Get your patient on Cefadroxil - Cefadroxil powder, For Suspension (Cefadroxil)
Cefadroxil - Cefadroxil powder, For Suspension prescribing information
INDICATIONS AND USAGE
Cefadroxil for oral suspension USP is indicated for the treatment of patients with infection caused by susceptible strains of the designated organisms in the following diseases:
Urinary tract infections caused by E. coli , P. mirabilis , and Klebsiella species.
Skin and skin structure infections caused by staphylococci and/or streptococci.
Pharyngitis and/or tonsillitis caused by Streptococcus pyogenes (Group A beta-hemolytic streptococci).
Note: Only penicillin by the intramuscular route of administration has been shown to be effective in the prophylaxis of rheumatic fever. Cefadroxil monohydrate is generally effective in the eradication of streptococci from the oropharynx. However, data establishing the efficacy of cefadroxil monohydrate for the prophylaxis of subsequent rheumatic fever are not available.
Note: Culture and susceptibility tests should be initiated prior to and during therapy. Renal function studies should be performed when indicated.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefadroxil for oral suspension and other antibacterial drugs, cefadroxil for oral suspension should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
Cefadroxil for oral suspension is acid-stable and may be administered orally without regard to meals. Administration with food may be helpful in diminishing potential gastrointestinal complaints occasionally associated with oral cephalosporin therapy.
Urinary Tract Infections
For uncomplicated lower urinary tract infections (i.e., cystitis) the usual dosage is 1 or 2 g per day in a single (q.d.) or divided doses (b.i.d.).
For all other urinary tract infections the usual dosage is 2 g per day in divided doses (b.i.d.).
Skin and Skin Structure Infections
For skin and skin structure infections the usual dosage is 1 g per day in single (q.d.) or divided doses (b.i.d.).
Pharyngitis and Tonsillitis
Treatment of group A beta-hemolytic streptococcal pharyngitis and tonsillitis— 1 g per day in single (q.d.) or divided doses (b.i.d.) for 10 days.
For urinary tract infections, the recommended daily dosage for children is 30 mg/kg/day in divided doses every 12 hours. For pharyngitis, tonsillitis, and impetigo, the recommended daily dosage for children is 30 mg/kg/day in a single dose or in equally divided doses every 12 hours. For other skin and skin structure infections, the recommended daily dosage is 30 mg/kg/day in equally divided doses every 12 hours. In the treatment of beta-hemolytic streptococcal infections, a therapeutic dosage of cefadroxil for oral suspension should be administered for at least 10 days.
See chart for total daily dosage for children.
| DAILY DOSAGE OF CEFADROXIL FOR ORAL SUSPENSION | |||
| Child's Weight | 250 mg/5 mL | 500 mg/5 mL | |
| lbs | kg | ||
| 10 | 4.5 | ½ tsp | - |
| 20 | 9.1 | 1 tsp | - |
| 30 | 13.6 | 1½ tsp | - |
| 40 | 18.2 | 2 tsp | 1 tsp |
| 50 | 22.7 | 2½ tsp | 1¼ tsp |
| 60 | 27.3 | 3 tsp | 1½ tsp |
| 70 & above | 31.8 + | -- | 2 tsp |
In patients with renal impairment, the dosage of cefadroxil monohydrate should be adjusted according to creatinine clearance rates to prevent drug accumulation. The following schedule is suggested. In adults, the initial dose is 1000 mg of cefadroxil monohydrate and the maintenance dose (based on the creatinine clearance rate [mL/min/1.73 m 2 ]) is 500 mg at the time intervals listed below.
| Creatinine Clearances | Dosage Interval |
| 0 to 10 mL/min | 36 hours |
| 10 to 25 mL/min | 24 hours |
| 25 to 50 mL/min | 12 hours |
Patients with creatinine clearance rates over 50 mL/min may be treated as if they were patients having normal renal function.
| Reconstitution Directions for Oral Suspension | |
| Bottle Size | Reconstitution Directions |
| 100 mL | Suspend in a total of 67 mL water. Method: Tap bottle lightly to loosen powder. Add 67 mL of water in two portions. Shake well after each addition. |
| 75 mL | Suspend in a total of 51 mL water. Method: Tap bottle lightly to loosen powder. Add 51 mL of water in two portions. Shake well after each addition. |
| 50 mL | Suspend in a total of 34 mL water. Method: Tap bottle lightly to loosen powder. Add 34 mL of water in two portions. Shake well after each addition |
After reconstitution, store in refrigerator. Shake well before using.
Keep container tightly closed. Discard unused portion after 14 days.
CONTRAINDICATIONS
Cefadroxil monohydrate is contraindicated in patients with known allergy to the cephalosporin group of antibiotics.
ADVERSE REACTIONS
Onset of pseudomembranous colitis symptoms may occur during or after antibiotic treatment (see WARNINGS ). Dyspepsia, nausea and vomiting have been reported rarely. Diarrhea has also occurred.
Allergies (in the form of rash, urticaria, angioedema, and pruritus) have been observed. These reactions usually subsided upon discontinuation of the drug. Anaphylaxis has also been reported.
Other reactions have included hepatic dysfunction including cholestasis and elevations in serum transaminase, genital pruritus, genital moniliasis, vaginitis, moderate transient neutropenia, fever. Agranulocytosis, thrombocytopenia, idiosyncratic hepatic failure, erythema multiforme, Stevens-Johnson syndrome, serum sickness, and arthralgia have been rarely reported.
In addition to the adverse reactions listed above which have been observed in patients treated with cefadroxil, the following adverse reactions and altered laboratory tests have been reported for cephalosporin-class antibiotics:
Toxic epidermal necrolysis, abdominal pain, superinfection, renal dysfunction, toxic nephropathy, aplastic anemia, hemolytic anemia, hemorrhage, prolonged prothrombin time, positive Coombs' test, increased BUN, increased creatinine, elevated alkaline phosphatase, elevated aspartate aminotransferase (AST), elevated alanine aminotransferase (ALT), elevated bilirubin, elevated LDH, eosinophilia, pancytopenia, neutropenia.
Several cephalosporins have been implicated in triggering seizures, particularly in patients with renal impairment, when the dosage was not reduced (see DOSAGE AND ADMINISTRATION and OVERDOSAGE ). If seizures associated with drug therapy occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
DESCRIPTION
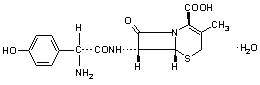
Cefadroxil monohydrate is a semisynthetic cephalosporin antibiotic intended for oral administration. It is a white to yellowish-white crystalline powder. It is soluble in water and it is acid-stable. It is chemically designated as 5-Thia-1-azabicyclo[4.2.O]oct-2-ene-2-carboxylic acid, 7-[[amino(4-hydroxyphenyl)acetyl]amino]-3-methyl-8-oxo-, monohydrate, [6R-[6(,7((R•)]]-. It has the formula C 16 H 17 N 3 O 5 S•H 2 O and the molecular weight of 381.40. It has the following structural formula:
Cefadroxil for oral suspension USP contains cefadroxil monohydrate USP. After reconstitution, each 5 mL contains cefadroxil monohydrate USP equivalent to 250 mg or 500 mg of cefadroxil. In addition, cefadroxil for oral suspension USP contains the following inactive ingredients: colloidal silicon dioxide, FD&C Yellow No. 6, powder flavor orange, powder flavor pineapple, sodium benzoate, sucrose, and xanthan gum.
Cefadroxil for oral suspension USP is a light orange colored powder, forming orange colored suspension on constitution.
CLINICAL PHARMACOLOGY
Cefadroxil monohydrate is rapidly absorbed after oral administration. Following single doses of 500 mg and 1000 mg, average peak serum concentrations were approximately 16 and 28 mcg/mL, respectively. Measurable levels were present 12 hours after administration. Over 90% of the drug is excreted unchanged in the urine within 24 hours. Peak urine concentrations are approximately 1800 mcg/mL during the period following a single 500-mg oral dose. Increases in dosage generally produce a proportionate increase in cefadroxil monohydrate urinary concentration. The urine antibiotic concentration, following a 1-g dose, was maintained well above the MIC of susceptible urinary pathogens for 20 to 22 hours.
Microbiology
In vitro tests demonstrate that the cephalosporins are bactericidal because of their inhibition of cell-wall synthesis. Cefadroxil has been shown to be active against the following organisms both in vitro and in clinical infections (see INDICATIONS AND USAGE ):
Beta-hemolytic streptococci
Staphylococci , including penicillinase-producing strains
Streptococcus (Diplococcus) pneumoniae
Escherichia coli
Proteus mirabilis
Klebsiella species
Moraxella (Branhamella) catarrhalis
Note: Most strains of Enterococcus faecalis (formerly Streptococcus faecalis ) and Enterococcus faecium (formerly Streptococcus faecium ) are resistant to cefadroxil monohydrate. It is not active against most strains of Enterobacter species, Morganella morganii (formerly Proteus morganii ), and P. vulgaris . It has no activity against Pseudomonas species and Acinetobacter calcoaceticus (formerly Mima and Herellea species).
The use of antibiotic disk susceptibility test methods which measure zone diameter give an accurate estimation of antibiotic susceptibility. One such standard procedure 1,3 which has been recommended for use with disks to test susceptibility of organisms to cefadroxil uses the cephalosporin class (cephalothin) disk. Interpretation involves the correlation of the diameters obtained in the disk test with the minimum inhibitory concentration (MIC) for cefadroxil.
Reports from the laboratory giving results of the standard single-disk susceptibility test with a 30 mcg cephalothin disk should be interpreted according to the following criteria:
| Zone Diameter ( mm ) | Interpretation | MIC ( mcg / mL ) |
| ≥18 | Susceptible (S) | ≤8 |
| 15 to 17 | Intermediate (I) | - |
| ≤14 | Resistant (R) | ≥32 |
A report of "Susceptible" indicates that the pathogen is likely to be inhibited by generally achievable blood levels. A report of "Intermediate susceptibility" suggests that the organism would be susceptible if high dosage is used or if the infection is confined to tissue and fluids (e.g., urine) in which high antibiotic levels are attained. A report of "Resistant" indicates that achievable concentrations of the antibiotic are unlikely to be inhibitory and other therapy should be selected.
Standardized procedures require the use of laboratory control organisms. The 30 mcg cephalothin disk should give the following zone diameters:
| Organism | Zone Diameter (mm) |
| Staphylococcus aureus ATCC 25923 | 29 to 37 |
| Escherichia coli ATCC 25922 | 15 to 21 |
When using the CLSI agar dilution or broth dilution (including microdilution) method 2,3 or equivalent, the MIC values should be interpreted according to the following criteria:
| MIC (mcg/mL) | Interpretation |
| ≤8 | Susceptible (S) |
| 16 | Intermediate (I) |
| ≥32 | Resistant (R) |
As with standard diffusion methods, dilution procedures require the use of laboratory control organisms. Standard cephalothin powder should provide the following MIC values:
| Microorganism | MIC (mcg/mL) | |
| Escherichia coli | ATCC 25922 | 4 to 16 |
| Staphylococcus aureus | ATCC 29213 | 0.12 to 0.5 |
HOW SUPPLIED
Cefadroxil for oral suspension USP is an orange-pineapple flavored, and is supplied as follows:
250 mg/5 mL NDC 68180-181-02 100 mL Bottle
500 mg/5 mL NDC 68180-182-02 75 mL Bottle
NDC 68180-182-03 100 mL Bottle
Prior to reconstitution: Store at 25°C (77°F); excursions permitted to 15° to 30° C (59° to 86° F). [See USP Controlled Room Temperature].
After reconstitution: Store in refrigerator. Shake well before using. Keep container tightly closed. Discard unused portion after 14 days.