Get your patient on Carbamazepine - Carbamazepine capsule, Extended Release (Carbamazepine)
Carbamazepine - Carbamazepine capsule, Extended Release prescribing information
WARNING
SERIOUS DERMATOLOGIC REACTIONS AND HLA-B•1502 ALLELE
SERIOUS AND SOMETIMES FATAL DERMATOLOGIC REACTIONS, INCLUDING TOXIC EPIDERMAL NECROLYSIS (TEN) AND STEVENS-JOHNSON SYNDROME (SJS), HAVE BEEN REPORTED DURING TREATMENT WITH CARBAMAZEPINE. THESE REACTIONS ARE ESTIMATED TO OCCUR IN 1 TO 6 PER 10,000 NEW USERS IN COUNTRIES WITH MAINLY CAUCASIAN POPULATIONS, BUT THE RISK IN SOME ASIAN COUNTRIES IS ESTIMATED TO BE ABOUT 10 TIMES HIGHER. STUDIES IN PATIENTS OF CHINESE ANCESTRY HAVE FOUND A STRONG ASSOCIATION BETWEEN THE RISK OF DEVELOPING SJS/TEN AND THE PRESENCE OF HLA-B•1502, AN INHERITED ALLELIC VARIANT OF THE HLA-B GENE. HLA-B•1502 IS FOUND ALMOST EXCLUSIVELY IN PATIENTS WITH ANCESTRY ACROSS BROAD AREAS OF ASIA. PATIENTS WITH ANCESTRY IN GENETICALLY AT-RISK POPULATIONS SHOULD BE SCREENED FOR THE PRESENCE OF HLA-B•1502 PRIOR TO INITIATING TREATMENT WITH CARBAMAZEPINE EXTENDED-RELEASE CAPSULES. PATIENTS TESTING POSITIVE FOR THE ALLELE SHOULD NOT BE TREATED WITH CARBAMAZEPINE EXTENDED-RELEASE CAPSULES UNLESS THE BENEFIT CLEARLY OUTWEIGHS THE RISK ( SEE WARNINGS AND PRECAUTIONS/LABORATORY TESTS ).
APLASTIC ANEMIA AND AGRANULOCYTOSIS
APLASTIC ANEMIA AND AGRANULOCYTOSIS HAVE BEEN REPORTED IN ASSOCIATION WITH THE USE OF CARBAMAZEPINE. DATA FROM A POPULATION-BASED CASE-CONTROL STUDY DEMONSTRATE THAT THE RISK OF DEVELOPING THESE REACTIONS IS 5 to 8 TIMES GREATER THAN IN THE GENERAL POPULATION. HOWEVER, THE OVERALL RISK OF THESE REACTIONS IN THE UNTREATED GENERAL POPULATION IS LOW, APPROXIMATELY SIX PATIENTS PER ONE MILLION POPULATION PER YEAR FOR AGRANULOCYTOSIS AND TWO PATIENTS PER ONE MILLION POPULATION PER YEAR FOR APLASTIC ANEMIA.
ALTHOUGH REPORTS OF TRANSIENT OR PERSISTENT DECREASED PLATELET OR WHITE BLOOD CELL COUNTS ARE NOT UNCOMMON IN ASSOCIATION WITH THE USE OF CARBAMAZEPINE, DATA ARE NOT AVAILABLE TO ESTIMATE ACCURATELY THEIR INCIDENCE OR OUTCOME. HOWEVER, THE VAST MAJORITY OF THE CASES OF LEUKOPENIA HAVE NOT PROGRESSED TO THE MORE SERIOUS CONDITIONS OF APLASTIC ANEMIA OR AGRANULOCYTOSIS.
BECAUSE OF THE VERY LOW INCIDENCE OF AGRANULOCYTOSIS AND APLASTIC ANEMIA, THE VAST MAJORITY OF MINOR HEMATOLOGIC CHANGES OBSERVED IN MONITORING OF PATIENTS ON CARBAMAZEPINE ARE UNLIKELY TO SIGNAL THE OCCURRENCE OF EITHER ABNORMALITY. NONETHELESS, COMPLETE PRETREATMENT HEMATOLOGICAL TESTING SHOULD BE OBTAINED AS A BASELINE. IF A PATIENT IN THE COURSE OF TREATMENT EXHIBITS LOW OR DECREASED WHITE BLOOD CELL OR PLATELET COUNTS, THE PATIENT SHOULD BE MONITORED CLOSELY. DISCONTINUATION OF THE DRUG SHOULD BE CONSIDERED IF ANY EVIDENCE OF SIGNIFICANT BONE MARROW DEPRESSION DEVELOPS.
INDICATIONS AND USAGE
Epilepsy
Carbamazepine extended-release capsules are indicated for use as an anticonvulsant drug. Evidence supporting efficacy of carbamazepine as an anticonvulsant was derived from active drug-controlled studies that enrolled patients with the following seizure types:
- Partial seizures with complex symptomatology (psychomotor, temporal lobe). Patients with these seizures appear to show greater improvements than those with other types.
- Generalized tonic-clonic seizures (grand mal).
- Mixed seizure patterns which include the above, or other partial or generalized seizures. Absence seizures (petit mal) do not appear to be controlled by carbamazepine (see PRECAUTIONS, General ).
Trigeminal Neuralgia
Carbamazepine extended-release capsules are indicated in the treatment of the pain associated with true trigeminal neuralgia. Beneficial results have also been reported in glossopharyngeal neuralgia. This drug is not a simple analgesic and should not be used for the relief of trivial aches or pains.
DOSAGE AND ADMINISTRATION
Monitoring of blood levels has increased the efficacy and safety of anticonvulsants (see PRECAUTIONS, Laboratory Tests ). Dosage should be adjusted to the needs of the individual patients. A low initial daily dosage with gradual increase is advised. As soon as adequate control is achieved, the dosage may be reduced very gradually to the minimum effective level. Carbamazepine extended-release capsules may be taken with or without food. Carbamazepine extended-release capsules may be swallowed whole or may be opened and all the beads sprinkled on a teaspoon of soft food such as applesauce. Make sure all of the food and medicine mixture is swallowed. Do not crush or chew carbamazepine extended-release capsules or the sprinkled beads.
Carbamazepine extended-release capsule is an extended-release formulation for twice a day administration. When converting patients from immediate release carbamazepine to carbamazepine extended-release capsules, the same total daily mg dose of carbamazepine should be administered. Following conversion to carbamazepine, patients should be closely monitored for seizure control. Depending on the therapeutic response after conversion, the total daily dose may need to be adjusted within the recommended dosing instructions.
Epilepsy (see INDICATIONS AND USAGE )
Adults and children over 12 years of age. Initial: 200 mg twice daily. Increase at weekly intervals by adding up to 200 mg/day until the optimal response is obtained. Dosage generally should not exceed 1,000 mg per day in children 12 to 15 years of age, and 1,200 mg daily in patients above 15 years of age. Doses up to 1,600 mg daily have been used in adults.
Maintenance: Adjust dosage to the minimum effective level, usually 800 to 1,200 mg daily.
Children under 12 years of age: Children taking total daily dosages of immediate-release carbamazepine of 400 mg or greater may be converted to the same total daily dosage of carbamazepine extended-release capsules, using a twice daily regimen. Ordinarily, optimal clinical response is achieved at daily doses below 35 mg/kg. If satisfactory clinical response has not been achieved, plasma levels should be measured to determine whether or not they are in the therapeutic range. No recommendation regarding the safety of carbamazepine extended-release capsules for use at doses above 35 mg/kg/24 hours can be made.
Combination Therapy: Carbamazepine extended-release capsules may be used alone or with other anticonvulsants. When added to existing anticonvulsant therapy, the drug should be added gradually while the other anticonvulsants are maintained or gradually decreased, except phenytoin, which may have to be increased (see PRECAUTIONS, Drug Interactions ).
Trigeminal Neuralgia (see INDICATIONS AND USAGE )
Initial: On the first day, start with one 200 mg capsule. This daily dose may be increased by up to 200 mg/day every 12 hours only as needed to achieve freedom from pain. Do not exceed 1,200 mg daily.
Maintenance: Control of pain can be maintained in most patients with 400 to 800 mg daily. However, some patients may be maintained on as little as 200 mg daily, while others may require as much as 1200 mg daily. At least once every 3 months throughout the treatment period, attempts should be made to reduce the dose to the minimum effective level or even to discontinue the drug.
CONTRAINDICATIONS
Carbamazepine should not be used in patients with a history of previous bone marrow depression, hypersensitivity to the drug, or known sensitivity to any of the tricyclic compounds, such as amitriptyline, desipramine, imipramine, protriptyline and nortriptyline. Likewise, on theoretical grounds its use with monoamine oxidase inhibitors is not recommended. Before administration of carbamazepine, MAO inhibitors should be discontinued for a minimum of 14 days, or longer if the clinical situation permits.
Coadministration of carbamazepine and nefazodone may result in insufficient plasma concentrations of nefazodone and its active metabolite to achieve a therapeutic effect. Coadministration of carbamazepine with nefazodone is contraindicated.
Coadministration of carbamazepine is contraindicated with delavirdine due to the potential for loss of virologic response and possible resistance to delavirdine or to the class of non-nucleoside reverse transcriptase inhibitors.
ADVERSE REACTIONS
General :
If adverse reactions are of such severity that the drug must be discontinued, the physician must be aware that abrupt discontinuation of any anticonvulsant drug in a responsive patient with epilepsy may lead to seizures or even status epilepticus with its life-threatening hazards.
The most severe adverse reactions previously observed with carbamazepine were reported in the hemopoietic system and skin (see BOXED WARNING ), and the cardiovascular system.
The most frequently observed adverse reactions, particularly during the initial phases of therapy, are dizziness, drowsiness, unsteadiness, nausea, and vomiting. To minimize the possibility of such reactions, therapy should be initiated at the lowest dosage recommended.
The following additional adverse reactions were previously reported with carbamazepine:
Hemopoietic System:
Aplastic anemia, agranulocytosis, pancytopenia, bone marrow depression, thrombocytopenia, leukopenia, leukocytosis, eosinophilia, acute intermittent porphyria.
Skin:
Toxic epidermal necrolysis (TEN) and Stevens-Johnson syndrome (SJS) (see BOXED WARNING ), Acute Generalized Exanthematous Pustulosis (AGEP), pruritic and erythematous rashes, urticaria, photosensitivity reactions, alterations in skin pigmentation, exfoliative dermatitis, erythema multiforme and nodosum, purpura, aggravation of disseminated lupus erythematosus, alopecia, diaphoresis, onychomadesis and hirsutism. In certain cases, discontinuation of therapy may be necessary.
Cardiovascular System :
Congestive heart failure, edema, aggravation of hypertension, hypotension, syncope and collapse, aggravation of coronary artery disease, arrhythmias and AV block, thrombophlebitis, thromboembolism, and adenopathy or lymphadenopathy. Some of these cardiovascular complications have resulted in fatalities. Myocardial infarction has been associated with other tricyclic compounds.
Immune system disorders : Hypogammaglobulinemia.
Liver :
Abnormalities in liver function tests, cholestatic and hepatocellular jaundice, hepatitis, and hepatic failure.
Pancreatic :
Pancreatitis.
Respiratory System:
Pulmonary hypersensitivity characterized by fever, dyspnea, pneumonitis, or pneumonia.
Genitourinary System :
Urinary frequency, acute urinary retention, oliguria with elevated blood pressure, azotemia, renal failure, and impotence. Albuminuria, glycosuria, elevated BUN, and microscopic deposits in the urine have also been reported. There have been rare reports of impaired male fertility and/or abnormal spermatogenesis.
Testicular atrophy occurred in rats receiving carbamazepine orally from 4 to 52 weeks at dosage levels of 50 to 400 mg/kg/day. Additionally, rats receiving carbamazepine in the diet for 2 years at dosage levels of 25, 75, and 250 mg/kg/day had a dose-related incidence of testicular atrophy and aspermatogenesis. In dogs, it produced a brownish discoloration, presumably a metabolite, in the urinary bladder at dosage levels of 50 mg/kg/day and higher. Relevance of these findings to humans is unknown.
Nervous System :
Dizziness, drowsiness, disturbances of coordination, confusion, headache, fatigue, blurred vision, visual hallucinations, transient diplopia, oculomotor disturbances, nystagmus, speech disturbances, abnormal involuntary movements, peripheral neuritis and paresthesias, depression with agitation, talkativeness, tinnitus, and hyperacusis.
There have been reports of associated paralysis and other symptoms of cerebral arterial insufficiency, but the exact relationship of these reactions to the drug has not been established.
Isolated cases of neuroleptic malignant syndrome have been reported with concomitant use of psychotropic drugs.
Digestive System :
Nausea, vomiting, gastric distress and abdominal pain, diarrhea, constipation, anorexia, and dryness of the mouth and pharynx, including glossitis and stomatitis.
Eyes:
Scattered punctate cortical lens opacities, as well as conjunctivitis, have been reported. Although a direct causal relationship has not been established, many phenothiazines and related drugs have been shown to cause eye changes.
Musculoskeletal System:
Bone loss, aching joints and muscles, and leg cramps.
Metabolism :
Fever and chills, decreased levels of plasma calcium leading to osteoporosis, and hyperammonemia have been reported.
Other:
Isolated cases of a lupus erythematosus-like syndrome have been reported. There have been occasional reports of elevated levels of cholesterol, HDL cholesterol, and triglycerides in patients taking anticonvulsants.
A case of aseptic meningitis, accompanied by myoclonus and peripheral eosinophilia, has been reported in a patient taking carbamazepine in combination with other medications. The patient was successfully dechallenged, and the meningitis reappeared upon rechallenge with carbamazepine.
Drug Interactions
Clinically meaningful drug interactions have occurred with concomitant medications and include, but are not limited to the following:
Agents Highly Bound to Plasma Protein:
Carbamazepine is not highly bound to plasma proteins; therefore, administration of carbamazepine extended-release capsules to a patient taking another drug that is highly protein bound should not cause increased free concentrations of the other drug.
Agents that Inhibit Cytochrome P450 Isoenzymes and/or Epoxide Hydrolase:
Carbamazepine is metabolized mainly by cytochrome P450 (CYP) 3A4 to the active carbamazepine 10, 11-epoxide, which is further metabolized to the trans-diol by epoxide hydrolase. Therefore, the potential exists for interaction between carbamazepine and any agent that inhibits CYP3A4 and/or epoxide hydrolase. Agents that are CYP3A4 inhibitors that have been found, or are expected, to increase plasma levels of carbamazepine extended-release capsules include, for example, the following:
Acetazolamide, aprepitant, azole antifungals (e.g., ketoconazole, itraconazole, fluconazole, voriconazole), cimetidine, ciprofloxacin, clarithromycin, dalfopristin, danazol, dantrolene, delavirdine, diltiazem, erythromycin, fluoxetine, fluvoxamine, grapefruit juice, ibuprofen, isoniazid, loratadine, macrolides, nefazodone, niacinamide, nicotinamide, olanzapine, omeprazole, oxybutynin, protease inhibitors, propoxyphene, quinine, quinupristin, ticlopidine, troleandomycin, valproate, verapamil, zileuton.
Human microsomal epoxide hydrolase has been identified as the enzyme responsible for the formation of the 10,11-transdiol derivative from carbamazepine-10,11 epoxide. Coadministration of inhibitors of human microsomal epoxide hydrolase may result in increased carbamazepine-10,11 epoxide plasma concentrations. Accordingly, the dosage of carbamazepine extended-release capsules should be adjusted and/or the plasma levels monitored when used concomitantly with loxapine, quetiapine, or valproic acid. Thus, if a patient has been titrated to a stable dosage of carbamazepine extended-release capsules, and then begins a course of treatment with one of these CYP3A4 or epoxide hydrolase inhibitors, it is reasonable to expect that a dose reduction for carbamazepine extended-release capsules may be necessary.
Agents that Induce Cytochrome P450 Isoenzymes :
Carbamazepine is metabolized by CYP3A4. Therefore, the potential exists for interaction between carbamazepine and any agent that induces CYP3A4. Agents that are CYP inducers that have been found, or are expected, to decrease plasma levels of carbamazepine extended-release capsules include, for example, the following:
Aminophylline, cisplatin, doxorubicin HCL, felbamate, fosphenytoin, methsuximide, phenobarbital, phenytoin( 1 ), primidone, rifampin and theophylline.
(1) Phenytoin plasma levels have also been reported to increase and decrease in the presence of carbamazepine, see below.
Thus, if a patient has been titrated to a stable dosage on carbamazepine extended-release capsules, and then begins a course of treatment with one of these CYP3A4 inducers, it is reasonable to expect that a dose increase for carbamazepine extended-release capsules may be necessary.
Agents with Decreased Levels in the Presence of Carbamazepine due to Induction of Cytochrome P450 Enzymes:
Carbamazepine is a potent inducer of hepatic CYP3A4 and is also known to be an inducer of CYP1A2, 2B6, 2C9/19 and may therefore reduce plasma concentrations of co-medications mainly metabolized by CYP 1A2, 2B6, 2C9/19 and 3A4, through induction of their metabolism. When used concomitantly with carbamazepine extended-release capsules, monitoring of concentrations or dosage adjustment of these agents may be necessary:
- When carbamazepine is added to aripiprazole, the aripiprazole dose should be doubled. Additional dose increases should be based on clinical evaluation. If carbamazepine is later withdrawn, the aripiprazole dose should be reduced.
- When carbamazepine is used with tacrolimus, monitoring of tacrolimus blood concentrations and appropriate dosage adjustments are recommended.
- The use of concomitant strong CYP3A4 inducers such as carbamazepine should be avoided with temsirolimus. If patients must be coadministered carbamazepine with temsirolimus, an adjustment of temsirolimus dosage should be considered.
- The use of carbamazepine with lapatinib should generally be avoided. If carbamazepine is started in a patient already taking lapatinib, the dose of lapatinib should be gradually titrated up. If carbamazepine is discontinued, the lapatinib dose should be reduced.
- Concomitant use of carbamazepine with nefazodone results in plasma concentrations of nefazodone and its active metabolite insufficient to achieve a therapeutic effect. Coadministration of carbamazepine with nefazodone is contraindicated (see CONTRAINDICATIONS ).
- Monitor concentrations of valproate when carbamazepine extended-release capsules are introduced or withdrawn in patients using valproic acid.
In addition, carbamazepine causes, or would be expected to cause, decreased levels of, for example, the following drugs, for which monitoring of concentrations or dosage adjustment may be necessary:
Acetaminophen, albendazole, alprazolam, aprepitant, buprenorphine, apixaban( 6 ), bupropion, buspirone, citalopram, clobazam, clonazepam, clozapine, corticosteroids (e.g. prednisolone, dexamethasone), cyclosporin, dabigatran( 6 ), delavirdine, desipramine, diazepam, dicumarol, dihydropyridine calcium channel blockers (e.g., felodipine), doxycycline, edoxaban( 6 ), eslicarbazepine, ethosuximide, everolimus, felbamate, haloperidol, imatinib, itraconazole, lamotrigine, levothyroxine, lorazepam, methadone, methsuximide, midazolam, mirtazapine, nefazodone, nortriptyline, olanzapine, oral and other hormonal contraceptives( 2 ), oxcarbazepine, paliperidone, phenytoin( 3 ), praziquantel, protease inhibitors, quetiapine, risperidone, rivaroxaban( 6 ), sertraline, sirolimus, tadalafil, theophylline, tiagabine, topiramate, tramadol, triazolam, trazodone( 4 ), tricyclic antidepressants (e.g., imipramine, amitriptyline, nortriptyline), valproate, warfarin (5) , ziprasidone, and zonisamide.
( 2 ) Concomitant use of carbamazepine with hormonal contraceptive products (e.g., oral and levonorgestrel subdermal implant contraceptives) may render the contraceptives less effective because the plasma concentrations of the hormones may be decreased. Breakthrough bleeding and unintended pregnancies have been reported with carbamazepine. Alternative or back-up methods of contraception should be considered.
( 3 ) Phenytoin has also been reported to increase in the presence of carbamazepine. Careful monitoring of phenytoin plasma levels following co-medication with carbamazepine is advised.
( 4 ) Following co-administration of carbamazepine 400 mg/day with trazodone 100 mg to 300 mg daily, carbamazepine reduced trough plasma concentrations of trazodone (as well as meta-chlorophenylpiperazine [mCPP]) by 76 and 60% respectively, compared to precarbamazepine values.
(5) Warfarin’s anticoagulant effect can be reduced in the presence of carbamazepine.
(6) Concomitant use of carbamazepine with rivaroxaban, apixaban, dabigatran, and edoxaban (direct acting oral anticoagulants) is expected to result in decreased plasma concentrations of these anticoagulants that may be insufficient to achieve the intended therapeutic effect. In general, coadministration of carbamazepine with rivaroxaban, apixaban, dabigatran, and edoxaban should be avoided.
Thus, if a patient has been titrated to a stable dosage on one of the agents in this category, and then begins a course of treatment with carbamazepine extended-release capsules, it is reasonable to expect that a dose increase for the concomitant agent may be necessary.
Agents with Increased Levels in the Presence of Carbamazepine
Carbamazepine extended-release capsules increase the plasma levels of the following agents.
Thus, if a patient has been titrated to a stable dosage on one of the agents in this category, and then begins a course of the treatment with carbamazepine extended-release capsules, it is reasonable to expect that a dose decrease for the concomitant agent may be necessary.
Clomipramine HCl, Phenytoin( 7 ), and Primidone
Carbamazepine extended-release capsules can increase the concentrations of clomipramine, phenytoin, and primidone. If a patient has been titrated to a stable dosage on one of these agents in this category, and then begins treatment with carbamazepine extended-release capsules, it may be necessary to decrease the dose of these drugs.
( 7 )Phenytoin has also been reported to decrease in the presence of carbamazepine. Careful monitoring of phenytoin plasma levels following co-medication with carbamazepine is advised.
Cyclophosphamide
Cyclophosphamide is an inactive prodrug and is converted to its active metabolite in part by CYP3A. The rate of metabolism and the leukopenic activity of cyclophosphamide are reportedly increased by chronic co-administration of CYP3A4 inducers. There is a potential for increased cyclophosphamide toxicity when coadministered with carbamazepine.
Pharmacological/Pharmacodynamic Interactions with Carbamazepine:
Delavirdine
Coadministration of carbamazepine with delavirdine may lead to loss of virologic response and possible resistance to delavirdine or to the class of non-nucleoside reverse transcriptase inhibitors (see CONTRAINDICATIONS ).
Lithium
Concomitant administration of carbamazepine and lithium may increase the risk of neurotoxic side effects.
Thyroid Effects with Other Anticonvulsant Medications
Alterations of thyroid function have been reported in combination therapy with other anticonvulsant medications.
Chloroquine and Mefloquine
Anti-malarial drugs, such as chloroquine and mefloquine, may antagonize the activity of carbamazepine.
Thus if a patient has been titrated to a stable dosage on one of the agents in this category, and then begins a course of treatment with carbamazepine extended-release capsules, it is reasonable to expect that a dose adjustment may be necessary.
CNS Depressants
The concomitant use of carbamazepine extended-release capsules and other CNS depressants can increase the risk of respiratory depression, profound sedation, hypotension, and syncope. CNS depressants include: alcohol, opioid analgesics, benzodiazepines, tricyclic antidepressants, sedative/hypnotics, anticonvulsants, antipsychotics, antihistamines, anticholinergics, alpha and beta blockers, general anesthetics, muscle relaxants, and illicit CNS depressants. Consider reducing the dose of CNS depressants or carbamazepine extended-release capsules when using these drugs concomitantly. Because of its primary CNS effect, caution should be used when carbamazepine extended-release capsules are taken with other centrally acting drugs and alcohol.
Neuromuscular Blocking Agents
Resistance to the neuromuscular blocking action of the nondepolarizing neuromuscular blocking agents pancuronium, vecuronium, rocuronium and cisatracurium has occurred in patients chronically administered carbamazepine. Whether or not carbamazepine has the same effect on other nondepolarizing agents is unknown. Patients should be monitored closely for more rapid recovery from neuromuscular blockade than expected, and infusion rate requirements may be higher.
DESCRIPTION
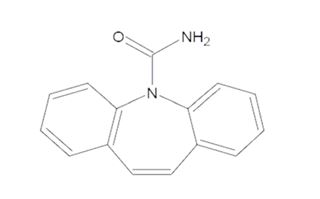
Carbamazepine extended-release capsules are an anticonvulsant and specific analgesic for trigeminal neuralgia, available for oral administration as 100 mg, 200 mg and 300 mg extended-release capsules of Carbamazepine, USP. Carbamazepine is a white to off-white powder, practically insoluble in water and soluble in alcohol and in acetone. Its molecular weight is 236.27 g/mol. Its chemical name is 5H-dibenz[b,f]azepine-5-carboxamide, and its structural formula is:
Carbamazepine extended-release capsules are a multi-component capsule formulation consisting of extended-release granules.
Inactive ingredients: colloidal silicon dioxide, ethylcellulose, and stearic acid.
The 100 mg, 200 mg and 300 mg capsule shells contain FD&C Blue #2, gelatin, iron oxide yellow, titanium dioxide and are imprinted with black ink (S-1-8114 and S-1-8115) contain FD&C BLUE #2, FD&C RED #40, FD&C BLUE #1, D&C YELLOW #10 and shellac.
CLINICAL PHARMACOLOGY
In controlled clinical trials, carbamazepine has been shown to be effective in the treatment of psychomotor and grand mal seizures, as well as trigeminal neuralgia.
Mechanism of Action
Carbamazepine has demonstrated anticonvulsant properties in rats and mice with electrically and chemically induced seizures. It appears to act by reducing polysynaptic responses and blocking the post-tetanic potentiation. Carbamazepine greatly reduces or abolishes pain induced by stimulation of the infraorbital nerve in cats and rats. It depresses thalamic potential and bulbar and polysynaptic reflexes, including the linguomandibular reflex in cats. Carbamazepine is chemically unrelated to other anticonvulsants or other drugs used to control the pain of trigeminal neuralgia. The mechanism of action remains unknown.
The principal metabolite of carbamazepine, carbamazepine-10, 11-epoxide, has anticonvulsant activity as demonstrated in several in vivo animal models of seizures. Though clinical activity for the epoxide has been postulated, the significance of its activity with respect to the safety and efficacy of carbamazepine has not been established.
Pharmacokinetics
Carbamazepine (CBZ):
Taken every 12 hours, carbamazepine extended-release capsules provide steady state plasma levels comparable to immediate-release carbamazepine tablets given every 6 hours, when administered at the same total mg daily dose.
Following a single 200 mg oral extended-release dose of carbamazepine, peak plasma concentration was 1.9 ± 0.3 mcg/mL and the time to reach the peak was 19 ± 7 hours. Following chronic administration (800 mg every 12 hours), the peak levels were 11.0 ± 2.5 mcg/mL and the time to reach the peak was 5.9 ± 1.8 hours. The pharmacokinetics of extended-release carbamazepine is linear over the single dose range of 200 to 800 mg.
Carbamazepine is 76% bound to plasma proteins. Carbamazepine is primarily metabolized in the liver. Cytochrome P450 3A4 was identified as the major isoform responsible for the formation of carbamazepine-10, 11-epoxide. Since carbamazepine induces its own metabolism, the half-life is also variable. Following a single extended-release dose of carbamazepine, the average half-life range from 35 to 40 hours and 12 to 17 hours on repeated dosing. The apparent oral clearance following a single dose was 25 ± 5 mL/min and following multiple dosing was 80 ± 30 mL/min.
After oral administration of 14 C-carbamazepine, 72% of the administered radioactivity was found in the urine and 28% in the feces. This urinary radioactivity was composed largely of hydroxylated and conjugated metabolites, with only 3% of unchanged carbamazepine.
Carbamazepine-10, 11-epoxide (CBZ-E):
Carbamazepine-10, 11-epoxide is considered to be an active metabolite of carbamazepine. Following a single 200 mg oral extended-release dose of carbamazepine, the peak plasma concentration of carbamazepine-10, 11-epoxide was 0.11 ± 0.012 mcg/mL and the time to reach the peak was 36 ± 6 hours. Following chronic administration of an extended-release dose of carbamazepine (800 mg every 12 hours), the peak levels of carbamazepine-10, 11-epoxide were 2.2 ± 0.9 mcg/mL and the time to reach the peak was 14 ± 8 hours. The plasma half-life of carbamazepine-10, 11-epoxide following administration of carbamazepine is 34 ± 9 hours. Following a single oral dose of extended-release carbamazepine (200 to 800 mg) the AUC and C max of carbamazepine-10, 11-epoxide were less than 10% of carbamazepine. Following multiple dosing of extended-release carbamazepine (800 to 1,600 mg daily for 14 days), the AUC and C max of carbamazepine-10, 11-epoxide were dose related, ranging from 15.7 mcg·hr/mL and 1.5 mcg/mL at 800 mg/day to 32.6 mcg·hr/mL and 3.2 mcg/mL at 1,600 mg/day, respectively, and were less than 30% of carbamazepine. Carbamazepine-10, 11-epoxide is 50% bound to plasma proteins.
Food Effect:
A high fat meal diet increased the rate of absorption of a single 400 mg dose (mean T max was reduced from 24 hours, in the fasting state, to 14 hours and C max increased from 3.2 to 4.3 mcg/mL) but not the extent (AUC) of absorption. The elimination half-life remains unchanged between fed and fasting state. The multiple dose study conducted in the fed state showed that the steady-state C max values were within the therapeutic concentration range. The pharmacokinetic profile of extended-release carbamazepine was similar when given by sprinkling the beads over applesauce compared to the intact capsule administered in the fasted state.
Special Populations
Hepatic Dysfunction:
The effect of hepatic impairment on the pharmacokinetics of carbamazepine is not known. However, given that carbamazepine is primarily metabolized in the liver, it is prudent to proceed with caution in patients with hepatic dysfunction.
Renal Dysfunction:
The effect of renal impairment on the pharmacokinetics of carbamazepine is not known.
Gender:
No difference in the mean AUC and C max of carbamazepine and carbamazepine-10, 11-epoxide was found between males and females.
Age:
Carbamazepine is more rapidly metabolized to carbamazepine-10, 11-epoxide in young children than adults. In children below the age of 15, there is an inverse relationship between CBZ-E/CBZ ratio and increasing age.
Race:
No information is available on the effect of race on the pharmacokinetics of carbamazepine.
HOW SUPPLIED
Carbamazepine extended-release capsules 100 mg are hard gelatin capsules with white opaque body and blue-green opaque cap, imprinted “APO C100” in black ink with white to off-white granule fill. They are supplied as follows:
Bottles of 30s (NDC 60505-2805-3) Bottles of 120s (NDC 60505-2805-7) Bottles of 1000s (NDC 60505-2805-8)
Carbamazepine extended-release capsules 200 mg are hard gelatin capsules with white opaque body and blue-green opaque cap, imprinted “APO C200” in black ink with white to off-white granule fill. They are supplied as follows:
Bottles of 30s (NDC 60505-2806-3) Bottles of 120s (NDC 60505-2806-7) Bottles of 1000s (NDC 60505-2806-8)
Carbamazepine extended-release capsules 300 mg are hard gelatin capsules with white opaque body and blue-green opaque cap, imprinted “APO C300” in black ink with white to off-white granule fill. They are supplied as follows:
Bottles of 30s (NDC 60505-2807-3) Bottles of 120s (NDC 60505-2807-7) Bottles of 500s (NDC 60505-2807-5)
Store at 20˚C to 25°C (68˚F to 77°F); excursions permitted to 15˚C to 30°C (59˚F to 86°F) [see USP Controlled Room Temperature]. Dispense in a tight, light-resistant container [see USP].
Dispense with Medication Guide available at https://www.apotex.com/products/us/mg.asp
APOTEX INC. CARBAMAZEPINE EXTENDED-RELEASE CAPSULES 100 mg, 200 mg and 300 mg
| Manufactured by: Apotex Inc. Toronto, Ontario Canada M9L 1T9 | Manufactured for: Apotex Corp. Weston, Florida USA 33326 |
Rev. 11
Carbamazepine - Carbamazepine capsule, Extended Release PubMed™ news
- Journal Article • 2026 MayCombined exposure to nano-TiO₂ and carbamazepine exacerbates reproductive toxicity in mussels Mytilus coruscus by disrupting steroid hormone homeostasis.
- Journal Article • 2026 MayCombined internal neurolysis and targeted proximal trigeminal root glycerol delivery for refractory multiple sclerosis-related trigeminal neuralgia: a case report.
- Journal Article • 2026 MayA prospective observational study evaluating drug-related problems and their impact on patient-related outcomes in hospitalized epileptic patients.
- Journal Article • 2026 MayA Brain-Permeable Selenocyanate Near-Infrared (NIR) Probe Platform for H(2)S Dynamics Imaging: Insights into Redox Homeostasis and Antiepileptic Therapy.
- Journal Article • 2026 MayFate, dissipation, and mass balance of antipsychotic pharmaceuticals in outdoor aquatic mesocosms.