Get your patient on Carboplatin - Carboplatin injection, Solution (Carboplatin)
Carboplatin - Carboplatin injection, Solution prescribing information
WARNING
Carboplatin Injection should be administered under the supervision of a qualified physician experienced in the use of cancer chemotherapeutic agents. Appropriate management of therapy and complications is possible only when adequate treatment facilities are readily available.
Bone marrow suppression is dose related and may be severe, resulting in infection and/or bleeding. Anemia may be cumulative and may require transfusion support. Vomiting is another frequent drug-related side effect.
Anaphylactic-like reactions to carboplatin have been reported and may occur within minutes of carboplatin administration. Epinephrine, corticosteroids, and antihistamines have been employed to alleviate symptoms.
INDICATIONS:
Initial Treatment of Advanced Ovarian Carcinoma
Carboplatin Injection is indicated for the initial treatment of advanced ovarian carcinoma in established combination with other approved chemotherapeutic agents. One established combination regimen consists of carboplatin and cyclophosphamide. Two randomized controlled studies conducted by the NCIC and SWOG with carboplatin versus cisplatin, both in combination with cyclophosphamide, have demonstrated equivalent overall survival between the two groups (see CLINICAL STUDIES ).
There is limited statistical power to demonstrate equivalence in overall pathologic complete response rates and long-term survival (≥3 years) because of the small number of patients with these outcomes: the small number of patients with residual tumor <2 cm after initial surgery also limits the statistical power to demonstrate equivalence in this subgroup.
Secondary Treatment of Advanced Ovarian Carcinoma
Carboplatin Injection is indicated for the palliative treatment of patients with ovarian carcinoma recurrent after prior chemotherapy, including patients who have been previously treated with cisplatin.
Within the group of patients previously treated with cisplatin, those who have developed progressive disease while receiving cisplatin therapy may have a decreased response rate.
DOSAGE AND ADMINISTRATION:
NOTE: Aluminum reacts with carboplatin causing precipitate formation and loss of potency, therefore, needles or intravenous sets containing aluminum parts that may come in contact with the drug must not be used for the preparation or administration of Carboplatin Injection.
Single-Agent Therapy
Carboplatin Injection, as a single agent, has been shown to be effective in patients with recurrent ovarian carcinoma at a dosage of 360 mg/m 2 IV on day 1 every 4 weeks (alternatively see Formula Dosing ). In general, however, single intermittent courses of Carboplatin Injection should not be repeated until the neutrophil count is at least 2,000 and the platelet count is at least 100,000.
Combination Therapy with Cyclophosphamide
In the chemotherapy of advanced ovarian cancer, an effective combination for previously untreated patients consists of: Carboplatin Injection–300 mg/m 2 IV on day 1 every 4 weeks for 6 cycles (alternatively see Formula Dosing ).
Cyclophosphamide–600 mg/m 2 IV on day 1 every 4 weeks for 6 cycles. For directions regarding the use and administration of cyclophosphamide please refer to its package insert (see CLINICAL STUDIES ).
Intermittent courses of Carboplatin Injection in combination with cyclophosphamide should not be repeated until the neutrophil count is at least 2,000 and the platelet count is at least 100,000.
Dose Adjustment Recommendations
Pretreatment platelet count and performance status are important prognostic factors for severity of myelosuppression in previously treated patients.
The suggested dose adjustments for single agent or combination therapy shown in the table below are modified from controlled trials in previously treated and untreated patients with ovarian carcinoma. Blood counts were done weekly, and the recommendations are based on the lowest post-treatment platelet or neutrophil value.
| Platelets | Neutrophils | Adjusted Dose • (From Prior Course) |
| > 100,000 | > 2000 | 125% |
| 50 to 100,000 | 500 to 2000 | No Adjustment |
| < 50,000 | <500 | 75% |
•Percentages apply to Carboplatin Injection as a single agent or to both carboplatin and cyclophosphamide in combination. In the controlled studies, dosages were also adjusted at a lower level (50 to 60%) for severe myelosuppression. Escalations above 125% were not recommended for these studies.
Carboplatin Injection is usually administered by an infusion lasting 15 minutes or longer. No pre- or post-treatment hydration or forced diuresis is required.
Patients with Impaired Kidney Function
Patients with creatinine clearance values below 60 mL/min are at increased risk of severe bone marrow suppression. In renally-impaired patients who received single-agent carboplatin therapy, the incidence of severe leukopenia, neutropenia, or thrombocytopenia has been about 25% when the dosage modifications in the table below have been used.
| Baseline | |
| Creatinine Clearance | Recommended Dose on Day 1 |
| 41 to 59 mL/min | 250 mg/m 2 |
| 16 to 40 mL/min | 200 mg/m 2 |
The data available for patients with severely impaired kidney function (creatinine clearance below 15 mL/min) are too limited to permit a recommendation for treatment.
These dosing recommendations apply to the initial course of treatment. Subsequent dosages should be adjusted according to the patient’s tolerance based on the degree of bone marrow suppression.
Formula Dosing
Another approach for determining the initial dose of Carboplatin Injection is the use of mathematical formulae, which are based on a patient’s pre-existing renal function or renal function and desired platelet nadir. Renal excretion is the major route of elimination for carboplatin (see CLINICAL PHARMACOLOGY ). The use of dosing formulae, as compared to empirical dose calculation based on body surface area, allows compensation for patient variations in pretreatment renal function that might otherwise result in either underdosing (in patients with above average renal function) or overdosing (in patients with impaired renal function).
A simple formula for calculating dosage, based upon a patient’s glomerular filtration rate (GFR in mL/min) and Carboplatin Injection target area under the concentration versus time curve (AUC in mg/mL•min), has been proposed by Calvert. In these studies, GFR was measured by 51 Cr-EDTA clearance.
| CALVERT FORMULA FOR CARBOPLATIN DOSING |
| Total Dose (mg) = (target AUC) x (GFR + 25) |
| Note: With the Calvert formula, the total dose of carboplatin is calculated in mg, not mg/m 2 . |
The target AUC of 4 mg/mL•min to 6 mg/mL•min using single-agent carboplatin appears to provide the most appropriate dose range in previously treated patients. This study also showed a trend between the AUC of single-agent carboplatin administered to previously treated patients and the likelihood of developing toxicity.
| AUC (mg/mL • min) | % Actual Toxicity in Previously Gr 3 or Gr 4 Thrombocytopenia | Treated Patients Gr 3 or Gr 4 Leukopenia |
| 4 to 5 | 16% | 13% |
| 6 to 7 | 33% | 34% |
Geriatric Dosing
Because renal function is often decreased in elderly patients, formula dosing of carboplatin based on estimates of GFR should be used in elderly patients to provide predictable plasma carboplatin AUCs and thereby minimize the risk of toxicity.
CONTRAINDICATIONS:
Carboplatin Injection is contraindicated in patients with a history of severe allergic reactions to cisplatin or other platinum-containing compounds.
Carboplatin Injection should not be employed in patients with severe bone marrow depression or significant bleeding.
ADVERSE REACTIONS:
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For a comparison of toxicities when carboplatin or cisplatin was given in combination with cyclophosphamide, see CLINICAL STUDIES , Use with Cyclophosphamide for Initial Treatment of Ovarian Cancer, Comparative Toxicity.
| ADVERSE EXPERIENCES IN PATIENTS WITH OVARIAN CANCER | ||
| First Line Combination Therapy • Percent | Second Line Single Agent Therapy •• Percent | |
| Bone Marrow | ||
| Thrombocytopenia | ||
| < 100,000/mm 3 | 66 | 62 |
| < 50,000/mm 3 | 33 | 35 |
| Neutropenia | ||
| < 2,000 cells/mm 3 | 96 | 67 |
| < 1,000 cells/mm 3 | 82 | 21 |
| Leukopenia | ||
| < 4,000 cells/mm 3 | 97 | 85 |
| < 2,000 cells/mm 3 | 71 | 26 |
| Anemia | ||
| < 11/g/dL | 90 | 90 |
| < 8 g/dL | 14 | 21 |
| Infections | 16 | 5 |
| Bleeding | 8 | 5 |
| Transfusions | 35 | 44 |
| Gastrointestinal | ||
| Nausea and vomiting | 93 | 92 |
| Vomiting | 83 | 81 |
| Other GI side effects | 46 | 21 |
| Neurologic | ||
| Peripheral neuropathies | 15 | 6 |
| Ototoxicity | 12 | 1 |
| Other sensory side effects | 5 | 1 |
| Central neurotoxicity | 26 | 5 |
| Renal | ||
| Serum creatinine elevations | 6 | 10 |
| Blood urea elevations | 17 | 22 |
| Hepatic | ||
| Bilirubin elevations | 5 | 5 |
| SGOT elevations | 20 | 19 |
| Alkaline phosphatase elevations | 29 | 37 |
| Electrolytes loss | ||
| Sodium | 10 | 47 |
| Potassium | 16 | 28 |
| Calcium | 16 | 31 |
| Magnesium | 61 | 43 |
| Other side effects | ||
| Pain | 44 | 23 |
| Asthenia | 41 | 11 |
| Cardiovascular | 19 | 6 |
| Respiratory | 10 | 6 |
| Allergic | 11 | 2 |
| Genitourinary | 10 | 2 |
| Alopecia | 49 | 2 |
| Mucositis | 8 | 1 |
•Use with Cyclophosphamide for Initial Treatment of Ovarian Cancer
Data are based on the experience of 393 patients with ovarian cancer (regardless of baseline status) who received initial combination therapy with carboplatin and cyclophosphamide in two randomized controlled studies conducted by SWOG and NCIC (see CLINICAL STUDIES ).
Combination with cyclophosphamide as well as duration of treatment may be responsible for the differences that can be noted in the adverse experience table.
••Single Agent Use for the Secondary Treatment of Ovarian Cancer
Data are based on the experience of 553 patients with previously treated ovarian carcinoma (regardless of baseline status) who received single-agent carboplatin.
In the narrative section that follows, the incidences of adverse events are based on data from 1,893 patients with various types of tumors who received carboplatin as single-agent therapy.
Hematologic Toxicity
Bone marrow suppression is the dose-limiting toxicity of carboplatin. Thrombocytopenia with platelet counts below 50,000/mm 3 occurs in 25% of the patients (35% of pretreated ovarian cancer patients); neutropenia with granulocyte counts below 1,000/mm 3 occurs in 16% of the patients (21% of pretreated ovarian cancer patients); leukopenia with WBC counts below 2,000/mm 3 occurs in 15% of the patients (26% of pretreated ovarian cancer patients). The nadir usually occurs about day 21 in patients receiving single-agent therapy. By day 28, 90% of patients have platelet counts above 100,000/mm 3 ; 74% have neutrophil counts above 2,000/mm 3 ; 67% have leukocyte counts above 4,000/mm 3 .
Marrow suppression is usually more severe in patients with impaired kidney function. Patients with poor performance status have also experienced a higher incidence of severe leukopenia and thrombocytopenia.
The hematologic effects, although usually reversible, have resulted in infectious or hemorrhagic complications in 5% of the patients treated with carboplatin, with drug-related death occurring in less than 1% of the patients. Fever has also been reported in patients with neutropenia.
Anemia with hemoglobin less than 11 g/dL has been observed in 71% of the patients who started therapy with a baseline above that value. The incidence of anemia increases with increasing exposure to carboplatin. Transfusions have been administered to 26% of the patients treated with carboplatin (44% of previously treated ovarian cancer patients).
Bone marrow depression may be more severe when carboplatin is combined with other bone marrow suppressing drugs or with radiotherapy.
Gastrointestinal Toxicity
Vomiting occurs in 65% of the patients (81% of previously treated ovarian cancer patients) and in about one-third of these patients it is severe. Carboplatin, as a single agent or in combination, is significantly less emetogenic than cisplatin; however, patients previously treated with emetogenic agents, especially cisplatin, appear to be more prone to vomiting. Nausea alone occurs in an additional 10% to 15% of patients. Both nausea and vomiting usually cease within 24 hours of treatment and are often responsive to antiemetic measures. Although no conclusive efficacy data exist with the following schedules, prolonged administration of carboplatin, either by continuous 24-hour infusion or by daily pulse doses given for 5 consecutive days, was associated with less severe vomiting than the single dose intermittent schedule. Emesis was increased when carboplatin was used in combination with other emetogenic compounds. Other gastrointestinal effects observed frequently were pain, in 17% of the patients; diarrhea, in 6%; and constipation, also in 6%.
Neurologic Toxicity
Peripheral neuropathies have been observed in 4% of the patients receiving carboplatin (6% of pretreated ovarian cancer patients) with mild paresthesias occurring most frequently. Carboplatin therapy produces significantly fewer and less severe neurologic side effects than does therapy with cisplatin. However, patients older than 65 years and/or previously treated with cisplatin appear to have an increased risk (10%) for peripheral neuropathies. In 70% of the patients with pre-existing cisplatin-induced peripheral neurotoxicity, there was no worsening of symptoms during therapy with carboplatin. Clinical ototoxicity and other sensory abnormalities such as visual disturbances and change in taste have been reported in only 1% of the patients. Central nervous system symptoms have been reported in 5% of the patients and appear to be most often related to the use of antiemetics.
Although the overall incidence of peripheral neurologic side effects induced by carboplatin is low, prolonged treatment, particularly in cisplatin pretreated patients, may result in cumulative neurotoxicity.
Nephrotoxicity
Development of abnormal renal function test results is uncommon, despite the fact that carboplatin, unlike cisplatin, has usually been administered without high-volume fluid hydration and/or forced diuresis. The incidences of abnormal renal function tests reported are 6% for serum creatinine and 14% for blood urea nitrogen (10% and 22%, respectively, in pretreated ovarian cancer patients). Most of these reported abnormalities have been mild and about one-half of them were reversible.
Creatinine clearance has proven to be the most sensitive measure of kidney function in patients receiving carboplatin, and it appears to be the most useful test for correlating drug clearance and bone marrow suppression. Twenty-seven percent of the patients who had a baseline value of 60 mL/min or more demonstrated a reduction below this value during carboplatin therapy.
Hepatic Toxicity
The incidences of abnormal liver function tests in patients with normal baseline values were reported as follows: total bilirubin, 5%; SGOT, 15%; and alkaline phosphatase, 24%; (5%, 19%, and 37%, respectively, in pretreated ovarian cancer patients). These abnormalities have generally been mild and reversible in about one-half of the cases, although the role of metastatic tumor in the liver may complicate the assessment in many patients. In a limited series of patients receiving very high dosages of carboplatin and autologous bone marrow transplantation, severe abnormalities of liver function tests were reported.
Electrolyte Changes
The incidences of abnormally decreased serum electrolyte values reported were as follows: sodium, 29%; potassium, 20%; calcium, 22%; and magnesium, 29%; (47%, 28%, 31%, and 43%, respectively, in pretreated ovarian cancer patients). Electrolyte supplementation was not routinely administered concomitantly with carboplatin, and these electrolyte abnormalities were rarely associated with symptoms.
Allergic Reactions
Hypersensitivity to carboplatin has been reported in 2% of the patients. These allergic reactions have been similar in nature and severity to those reported with other platinum-containing compounds, ie, rash, urticaria, erythema, pruritus, and rarely bronchospasm and hypotension. Anaphylactic reactions have been reported as part of postmarketing surveillance (see WARNINGS ). These reactions have been successfully managed with standard epinephrine, corticosteroid, and antihistamine therapy.
Injection Site Reactions
Injection site reactions, including redness, swelling, and pain, have been reported during postmarketing surveillance. Necrosis associated with extravasation has also been reported.
Other Events
Pain and asthenia were the most frequently reported miscellaneous adverse effects; their relationship to the tumor and to anemia was likely. Alopecia was reported (3%). Cardiovascular, respiratory, genitourinary, and mucosal side effects have occurred in 6% or less of the patients. Cardiovascular events (cardiac failure, embolism, cerebrovascular accidents) were fatal in less than 1% of the patients and did not appear to be related to chemotherapy. Cancer-associated hemolytic uremic syndrome has been reported rarely.
Malaise, anorexia, hypertension, dehydration, and stomatitis have been reported as part of postmarketing surveillance.
DESCRIPTION:
Carboplatin Injection is supplied as a sterile, pyrogen-free solution available in 10 mg per mL multiple dose vials containing 50 mg, 150 mg, 450 mg or 600 mg of carboplatin for administration by intravenous infusion. Each mL contains: carboplatin 10 mg, and water for injection to volume.
Carboplatin is a platinum coordination compound. The chemical name for carboplatin is platinum, diammine [1,1-cyclobutane-dicarboxylato(2-)-0,0’]-, (SP-4-2), and has the following structural formula:
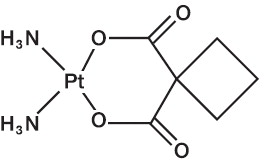
C 6 H 12 N 2 O 4 Pt M.W. 371.25
Carboplatin is a crystalline powder. It is soluble in water at a rate of approximately 14 mg/mL, and the pH of a 1% solution is 5 to 7. It is virtually insoluble in ethanol, acetone, and dimethylacetamide.
CLINICAL PHARMACOLOGY:
Carboplatin, like cisplatin, produces predominantly interstrand DNA cross-links rather than DNA-protein cross-links. This effect is apparently cell-cycle nonspecific. The aquation of carboplatin, which is thought to produce the active species, occurs at a slower rate than in the case of cisplatin. Despite this difference, it appears that both carboplatin and cisplatin induce equal numbers of drug-DNA cross-links, causing equivalent lesions and biological effects. The differences in potencies for carboplatin and cisplatin appear to be directly related to the difference in aquation rates.
In patients with creatinine clearances of about 60 mL/min, or greater, plasma levels of intact carboplatin decay in a biphasic manner after a 30-minute intravenous infusion of 300 to 500 mg/m 2 of carboplatin. The initial plasma half-life (alpha) was found to be 1.1 to 2 hours (n=6), and the post distribution plasma half-life (beta) was found to be 2.6 to 5.9 hours (n=6). The total body clearance, apparent volume of distribution and mean residence time for carboplatin are 4.4 L/hour, 16 L and 3.5 hours, respectively. The C max values and areas under the plasma concentration versus time curves from 0 to infinity (AUC inf) increase linearly with dose, although the increase was slightly more than dose proportional. Carboplatin, therefore, exhibits linear pharmacokinetics over the dosing range studied (300 to 500 mg/m 2 ). Carboplatin is not bound to plasma proteins. No significant quantities of protein-free, ultrafilterable platinum-containing species other than carboplatin are present in plasma. However, platinum from carboplatin becomes irreversibly bound to plasma proteins and is slowly eliminated with a minimum half-life of 5 days.
The major route of elimination of carboplatin is renal excretion. Patients with creatinine clearances of approximately 60 mL/min or greater excrete 65% of the dose in the urine within 12 hours and 71% of the dose within 24 hours. All of the platinum in the 24-hour urine is present as carboplatin. Only 3% to 5% of the administered platinum is excreted in the urine between 24 and 96 hours. There are insufficient data to determine whether biliary excretion occurs.
In patients with creatinine clearances below 60 mL/min, the total body and renal clearances of carboplatin decrease as the creatinine clearance decreases. Carboplatin dosages should therefore be reduced in these patients (see DOSAGE AND ADMINISTRATION ).
The primary determinant of carboplatin clearance is glomerular filtration rate (GFR) and this parameter of renal function is often decreased in elderly patients. Dosing formulas incorporating estimates of GFR (see DOSAGE AND ADMINISTRATION ) to provide predictable carboplatin plasma AUCs should be used in elderly patients to minimize the risk of toxicity.
CLINICAL STUDIES:
Use with Cyclophosphamide for Initial Treatment of Ovarian Cancer
In two prospectively randomized, controlled studies conducted by the National Cancer Institute of Canada, Clinical Trials Group (NCIC) and the Southwest Oncology Group (SWOG), 789 chemotherapy naive patients with advanced ovarian cancer were treated with carboplatin or cisplatin, both in combination with cyclophos-phamide every 28 days for 6 courses before surgical reevaluation. The following results were obtained from both studies:
| Comparative Efficacy | ||
| Overview of Pivotal Trials | ||
| NCIC | SWOG | |
| Number of patients randomized | 447 | 342 |
| Median age (years) | 60 | 62 |
| Dose of cisplatin | 75 mg/m 2 | 100 mg/m 2 |
| Dose of carboplatin | 300 mg/m 2 | 300 mg/m 2 |
| Dose of cyclophosphamide | 600 mg/m 2 | 600 mg/m 2 |
| Residual tumor < 2 cm (number of patients) | 39% (174/447) | 14% (49/342) |
| Clinical Response in Measurable Disease Patients | ||
| NCIC | SWOG | |
| Carboplatin (number of patients) | 60% (48/80) | 58% (48/83) |
| Cisplatin (number of patients) | 58% (49/85) | 43% (33/76) |
| 95% C.I. of difference (Carboplatin-Cisplatin) | (-13.9%, 18.6%) | (-2.3%, 31.1%) |
| Pathologic Complete Response • | ||
| NCIC | SWOG | |
| Carboplatin (number of patients) | 11% (24/224) | 10% (17/171) |
| Cisplatin (number of patients) | 15% (33/223) | 10% (17/171) |
| 95% C.I. of difference (Carboplatin-Cisplatin) | (-10.7%, 2.5%) | (-6.9%, 6.9%) |
•114 Carboplatin and 109 Cisplatin patients did not undergo second look surgery in NCIC study.
90 Carboplatin and 106 Cisplatin patients did not undergo second look surgery in SWOG study.
| Progression-Free Survival (PFS) | ||
| NCIC | SWOG | |
| Median | ||
| Carboplatin | 59 weeks | 49 weeks |
| Cisplatin | 61 weeks | 47 weeks |
| 2-year PFS • | ||
| Carboplatin | 31% | 21% |
| Cisplatin | 31% | 21% |
| 95% C.I. of difference (Carboplatin-Cisplatin) | (-9.3, 8.7) | (-9.0, 9.4) |
| 3-year PFS • | ||
| Carboplatin | 19% | 8% |
| Cisplatin | 23% | 14% |
| 95% C.I. of difference (Carboplatin-Cisplatin) | (-11.5, 4.5) | (-14.1, 0.3) |
| Hazard Ratio •• | 1.10 | 1.02 |
| 95% C.I. (Carboplatin-Cisplatin) | (0.89, 1.35) | (0.81, 1.29) |
•Kaplan-Meier Estimates
Unrelated deaths occurring in the absence of progression were counted as events (progression) in this analysis.
••Analysis adjusted for factors found to be of prognostic significance were consistent with unadjusted analysis.
| Survival | ||
| NCIC | SWOG | |
| Median | ||
| Carboplatin | 110 weeks | 86 weeks |
| Cisplatin | 99 weeks | 79 weeks |
| 2-year Survival • | ||
| Carboplatin | 51.9% | 40.2% |
| Cisplatin | 48.4% | 39% |
| 95% C.I. of difference (Carboplatin-Cisplatin) | (-6.2, 13.2) | (-9.8, 12.2) |
| 3-year Survival • | ||
| Carboplatin | 34.6% | 18.3% |
| Cisplatin | 33.1% | 24.9% |
| 95% C.I. of difference (Carboplatin-Cisplatin) | (-7.7, 10.7) | (-15.9, 2.7) |
| Hazard Ratio•• | ||
| 95% C.I. | 0.98 | 1.01 |
| (Carboplatin-Cisplatin) | (0.78, 1.23) | (0.78, 1.30) |
•Kaplan-Meier Estimates
••Analysis adjusted for factors found to be of prognostic significance were consistent with unadjusted analysis.
Comparative Toxicity
The pattern of toxicity exerted by the carboplatin-containing regimen was significantly different from that of the cisplatin-containing combinations. Differences between the two studies may be explained by different cisplatin dosages and by different supportive care.
The carboplatin-containing regimen induced significantly more thrombocytopenia and, in one study, significantly more leukopenia and more need for transfusional support. The cisplatin-containing regimen produced significantly more anemia in one study. However, no significant differences occurred in incidences of infections and hemorrhagic episodes.
Non-hematologic toxicities (emesis, neurotoxicity, ototoxicity, renal toxicity, hypomagnesemia, and alopecia) were significantly more frequent in the cisplatin-containing arms.
| ADVERSE EXPERIENCES IN PATIENTS WITH OVARIAN CANCER NCIC STUDY | |||
| Carboplatin Arm Percent • | Cisplatin Arm Percent • | P-Values •• | |
| Bone Marrow | |||
| Thrombocytopenia | |||
| < 100,000/mm 3 | 70 | 29 | <0.001 |
| < 50,000/mm 3 | 41 | 6 | <0.001 |
| Neutropenia | |||
| < 2,000 cells/mm 3 | 97 | 96 | n.s. |
| < 1,000 cells/mm 3 | 81 | 79 | n.s. |
| Leukopenia | |||
| < 4,000 cells/mm 3 | 98 | 97 | n.s. |
| < 2,000 cells/mm 3 | 68 | 52 | 0.001 |
| Anemia | |||
| < 11 g/dL | 91 | 91 | n.s. |
| < 8 g/dL | 18 | 12 | n.s. |
| Infections | 14 | 12 | n.s. |
| Bleeding | 10 | 4 | n.s. |
| Transfusions | 42 | 31 | 0.018 |
| Gastrointestinal | |||
| Nausea and vomiting | 93 | 98 | 0.010 |
| Vomiting | 84 | 97 | <0.001 |
| Other GI side effects | 50 | 62 | 0.013 |
| Neurologic | |||
| Peripheral neuropathies | 16 | 42 | <0.001 |
| Ototoxicity | 13 | 33 | <0.001 |
| Other sensory side effects | 6 | 10 | n.s. |
| Central neurotoxicity | 28 | 40 | 0.009 |
| Renal | |||
| Serum creatinine elevations | 5 | 13 | 0.006 |
| Blood urea elevations | 17 | 31 | <0.001 |
| Hepatic | |||
| Bilirubin elevations | 5 | 3 | n.s. |
| SGOT elevations | 17 | 13 | n.s. |
| Alkaline phosphatase elevations | — | — | — |
| Electrolytes loss | |||
| Sodium | 10 | 20 | 0.005 |
| Potassium | 16 | 22 | n.s. |
| Calcium | 16 | 19 | n.s. |
| Magnesium | 63 | 88 | <0.001 |
| Other side effects | |||
| Pain | 36 | 37 | n.s. |
| Asthenia | 40 | 33 | n.s. |
| Cardiovascular | 15 | 19 | n.s. |
| Respiratory | 8 | 9 | n.s. |
| Allergic | 12 | 9 | n.s. |
| Genitourinary | 10 | 10 | n.s. |
| Alopecia+ | 50 | 62 | 0.017 |
| Mucositis | 10 | 9 | n.s. |
•Values are in percent of evaluable patients.
••n.s.= not significant, p>0.05.
+May have been affected by cyclophosphamide dosage delivered.
| ADVERSE EXPERIENCES IN PATIENTS WITH OVARIAN CANCER SWOG STUDY | |||
| Carboplatin Arm Percent• | Cisplatin Arm Percent• | P-Values•• | |
| Bone Marrow | |||
| Thrombocytopenia | |||
| < 100,000/mm 3 | 59 | 35 | <0.001 |
| <50,000/mm 3 | 22 | 11 | 0.006 |
| Neutropenia | |||
| < 2,000 cells/mm 3 | 95 | 97 | n.s. |
| < 1,000 cells/mm 3 | 84 | 78 | n.s. |
| Leukopenia | |||
| < 4,000 cells/mm 3 | 97 | 97 | n.s. |
| < 2,000 cells/mm 3 | 76 | 67 | n.s. |
| Anemia | |||
| < 11 g/dL | 88 | 87 | n.s. |
| < 8 g/dL | 8 | 24 | < 0.001 |
| Infections | 18 | 21 | n.s. |
| Bleeding | 6 | 4 | n.s. |
| Transfusions | 25 | 33 | n.s. |
| Gastrointestinal | |||
| Nausea and vomiting | 94 | 96 | n.s. |
| Vomiting | 82 | 91 | 0.007 |
| Other GI side effects | 40 | 48 | n.s. |
| Neurologic | |||
| Peripheral neuropathies | 13 | 28 | 0.001 |
| Ototoxicity | 12 | 30 | <0.001 |
| Other sensory side effects | 4 | 6 | n.s. |
| Central neurotoxicity | 23 | 29 | n.s. |
| Renal | |||
| Serum creatinine elevations | 7 | 38 | <0.001 |
| Blood urea elevations | - | - | - |
| Hepatic | |||
| Bilirubin elevations | 5 | 3 | n.s. |
| SGOT elevations | 23 | 16 | n.s. |
| Alkaline phosphatase elevations | 29 | 20 | n.s. |
| Electrolytes loss | |||
| Sodium | - | - | - |
| Potassium | - | - | - |
| Calcium | - | - | - |
| Magnesium | 58 | 77 | <0.001 |
| Other side effects | |||
| Pain | 54 | 52 | n.s. |
| Asthenia | 43 | 46 | n.s. |
| Cardiovascular | 23 | 30 | n.s. |
| Respiratory | 12 | 11 | n.s. |
| Allergic | 10 | 11 | n.s. |
| Genitourinary | 11 | 13 | n.s. |
| Alopecia+ | 43 | 57 | 0.009 |
| Mucositis | 6 | 11 | n.s. |
•Values are in percent of evaluable patients.
••n.s.= not significant, p>0.05.
+May have been affected by cyclophosphamide dosage delivered.
Use as a Single Agent for Secondary Treatment of Advanced Ovarian Cancer
In two prospective, randomized controlled studies in patients with advanced ovarian cancer previously treated with chemotherapy, carboplatin achieved 6 clinical complete responses in 47 patients. The duration of these responses ranged from 45 to 71+ weeks.
HOW SUPPLIED:
| Product No. | NDC No. | |
| 107260 | 63323-172-60 | CARBOplatin Injection, 600 mg per 60 mL (10 mg per mL), in a 60 mL multiple dose vial packaged individually. |
The container closure is not made with natural rubber latex.
Storage
Unopened vials of Carboplatin Injection are stable to the date indicated on the package when stored at 25°C (77°F); [excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Protect from light.
Carboplatin Injection multidose vials maintain microbial, chemical, and physical stability for up to 14 days at 25°C following multiple needle entries.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Solutions for infusion should be discarded 8 hours after preparation.
Handling and Disposal
Caution should be exercised in handling and preparing carboplatin injection. Several guidelines on this subject have been published. 1-4
To minimize the risk of dermal exposure, always wear impervious gloves when handling vials containing carboplatin injection. If carboplatin injection contacts the skin, immediately wash the skin thoroughly with soap and water. If carboplatin injection contacts mucous membranes, the membranes should be flushed immediately and thoroughly with water. More information is available in the references listed below.
Carboplatin - Carboplatin injection, Solution PubMed™ news
- Journal Article • 2026 MayCarboplatin with or without nivolumab in metastatic triple-negative breast cancer: a randomized phase II trial.
- Journal Article • 2026 MayIdentifying therapeutic targets in low-grade serous ovarian carcinomas with no specific molecular profile.
- Journal Article • 2026 MayAtezolizumab + Chemotherapy in Older Patients With Lung Cancer in Japan.
- Journal Article • 2026 MayComparative neurologic toxicity profiles of chemotherapy versus immune checkpoint inhibitors in melanoma: a propensity score-matched analysis.
- Journal Article • 2026 MayCarboplatin alleviates astrocytic TDP-43 neurotoxicity by inhibiting NF-κB activation.