine Mesylate)
Benztropine Mesylate - Benztropine Mesylate tablet prescribing information
INDICATIONS AND USAGE
Benztropine Mesylate Tablets, USP are indicated for use as an adjunct in the therapy of all forms of parkinsonism.
Useful also in the control of extrapyramidal disorders (except tardive dyskinesia-see PRECAUTIONS ) due to neuroleptic drugs (e.g., phenothiazines).
DOSAGE AND ADMINISTRATION
Benztropine mesylate tablets should be used when patients are able to take oral medication.
The injection is especially useful for psychotic patients with acute dystonic reactions or other reactions that make oral medication difficult or impossible. It is recommended also when a more rapid response is desired than can be obtained with the tablets.
Because of cumulative action, therapy should be initiated with a low dose which is increased gradually at five or six-day intervals to the smallest amount necessary for optimal relief. Increases should be made in increments of 0.5 mg, to a maximum of 6 mg, or until optimal results are obtained without excessive adverse reactions.
Postencephalitic and Idiopathic Parkinsonism - The usual daily dose is 1 to 2 mg, with a range of 0.5 to 6 mg orally or parenterally.
As with any agent used in parkinsonism, dosage must be individualized according to age and weight, and the type of parkinsonism being treated. Generally, older patients, and thin patients cannot tolerate large doses. Most patients with postencephalitic parkinsonism need fairly large doses and tolerate them well. Patients with a poor mental outlook are usually poor candidates for therapy.
In idiopathic parkinsonism, therapy may be initiated with a single daily dose of 0.5 to 1 mg at bedtime. In some patients, this will be adequate; in others 4 to 6 mg a day may be required.
In postencephalitic parkinsonism, therapy may be initiated in most patients with 2 mg a day in one or more doses. In highly sensitive patients, therapy may be initiated with 0.5 mg at bedtime, and increased as necessary.
Some patients experience greatest relief by taking the entire dose at bedtime; others react more favorably to divided doses, two to four times a day. Frequently, one dose a day is sufficient, and divided doses may be unnecessary or undesirable.
The long duration of action of this drug makes it particularly suitable for bedtime medication when its effects may last throughout the night, enabling patients to turn in bed during the night more easily, and to rise in the morning.
When benztropine mesylate is started, do not terminate therapy with other antiparkinsonian agents abruptly. If the other agents are to be reduced or discontinued, it must be done gradually. Many patients obtain greatest relief with combination therapy.
Benztropine mesylate may be used concomitantly with Carbidopa-Levodopa, or with levodopa, in which case periodic dosage adjustment may be required in order to maintain optimum response.
Drug-Induced Extrapyramidal Disorders - In treating extrapyramidal disorders due to neuroleptic drugs (e.g., phenothiazines), the recommended dosage is 1 to 4 mg once or twice a day orally or parenterally. Dosage must be individualized according to the need of the patient. Some patients require more than recommended; others do not need as much.
In acute dystonic reactions, 1 to 2 mL of the injection usually relieves the condition quickly. After that, the tablets, 1 to 2 mg twice a day, usually prevent recurrence.
When extrapyramidal disorders develop soon after initiation of treatment with neuroleptic drugs (e.g., phenothiazines), they are likely to be transient. One to 2 mg of benztropine mesylate tablets two or three times a day usually provides relief within one or two days. After one or two weeks, the drug should be withdrawn to determine the continued need for it. If such disorders recur, benztropine mesylate can be reinstituted.
Certain drug-induced extrapyramidal disorders that develop slowly may not respond to benztropine mesylate.
CONTRAINDICATIONS
Hypersensitivity to benztropine mesylate tablets or to any component of the tablets.
Because of its atropine-like side effects, this drug is contraindicated in pediatric patients under three years of age, and should be used with caution in older pediatric patients.
ADVERSE REACTIONS
The adverse reactions below, most of which are anticholinergic in nature, have been reported and within each category are listed in order of decreasing severity.
Cardiovascular
Tachycardia.
Digestive
Paralytic ileus, constipation, vomiting, nausea, dry mouth.
If dry mouth is so severe that there is difficulty in swallowing or speaking, or loss of appetite and weight, reduce dosage, or discontinue the drug temporarily.
Slight reduction in dosage may control nausea and still give sufficient relief of symptoms. Vomiting may be controlled by temporary discontinuation, followed by resumption at a lower dosage.
Nervous System
Toxic psychosis, including confusion, disorientation, memory impairment, visual hallucinations; exacerbation of preexisting psychotic symptoms; nervousness; depression; listlessness; numbness of fingers.
Special Senses
Blurred vision, dilated pupils.
Urogenital
Urinary retention, dysuria.
Metabolic/Immune or Skin
Occasionally, an allergic reaction, e.g., skin rash, develops. If this cannot be controlled by dosage reduction, the medication should be discontinued.
Other
Heat stroke, hyperthermia, fever.
Drug Interactions
Antipsychotic drugs such as phenothiazines or haloperidol; tricyclic antidepressants (see WARNINGS ).
DESCRIPTION
Benztropine mesylate is a synthetic compound containing structural features found in atropine and diphenhydramine.
It is designated chemically as 3α-(Diphenylmethoxy)-1αH, 5αH-tropane methanesulfonate. Its molecular formula is C 21 H 25 NO•CH 4 O 3 S, and its structural formula is:
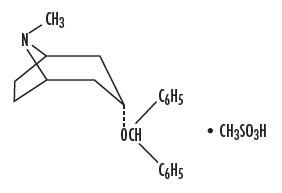
Benztropine mesylate is a crystalline white powder, very soluble in water, and has a molecular weight of 403.54.
Each benztropine mesylate tablet for oral administration contains benztropine mesylate, USP, 0.5 mg, 1 mg, or 2 mg.
Each tablet contains the following inactive ingredients: dibasic calcium phosphate (hydrous), anhydrous lactose, magnesium stearate, microcrystalline cellulose, and pregelatinized starch.
CLINICAL PHARMACOLOGY
Benztropine mesylate possesses both anticholinergic and antihistaminic effects, although only the former have been established as therapeutically significant in the management of parkinsonism.
In the isolated guinea pig ileum, the anticholinergic activity of this drug is about equal to that of atropine; however, when administered orally to unanesthetized cats, it is only about half as active as atropine.
In laboratory animals, its antihistaminic activity and duration of action approach those of pyrilamine maleate.
HOW SUPPLIED
Benztropine Mesylate Tablets, USP 0.5 mg are round, white, debossed with ‘CE’ on the left side of the functional score and ‘41’ on the right side on one side and plain on the other side. They are supplied in: Bottles of 30 NDC 62135-141-30 Bottles of 100 NDC 62135-141-01 Bottles of 180 NDC 62135-141-18 Bottles of 540 NDC 62135-141-57 Bottles of 1000 NDC 62135-141-10
Benztropine Mesylate Tablets, USP 1 mg are oval shaped, white, debossed with ‘CE’ on the left side of the functional score and ‘44’ on the right side on one side and plain on the other side.
They are supplied in:
Bottles of 30 NDC 62135-144-30
Bottles of 100 NDC 62135-144-01
Bottles of 180 NDC 62135-144-18
Bottles of 540 NDC 62135-144-57
Bottles of 1000 NDC 62135-144-10
Benztropine Mesylate Tablets, USP 2 mg are round, white, debossed with ‘CE’ on the upper side of the functional score and ‘42’ on the lower side on one side and plain on the other side.
They are supplied in:
Bottles of 30 NDC 62135-142-30
Bottles of 100 NDC 62135-142-01
Bottles of 180 NDC 62135-142-18
Bottles of 540 NDC 62135-142-57
Bottles of 1000 NDC 62135-142-10
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
| Dispense in a tight, light-resistant container [see USP]. |
• Duvoisin, R.C.; Katz, R.J; Amer. Med. Ass. 206: 1963-1965, Nov. 25, 1968.
Manufactured by: Chartwell Pharmaceuticals, LLC.
Congers, NY 10920
Manufactured for: Chartwell RX, LLC.
Congers, NY 10920
Made in USA
Rev: 08/2022
L70507