Brevital Sodium - Methohexital Sodium injection, Powder, Lyophilized, For Solution prescribing information
WARNING
BREVITAL should be used only in hospital or ambulatory care settings that provide for continuous monitoring of respiratory (e.g. pulse oximetry) and cardiac function. Immediate availability of resuscitative drugs and age- and size-appropriate equipment for bag/valve/mask ventilation and intubation and personnel trained in their use and skilled in airway management should be assured. For deeply sedated patients, a designated individual other than the practitioner performing the procedure should be present to continuously monitor the patient. (See WARNINGS .)
INDICATIONS AND USAGE
BREVITAL can be used in adults as follows:
1. For intravenous induction of anesthesia prior to the use of other general anesthetic agents.
2. For intravenous induction of anesthesia and as an adjunct to subpotent inhalational anesthetic agents (such as nitrous oxide in oxygen) for short surgical procedures; BREVITAL may be given by infusion or intermittent injection.
3. For use along with other parenteral agents, usually narcotic analgesics, to supplement subpotent inhalational anesthetic agents (such as nitrous oxide in oxygen) for longer surgical procedures.
4. As intravenous anesthesia for short surgical, diagnostic, or therapeutic procedures associated with minimal painful stimuli (see WARNINGS ).
5. As an agent for inducing a hypnotic state.
BREVITAL can be used in pediatric patients older than 1 month as follows:
1. For rectal or intramuscular induction of anesthesia prior to the use of other general anesthetic agents.
2. For rectal or intramuscular induction of anesthesia and as an adjunct to subpotent inhalational anesthetic agents for short surgical procedures.
3. As rectal or intramuscular anesthesia for short surgical, diagnostic, or therapeutic procedures associated with minimal painful stimuli.
DOSAGE AND ADMINISTRATION
Facilities for assisting ventilation and administering oxygen are necessary adjuncts for all routes of administration of anesthesia. Since cardiorespiratory arrest may occur, patients should be observed carefully during and after use of BREVITAL. Age- and size- appropriate resuscitative equipment (i.e., intubation and cardioversion equipment, oxygen, suction, and a secure intravenous line) and personnel qualified in its use must be immediately available.
Preanesthetic medication is generally advisable. BREVITAL may be used with any of the recognized preanesthetic medications.
Preparation of Solution
FOLLOW DILUTION INSTRUCTIONS EXACTLY.
Freshly prepare solutions of BREVITAL and use promptly. Reconstituted solutions of BREVITAL are chemically stable at room temperature for 24 hours.
Diluents
ONLY USE BACTERIOSTATIC-FREE DILUENT – Recommended diluents are based on route of administration (see Dilution Instructions ).
Incompatible diluents: Lactated Ringer’s Injection
Dilution Instructions
1% solutions (10 mg/mL) should be prepared for intermittent intravenous and rectal administration; 0.2% solutions (2 mg/mL) should be prepared for continuous intravenous drug administration; 5% solutions (50 mg/mL) should be prepared for intramuscular administration.
Contents of vials should be diluted as follows:
FOR INTERMITTENT INTRAVENOUS and RECTAL ADMINISTRATION
The preferred diluent for intermittent intravenous and rectal administration is Sterile Water for Injection. 5% Dextrose Injection, or 0.9% Sodium Chloride Injection are also acceptable diluents.
| Strength | Amount of Diluent to Be Added to the Contents of the BREVITAL Vial | For 1% methohexital solution (10 mg/mL) |
|---|---|---|
500 mg | 50 mL | no further dilution needed |
FOR CONTINUOUS INTRAVENOUS ADMINISTRATION
For continuous drip anesthesia, prepare a 0.2% solution by adding 500 mg of BREVITAL Sodium to 250 mL of diluent. For this dilution, either 5% glucose solution or isotonic (0.9%) sodium chloride solution ONLY is recommended as the diluent instead of sterile water for injection in order to avoid extreme hypotonicity.
| Strength | Amount of Diluent to Be Added to the Contents of the BREVITAL Vial | For 0.2% methohexital solution (2 mg/mL) |
|---|---|---|
500 mg | 15 mL | add to 235 mL diluent for 250 mL total volume |
FOR INTRAMUSCULAR ADMINISTRATION
The preferred diluent for intramuscular administration is Sterile Water for Injection. 0.9% Sodium Chloride Injection is also an acceptable diluent.
| Strength | Amount of Diluent to Be Added to the Contents of the BREVITAL Vial | For 5% methohexital solution (50 mg/mL) |
|---|---|---|
500 mg | 10 mL | no further dilution needed |
Administration
Dosage is highly individualized; the drug should be administered only by those completely familiar with its quantitative differences from other barbiturate anesthetics.
Adults
BREVITAL is administered intravenously in a concentration of no higher than 1%. Higher concentrations markedly increase the incidence of muscular movements and irregularities in respiration and blood pressure.
Induction of anesthesia
For induction of anesthesia, a 1% solution is administered at a rate of about 1 mL/5 seconds. Gaseous anesthetics and/or skeletal muscle relaxants may be administered concomitantly. The dose required for induction may range from 50 to 120 mg or more but averages about 70 mg. The usual dosage in adults ranges from 1 to 1.5 mg/kg. The induction dose usually provides anesthesia for 5 to 7 minutes.
Maintenance of anesthesia
Maintenance of anesthesia may be accomplished by intermittent injections of the 1% solution or, more easily, by continuous intravenous drip of a 0.2% solution. Intermittent injections of about 20 to 40 mg (2 to 4 mL of a 1% solution) may be given as required, usually every 4 to 7 minutes. For continuous drip, the average rate of administration is about 3 mL of a 0.2% solution/minute (1 drop/second). The rate of flow must be individualized for each patient. For longer surgical procedures, gradual reduction in the rate of administration is recommended (see discussion of prolonged administration in WARNINGS ). Other parenteral agents, usually narcotic analgesics, are ordinarily employed along with BREVITAL during longer procedures.
Pediatric Patients
BREVITAL is administered intramuscularly in a 5% concentration and administered rectally as a 1% solution.
Induction of anesthesia
For the induction of anesthesia by the intramuscular route of administration, the usual dose ranges from 6.6 to 10 mg/kg of the 5% concentration. For rectal administration, the usual dose for induction is 25 mg/kg using the 1% solution.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Compatibility Information
Solutions of BREVITAL should not be mixed in the same syringe or administered simultaneously during intravenous infusion through the same needle with acid solutions, such as atropine sulfate, metocurine iodide, and succinylcholine chloride. Alteration of pH may cause free barbituric acid to be precipitated. Solubility of the soluble sodium salts of barbiturates, including BREVITAL, is maintained only at a relatively high (basic) pH.
Because of numerous requests from anesthesiologists for information regarding the chemical compatibility of these mixtures, the following chart contains information obtained from compatibility studies in which a 1% solution of BREVITAL was mixed with therapeutic amounts of agents whose solutions have a low (acid) pH.
| Active Ingredient | Potency per mL | Volume Used | Immediate | 15 min | Physical Change 30 min | 1 h |
|---|---|---|---|---|---|---|
BREVITAL | 10 mg | 10 mL | CONTROL | |||
Atropine Sulfate | 1/150 gr | 1 mL | None | Haze | ||
Atropine Sulfate | 1/100 gr | 1 mL | None | Ppt | Ppt | |
Succinylcholine chloride | 0.5 mg | 4 mL | None | None | Haze | |
Succinylcholine chloride | 1 mg | 4 mL | None | None | Haze | |
Metocurine Iodide | 0.5 mg | 4 mL | None | None | Ppt | |
Metocurine Iodide | 1 mg | 4 mL | None | None | Ppt | |
Scopolamine Hydrobromide | 1/120 gr | 1 mL | None | None | None | Haze |
Tubocurarine chloride | 3 mg | 4 mL | None | Haze |
CONTRAINDICATIONS
BREVITAL is contraindicated in patients in whom general anesthesia is contraindicated, in those with latent or manifest porphyria, or in patients with a known hypersensitivity to barbiturates.
ADVERSE REACTIONS
Side effects associated with BREVITAL are extensions of pharmacologic effects and include:
Cardiovascular
Circulatory depression, thrombophlebitis, hypotension, tachycardia, peripheral vascular collapse, and convulsions in association with cardiorespiratory arrest
Respiratory
Respiratory depression (including apnea), cardiorespiratory arrest, laryngospasm, bronchospasm, hiccups, and dyspnea
Neurologic
Skeletal muscle hyperactivity (twitching), injury to nerves adjacent to injection site, and seizures
Psychiatric
Emergence delirium, restlessness, and anxiety may occur, especially in the presence of postoperative pain
Gastrointestinal
Nausea, emesis, abdominal pain, and liver function tests abnormal
Allergic
Erythema, pruritus, urticaria, and cases of anaphylaxis have been reported rarely
Other
Other adverse reactions include pain at injection site, salivation, headache, and rhinitis
For medical advice about adverse reactions contact your medical professional. To report SUSPECTED ADVERSE REACTIONS, contact Endo at 1-800-828-9393 or FDA at 1-800-FDA-1088 (1-800-332-1088) or www.fda.gov/medwatch/.
Drug Interactions
Prior chronic administration of barbiturates or phenytoin (e.g. for seizure disorder) appears to reduce the effectiveness of BREVITAL. Barbiturates may influence the metabolism of other concomitantly used drugs, such as phenytoin, halothane, anticoagulants, corticosteroids, ethyl alcohol, and propylene glycol-containing solutions.
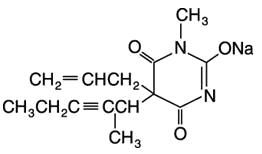
DESCRIPTION
BREVITAL ® (methohexital sodium, USP) for injection, is 2,4,6 (1 H , 3 H , 5 H )-Pyrimidinetrione, 1-methyl-5-(1-methyl-2-pentynyl)-5-(2-propenyl)-, (±)-, monosodium salt and has the empirical formula C 14 H 17 N 2 NaO 3 . Its molecular weight is 284.29.
The structural formula is as follows:
Methohexital sodium is a rapid, ultrashort-acting barbiturate anesthetic. Methohexital sodium for injection is a freeze-dried, sterile, nonpyrogenic mixture of methohexital sodium with 6% anhydrous sodium carbonate added as a buffer. It contains not less than 90% and not more than 110% of the labeled amount of methohexital sodium. It occurs as a white, freeze-dried plug that is freely soluble in water.
This product is oxygen sensitive. The pH of the 1% solution is between 10 and 11; the pH of the 0.2% solution in 5% dextrose is between 9.5 and 10.5.
Methohexital sodium may be administered by direct intravenous injection or continuous intravenous drip, intramuscular or rectal routes (see PRECAUTIONS/Pediatric Use ). Reconstituting instructions vary depending on the route of administration (see DOSAGE AND ADMINISTRATION ).
CLINICAL PHARMACOLOGY
Compared with thiamylal and thiopental, methohexital is at least twice as potent on a weight basis, and its duration of action is only about half as long. Although the metabolic fate of methohexital in the body is not clear, the drug does not appear to concentrate in fat depots to the extent that other barbiturate anesthetics do. Thus, cumulative effects are fewer and recovery is more rapid with methohexital than with thiobarbiturates. In experimental animals, the drug cannot be detected in the blood 24 hours after administration.
Methohexital differs chemically from the established barbiturate anesthetics in that it contains no sulfur. Little analgesia is conferred by barbiturates; their use in the presence of pain may result in excitation.
Intravenous administration of methohexital results in rapid uptake by the brain (within 30 seconds) and rapid induction of sleep.
Following intramuscular administration to pediatric patients, the onset of sleep occurs in 2 to 10 minutes. A plasma concentration of 3 mcg/mL was achieved in pediatric patients 15 minutes after an intramuscular dose (10 mg/kg) of a 5% solution. Following rectal administration to pediatric patients, the onset of sleep occurs in 5 to 15 minutes. Plasma methohexital concentrations achieved following rectal administration tend to increase both with dose and with the use of more dilute solution concentrations when using the same dose. A 25 mg/kg dose of a 1% methohexital solution yielded plasma concentrations of 6.9 to 7.9 mcg/mL 15 minutes after dosing. The absolute bioavailability of rectal methohexital sodium is 17%.
With single doses, the rate of redistribution determines duration of pharmacologic effect. Metabolism occurs in the liver through demethylation and oxidation. Side-chain oxidation is the most important biotransformation involved in termination of biologic activity. Excretion occurs via the kidneys through glomerular filtration.
HOW SUPPLIED
Store between 20° to 25°C (68° to 77°F). (See USP Controlled Room Temperature.)
BREVITAL ® Vials•:
The 500 mg vials (with 30 mg anhydrous sodium carbonate) are available as follows:
NDC 42023-105-01 – Multi-Dose Vial – Pack of 1
•In crystalline form.