Get your patient on Cefdinir - Cefdinir powder, For Suspension (Cefdinir)
Cefdinir - Cefdinir powder, For Suspension prescribing information
INDICATIONS & USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefdinir and other antibacterial drugs, cefdinir should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Cefdinir for oral suspension is indicated for the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the conditions listed below.
Adults and Adolescents Community-Acquired Pneumonia caused by Haemophilus influenzae (including β-lactamase producing strains), Haemophilus parainfluenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains) (see CLINICAL STUDIES ).
Acute Exacerbations of Chronic Bronchitis caused by Haemophilus influenzae (including β-lactamase producing strains), Haemophilus parainfluenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains). Acute Maxillary Sinusitis caused by Haemophilus influenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains).
NOTE: For information on use in pediatric patients, see Pediatric Use and DOSAGE AND ADMINISTRATION . Pharyngitis/Tonsillitis caused by Streptococcus pyogenes (see CLINICAL STUDIES ).
NOTE: Cefdinir is effective in the eradication of S. pyogenes from the oropharynx. Cefdinir has not, however, been studied for the prevention of rheumatic fever following S. pyogenes pharyngitis/tonsillitis. Only intramuscular penicillin has been demonstrated to be effective for the prevention of rheumatic fever. Uncomplicated Skin and Skin Structure Infections caused by Staphylococcus aureus (including β-lactamase producing strains) and Streptococcus pyogenes . Pediatric Patients
Acute Bacterial Otitis Media caused by Haemophilus influenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains).Pharyngitis/Tonsillitis caused by Streptococcus pyogenes (see CLINICAL STUDIES ).
NOTE: Cefdinir is effective in the eradication of S. pyogenes from the oropharynx. Cefdinir has not, however, been studied for the prevention of rheumatic fever following S. pyogenes pharyngitis/tonsillitis. Only intramuscular penicillin has been demonstrated to be effective for the prevention of rheumatic fever.
Uncomplicated Skin and Skin Structure Infections caused by Staphylococcus aureus (including β-lactamase producing strains) and Streptococcus pyogenes .
DOSAGE & ADMINISTRATION
(see INDICATIONS AND USAGE for Indicated Pathogens)
Powder for Oral Suspension
The recommended dosage and duration of treatment for infections in pediatric patients are described in the following chart; the total daily dose for all infections is 14 mg/kg, up to a maximum dose of 600 mg per day. Once-daily dosing for 10 days is as effective as BID. dosing. Once-daily dosing has not been studied in skin infections; therefore, cefdinir for oral suspension should be administered twice daily in this infection. Cefdinir for oral suspension may be administered without regard to meals. Pediatric Patients (Age 6 Months Through 12 Years)
| Type of Infection | Dosage | Duration |
| Acute Bacterial Otitis Media | 7 mg/kg q12h or 14 mg/kg q24h | 5 to 10 days 10 days |
| Acute Maxillary Sinusitis | 7 mg/kg q12h or 14 mg/kg q24h | 10 days 10 days |
| Pharyngitis/Tonsillitis | 7 mg/kg q12h or 14 mg/kg q24h | 5 to 10 days 10 days |
| Uncomplicated Skin and Skin Structure Infections | 7 mg/kg q12h | 10 days |
CEFDINIR FOR ORAL SUSPENSION PEDIATRIC DOSAGE CHART
| Weight | 125 mg/5 mL | 250 mg/5 mL |
| 9 kg/20 lbs | 2.5 mL q12h or 5 mL q24h | Use 125 mg/5 mL product |
| 18 kg/40 lbs | 5 mL q12h or 10 mL q24h | 2.5 mL q12h or 5 mL q24h |
| 27 kg/60 lbs | 7.5 mL q12h or 15 mL q24h | 3.75 mL q12h or 7.5 mL q24h |
| 36 kg/80 lbs | 10 mL q12h or 20 mL q24h | 5 mL q12h or 10 mL q24h |
| ≥43 kg• /95 lbs | 12 mL q12h or 24 mL q24h | 6 mL q12h or 12 mL q24h |
• Pediatric patients who weigh ≥ 4 3 kg should receive the maximum daily dose of 600 mg.
Patients with Renal Insufficiency
For adult patients with creatinine clearance < 30 mL/min, the dose of cefdinir should be 300 mg given once daily.
Creatinine clearance is difficult to measure in outpatients. However, the following formula may be used to estimate creatinine clearance (CLcr) in adult patients. For estimates to be valid, serum creatinine levels should reflect steady-state levels of renal function.
| Males: | CLcr = | (weight) (140 – age) |
| (72) (serum creatinine) | ||
| Females: | CLcr = | 0.85 x above value |
where creatinine clearance is in mL/min, age is in years, weight is in kilograms, and serum creatinine is in mg/dL .1 The following formula may be used to estimate creatinine clearance in pediatric patients:
| CLcr = K x | body length or height |
| serum creatinine |
where K=0.55 for pediatric patients older than 1 year 2 and 0.45 for infants (up to 1 year). 3 In the above equation, creatinine clearance is in mL/min/1.73 m 2 , body length or height is in centimeters, and serum creatinine is in mg/dL. For pediatric patients with a creatinine clearance of < 30 mL/min/1.73 m 2 , the dose of cefdinir should be 7 mg/kg (up to 300 mg) given once daily
Patients on Hemodialysis
Hemodialysis removes cefdinir from the body. In patients maintained on chronic hemodialysis, the recommended initial dosage regimen is a 300- mg or 7- mg/kg dose every other day. At the conclusion of each hemodialysis session, 300 mg (or 7 mg/kg) should be given. Subsequent doses (300 mg or 7 mg/kg) are then administered every other day. Directions for Mixing Cefdinir for Oral Suspension
| Final Concentration | Final Volume(mL) | Amount of Water | Directions |
| 125 mg/5 mL | 60 100 | 40 mL 64 mL | Tap bottle to loosen powder, then add water in 2 portions. Shake well after each aliquot. |
| 250 mg/5 mL | 60 100 | 40 mL 64 mL | Tap bottle to loosen powder, then add water in 2 portions. Shake well after each aliquot. |
After mixing, the suspension can be stored at controlled room temperature (20 ° to 25 ° C/68 ° to 77 ° F). The container should be kept tightly closed, and the suspension should be shaken well before each administration. The suspension may be used for 10 days, after which any unused portion must be discarded.
CONTRAINDICATIONS
Cefdinir is contraindicated in patients with known allergy to the cephalosporin class of antibiotics.
ADVERSE EVENTS
Clinical Trials-Cefdinir for Oral Suspension (Pediatric Patients) In clinical trials, 2289 pediatric patients (1783 U.S. and 506 non-U.S.) were treated with the recommended dose of cefdinir suspension (14 mg/kg/day). Most adverse events were mild and self-limiting. No deaths or permanent disabilities were attributed to cefdinir. Forty of 2289 (2%) patients discontinued medication due to adverse events considered by the investigators to be possibly, probably, or definitely associated with cefdinir therapy. Discontinuations were primarily for gastrointestinal disturbances, usually diarrhea. Five of 2289 (0.2%) patients were discontinued due to rash thought related to cefdinir administration. In the U.S., the following adverse events were thought by investigators to be possibly, probably, or definitely related to cefdinir suspension in multiple-dose clinical trials (N = 1783 cefdinir-treated patients):
| ADVERSE EVENTS ASSOCIATED WITH CEFDINIR SUSPENSION U.S. TRIALS IN PEDIATRIC PATIENTS (N = 1783)• | ||
| Incidence ≥ 1% | Diarrhea Rash Vomiting | 8% 3% 1% |
| Incidence < 1% but > 0.1% | Cutaneous moniliasis Abdominal pain Leukopenia† Vaginal moniliasis Vaginitis Abnormal stools Dyspepsia Hyperkinesia Increased AST† Maculopapular rash Nausea | 0.9% 0.8% 0.3% 0.3% of girls 0.3% of girls 0.2% 0.2% 0.2% 0.2% 0.2% 0.2% |
• 977 males, 806 females † Laboratory changes were occasionally reported as adverse events.
NOTE: In both cefdinir- and control-treated patients, rates of diarrhea and rash were higher in the youngest pediatric patients. The incidence of diarrhea in cefdinir-treated patients ≤ 2 years of age was 17% (95/557) compared with 4% (51/1226) in those >2 years old. The incidence of rash (primarily diaper rash in the younger patients) was 8% (43/557) in patients ≤ 2 years of age compared with 1% (8/1226) in those >2 years old. The following laboratory value changes of possible clinical significance, irrespective of relationship to therapy with cefdinir, were seen during clinical trials conducted in the U.S.:
| LABORATORY VALUE CHANGES OF POSSIBLE CLINICAL SIGNIFICANCE OBSERVED WITH CEFDINIR SUSPENSION U.S. TRIALS IN PEDIATRIC PATIENTS (N = 1783) | ||
| Incidence ≥1% | ↑Lymphocytes, ↓Lymphocytes ↑Alkaline phosphatase ↓Bicarbonate• ↑Eosinophils ↑Lactate dehydrogenase ↑Platelets ↑PMNs, ↓PMNs ↑Urine protein | 2%, 0.8% 1% 1% 1% 1% 1% 1%, 1% 1% |
| Incidence < 1% but > 0.1% | ↑Phosphorus, ↓Phosphorus ↑Urine pH ↓White blood cells, ↑White blood cells ↓Calcium• ↓Hemoglobin ↑Urine leukocytes ↑Monocytes ↑AST ↑Potassium• ↑Urine specific gravity, ↓Urine specific gravity ↓Hematocrit• | 0.9%, 0.4% 0.8% 0.7%, 0.3% 0.5% 0.5% 0.5% 0.4% 0.3% 0.3% 0.3%, 0.1% 0.2% |
• N = 1387 for these parameters
Postmarketing Experience
The following adverse experiences and altered laboratory tests, regardless of their relationship to cefdinir, have been reported during extensive postmarketing experience, beginning with approval in Japan in 1991: shock, anaphylaxis with rare cases of fatality, facial and laryngeal edema, feeling of suffocation, serum sickness-like reactions, conjunctivitis, stomatitis, Stevens-Johnson syndrome, toxic epidermal necrolysis, exfoliative dermatitis, erythema multiforme, erythema nodosum, acute hepatitis, cholestasis, fulminant hepatitis, hepatic failure, jaundice, increased amylase, acute enterocolitis, bloody diarrhea, hemorrhagic colitis, melena, pseudomembranous colitis, pancytopenia, granulocytopenia, leukopenia, thrombocytopenia, idiopathic thrombocytopenic purpura, haemolytic anemia, acute respiratory failure, asthmatic attack, drug-induced pneumonia, eosinophilic pneumonia, idiopathic interstitial pneumonia, fever, acute renal failure, nephropathy, bleeding tendency, coagulation disorder, disseminated intravascular coagulation, upper GI bleed, peptic ulcer, ileus, loss of consciousness, allergic vasculitis, possible cefdinir-diclofenac interaction, cardiac failure, chest pain, myocardial infarction, hypertension, involuntary movements, and rhabdomyolysis.
Cephalosporin Class Adverse Events
The following adverse events and altered laboratory tests have been reported for cephalosporin-class antibiotics in general: Allergic reactions, anaphylaxis, Stevens-Johnson syndrome, erythema multiforme, toxic epidermal necrolysis, renal dysfunction, toxic nephropathy, hepatic dysfunction including cholestasis, aplastic anemia, hemolytic anemia, hemorrhage, false-positive test for urinary glucose, neutropenia, pancytopenia, and agranulocytosis. Pseudomembranous colitis symptoms may begin during or after antibiotic treatment (see WARNINGS ). Several cephalosporins have been implicated in triggering seizures, particularly in patients with renal impairment when the dosage was not reduced (see DOSAGE AND ADMINISTRATION and OVERDOSAGE ). If seizures associated with drug therapy occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
DRUG INTERACTIONS
Antacids (aluminum- or magnesium-containing) Concomitant administration of 300- mg cefdinir capsules with 30 mL Maalox ® TC suspension reduces the rate (C max ) and extent (AUC) of absorption by approximately 40%. Time to reach Cmax is also prolonged by 1 hour. There are no significant effects on cefdinir pharmacokinetics if the antacid is administered 2 hours before or 2 hours after cefdinir. If antacids are required during cefdinir therapy, cefdinir should be taken at least 2 hours before or after the antacid Probenecid As with other β-lactam antibiotics, probenecid inhibits the renal excretion of cefdinir, resulting in an approximate doubling in AUC, a 54% increase in peak cefdinir plasma levels, and a 50% prolongation in the apparent elimination t 1/2 . Iron Supplements and Foods Fortified With Iron Concomitant administration of cefdinir with a therapeutic iron supplement containing 60 mg of elemental iron (as FeSO 4 ) or vitamins supplemented with 10 mg of elemental iron reduced extent of absorption by 80% and 31%, respectively. If iron supplements are required during cefdinir therapy, cefdinir should be taken at least 2 hours before or after the supplement. The effect of foods highly fortified with elemental iron (primarily iron-fortified breakfast cereals) on cefdinir absorption has not been studied. Concomitantly administered iron-fortified infant formula (2.2 mg elemental iron/6 oz) has no significant effect on cefdinir pharmacokinetics. Therefore, cefdinir for oral suspension can be administered with iron-fortified infant formula. There have been reports of reddish stools in patients receiving cefdinir. In many cases, patients were also receiving iron-containing products. The reddish color is due to the formation of a nonabsorbable complex between cefdinir or its breakdown products and iron in the gastrointestinal tract.
DESCRIPTION
Cefdinir for Oral Suspension USP contains the active ingredient cefdinir, an extended-spectrum, semisynthetic cephalosporin, for oral administration. Chemically, cefdinir is [6R-[6α, 7β (Z)]]-7-[[(2-amino-4-thiazolyl)(hydroxyimino)acetyl]amino]-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid. Cefdinir is a white to slightly brownish-yellow solid. It is slightly soluble in dilute hydrochloric acid and sparingly soluble in 0.1 M pH 7.0 phosphate buffer. The empirical formula is C 14 H 13 N 5 O 5 S 2 and the molecular weight is 395.42. Cefdinir has the structural formula shown below:
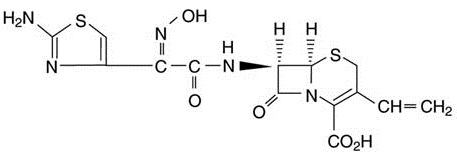
Cefdinir for Oral Suspension USP, after reconstitution, contains 125 mg cefdinir per 5 mL or 250 mg cefdinir per 5 mL and the following inactive ingredients: sucrose, sodium benzoate, xanthan gum, guar gum, colloidal silicone dioxide, magnesium stearate, sodium citrate anhydrous, citric acid anhydrous and strawberry flavoring.
CLINICAL PHARMACOLOGY
Pharmacokinetics and Drug Metabolism Absorption Oral Bioavailability Maximal plasma cefdinir concentrations occur 2 to 4 hours postdose following capsule or suspension administration. Plasma cefdinir concentrations increase with dose, but the increases are less than dose-proportional from 300 mg (7 mg/kg) to 600 mg (14 mg/kg). Following administration of suspension to healthy adults, cefdinir bioavailability is 120% relative to capsules. Estimated bioavailability of cefdinir capsules is 21% following administration of a 300 mg capsule dose, and 16% following administration of a 600 mg capsule dose. Estimated absolute bioavailability of cefdinir suspension is 25%. Cefdinir oral suspension of 250 mg/5 mL strength was shown to be bioequivalent to the 125 mg/5 mL strength in healthy adults under fasting conditions. Effect of Food The C max and AUC of cefdinir from the capsules are reduced by 16% and 10%, respectively, when given with a high-fat meal. In adults given the 250 mg/5 mL oral suspension with a high-fat meal, the Cmax and AUC of cefdinir are reduced by 44% and 33%, respectively. The magnitude of these reductions is not likely to be clinically significant because the safety and efficacy studies of oral suspension in pediatric patients were conducted without regard to food intake. Therefore, cefdinir may be taken without regard to food. Cefdinir Suspension Cefdinir plasma concentrations and pharmacokinetic parameter values following administration of single 7- and 14- mg/kg oral doses of cefdinir to pediatric subjects (age 6 months to 12 years) are presented in the following table: Mean (±SD) Plasma Cefdinir Pharmacokinetic Parameter Values Following Administration of Suspension to Pediatric Subjects
| Dose | Cmax(mcg /mL) | tmax(hr) | AUC(mcg .hr/mL) |
| 7 mg /kg | 2.30 (0 .6 5) | 2.2(0 .6 ) | 8 .31(2.50 ) |
| 14 mg /kg | 3.8 6 (0 .6 2) | 1.8 (0 .4 ) | 13.4 (2.6 4 ) |
Multiple Dosing Cefdinir does not accumulate in plasma following once- or twice-daily administration to subjects with normal renal function. Distribution The mean volume of distribution (Vd area ) of cefdinir in adult subjects is 0.35 L/kg (±0.29); in pediatric subjects (age 6 months to 12 years), cefdinir Vd area is 0.67 L/kg (±0.38). Cefdinir is 60% to 70% bound to plasma proteins in both adult and pediatric subjects; binding is independent of concentration. Skin Blister In adult subjects, median (range) maximal blister fluid cefdinir concentrations of 0.65 (0.33 to 1.1) and 1.1 (0.49 to 1.9) mcg/mL were observed 4 to 5 hours following administration of 300- and 600- mg doses, respectively. Mean (±SD) blister C max and AUC (0-∞) values were 48% (±13) and 91% (±18) of corresponding plasma values. Tonsil Tissue In adult patients undergoing elective tonsillectomy, respective median tonsil tissue cefdinir concentrations 4 hours after administration of single 300- and 600- mg doses were 0.25 (0.22 to 0.46) and 0.36 (0.22 to 0.80) mcg/g. Mean tonsil tissue concentrations were 24% (±8) of corresponding plasma concentrations. Sinus Tissue In adult patients undergoing elective maxillary and ethmoid sinus surgery, respective median sinus tissue cefdinir concentrations 4 hours after administration of single 300- and 600- mg doses were < 0.12 (< 0.12 to 0.46) and 0.21 (< 0.12 to 2.0) mcg/g. Mean sinus tissue concentrations were 16% (±20) of corresponding plasma concentrations. Lung Tissue In adult patients undergoing diagnostic bronchoscopy, respective median bronchial mucosa cefdinir concentrations 4 hours after administration of single 300- and 600- mg doses were 0.78 (< 0.06 to 1.33) and 1.14 (< 0.06 to 1.92) mcg/mL, and were 31% (±18) of corresponding plasma concentrations. Respective median epithelial lining fluid concentrations were 0.29 (< 0.3 to 4.73) and 0.49 (< 0.3 to 0.59) mcg/mL, and were 35% (±83) of corresponding plasma concentrations. Middle Ear Fluid In 14 pediatric patients with acute bacterial otitis media, respective median middle ear fluid cefdinir concentrations 3 hours after administration of single 7- and 14- mg/kg doses were 0.21 (< 0.09 to 0.94) and 0.72 (0.14 to 1.42) mcg/mL. Mean middle ear fluid concentrations were 15% (±15) of corresponding plasma concentrations. CSF Data on cefdinir penetration into human cerebrospinal fluid are not available. Metabolism and Excretion Cefdinir is not appreciably metabolized. Activity is primarily due to parent drug. Cefdinir is eliminated principally via renal excretion with a mean plasma elimination half-life (t 1/2 ) of 1.7 (±0.6) hours. In healthy subjects with normal renal function, renal clearance is 2.0 (±1.0) mL/min/kg, and apparent oral clearance is 11.6 (±6.0) and 15.5 (±5.4) mL/min/kg following doses of 300- and 600- mg, respectively. Mean percent of dose recovered unchanged in the urine following 300- and 600- mg doses is 18.4% (±6.4) and 11.6% (±4.6), respectively. Cefdinir clearance is reduced in patients with renal dysfunction (see Special Populations: Patients with Renal Insufficiency ). Because renal excretion is the predominant pathway of elimination, dosage should be adjusted in patients with markedly compromised renal function or who are undergoing hemodialysis (see DOSAGE AND ADMINISTRATION ). Special Populations Patients with Renal Insufficiency Cefdinir pharmacokinetics were investigated in 21 adult subjects with varying degrees of renal function. Decreases in cefdinir elimination rate, apparent oral clearance (CL/F), and renal clearance were approximately proportional to the reduction in creatinine clearance (CLcr). As a result, plasma cefdinir concentrations were higher and persisted longer in subjects with renal impairment than in those without renal impairment. In subjects with CLcr between 30 and 60 mL/min, C max and t 1/2 increased by approximately 2-fold and AUC by approximately 3-fold. In subjects with CLcr < 30 mL/min, C max increased by approximately 2-fold, t 1/2 by approximately 5-fold, and AUC by approximately 6-fold. Dosage adjustment is recommended in patients with markedly compromised renal function (creatinine clearance < 30 mL/min; see DOSAGE AND ADMINISTRATION ). Hemodialysis Cefdinir pharmacokinetics were studied in 8 adult subjects undergoing hemodialysis. Dialysis (4 hours duration) removed 63% of cefdinir from the body and reduced apparent elimination t 1/2 from 16 (±3.5) to 3.2 (±1.2) hours. Dosage adjustment is recommended in this patient population (see DOSAGE AND ADMINISTRATION ). Hepatic Disease Because cefdinir is predominantly renally eliminated and not appreciably metabolized, studies in patients with hepatic impairment were not conducted. It is not expected that dosage adjustment will be required in this population. Geriatric Patients The effect of age on cefdinir pharmacokinetics after a single 300- mg dose was evaluated in 32 subjects 19 to 91 years of age. Systemic exposure to cefdinir was substantially increased in older subjects (N=16), C max by 44% and AUC by 86%. This increase was due to a reduction in cefdinir clearance. The apparent volume of distribution was also reduced, thus no appreciable alterations in apparent elimination t 1/2 were observed (elderly: 2.2 ± 0.6 hours vs young: 1.8 ± 0.4 hours). Since cefdinir clearance has been shown to be primarily related to changes in renal function rather than age, elderly patients do not require dosage adjustment unless they have markedly compromised renal function (creatinine clearance < 30 mL/min, see Patients with Renal Insufficiency , above). Gender and Race The results of a meta-analysis of clinical pharmacokinetics (N=217) indicated no significant impact of either gender or race on cefdinir pharmacokinetics. Microbiology: Mechanism of Action As with other cephalosporins, bactericidal activity of cefdinir results from inhibition of cell wall synthesis. Cefdinir is stable in the presence of some, but not all, β-lactamase enzymes. As a result, many organisms resistant to penicillins and some cephalosporins are susceptible to cefdinir. Mechanism of Resistance Resistance to cefdinir is primarily through hydrolysis by some β-lactamases, alteration of penicillin-binding proteins (PBPs) and decreased permeability. Cefdinir is inactive against most strains of Enterobacter spp., Pseudomonas spp., Enterococcus spp., penicillin-resistant streptococci, and methicillin-resistant staphylococci. β-lactamase negative, ampicillin-resistant (BLNAR) H. influenza strains are typically non-susceptible to cefdinir. Antimicrobial Activity Cefdinir has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in INDICATIONS AND USAGE . Gram-Positive Bacteria Staphylococcus aureus (methicillin-susceptible strains only) Streptococcus pneumoniae (penicillin-susceptible strains only) Streptococcus pyogenes Gram-Negative Bacteria Haemophilus influenzae Haemophilus parainfluenzae Moraxella catarrhalis The following in vitro data are available, but their clinical significance is unknown. Cefdinir exhibits in vitro minimum inhibitory concentrations (MICs) of 1 mcg/mL or less against (≥ 90%) strains of the following microorganisms; however, the safety and effectiveness of cefdinir in treating clinical infections due to these microorganisms have not been established in adequate and well controlled clinical trials. Gram-Positive Bacteria Staphylococcus epidermidis (methicillin-susceptible strains only) Streptococcus agalactiae Viridans group streptococci Gram-Negative Bacteria Citrobacter koseri Escherichia coli Klebsiella pneumoniae Proteus mirabilis Susceptibility Testing For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
CLINICAL STUDIES
Community-Acquired Bacterial Pneumonia
In a controlled, double-blind study in adults and adolescents conducted in the US, cefdinir BID was compared with cefaclor 500 mg TID. Using strict evaluability and microbiologic/clinical response criteria 6 to 14 days posttherapy, the following clinical cure rates, presumptive microbiologic eradication rates, and statistical outcomes were obtained: U.S. Community-Acquired Pneumonia Study Cefdinir vs Cefaclor
| Cefdinir BID | Cefdinir TID | Outcome | |
| Clinical Cure Rates | 150/187 (80%) | 147/186 (79%) | Cefdinir equivalent to control |
| Eradication Rates Overall S. pneumoniae H. influenzae M. catarrhalis H. parainfluenzae | 177/195 (91%) 31/31 (100%) 55/65 (85%) 10/10 (100%) 81/89 (91%) | 184/200 (92%) 35/35 (100%) 60/72 (83%) 11/11 (100%) 78/82 (95%) | Cefdinir equivalent to control |
In a second controlled, investigator-blind study in adults and adolescents conducted primarily in Europe, cefdinir BID was compared with amoxicillin/clavulanate 500/125 mg TID. Using strict evaluability and clinical response criteria 6 to 14 days posttherapy, the following clinical cure rates, presumptive microbiologic eradication rates, and statistical outcomes were obtained: European Community-Acquired Pneumonia Study Cefdinir vs Amoxicillin/Clavulanate
| Cefdinir BID | Amoxicillin/ Clavulanate TID | Outcome | |
| Clinical Cure Rates | 83/104 (80%) | 86/97 (89%) | Cefdinir not equivalent to control |
| Eradication Rates Overall S. pneumoniae H. influenzae M. catarrhalis H. parainfluenzae | 85/96 (89%) 42/44 (95%) 26/35 (74%) 6/6 (100%) 11/11 (100%) | 84/90 (93%) 43/44 (98%) 21/26 (81%) 8/8 (100%) 12/12 (100%) | Cefdinir equivalent to control |
Streptococcal Pharyngitis /Tonsillitis
In four controlled studies conducted in the United States, cefdinir was compared with 10 days of penicillin in adult, adolescent, and pediatric patients. Two studies (one in adults and adolescents, the other in pediatric patients) compared 10 days of cefdinir QD or BID to penicillin 250 mg or 10 mg/kg QID. Using strict evaluability and microbiologic/clinical response criteria 5 to 10 days posttherapy, the following clinical cure rates, microbiologic eradication rates, and statistical outcomes were obtained: Pharyngitis /Tonsillitis Studies Cefdinir (10 days) vs Penicillin (10 days)
| Study | Efficacy Parameter | Cefdinir QD | Cefdinir BID | Penicillin QID | Outcome |
| Adults/ Adolescents | Eradication of S. Pyogenes Clinical Cure Rates | 192/210 (91%) | 199/217 (92%) | 181/217 (83%) | Cefdinir superior to control |
| 199/210 (95%) | 209/217 (96%) | 193/217 (89%) | Cefdinir superior to control | ||
| Pediatric Patients | Eradication of S. pyogenes Clinical Cure Rates | 215/228 (94%) | 214/227 (94%) | 159/227 (70%) | Cefdinir superior to control |
| 222/228 (97%) | 218/227 (96%) | 196/227 (86%) | Cefdinir superior to control |
Two studies (one in adults and adolescents, the other in pediatric patients) compared 5 days of cefdinir BID to 10 days of penicillin 250 mg or 10 mg/kg QID.Using strict evaluability and microbiologic/clinical response criteria 4 to 10 days posttherapy, the following clinical cure rates, microbiologic eradication rates, and statistical outcomes were obtained: Pharyngitis /Tonsillitis Studies Cefdinir (5 days) vs Penicillin (10 days)
| Study | Efficacy Parameter | Cefdinir BID | Penicillin QID | Outcome |
| Adults/ Adolescents | Eradication of S. Pyogenes Clinical Cure Rates | 193/218 (89%) | 176/214 (82%) | Cefdinir equivalent to control |
| 194/218 (89%) | 181/214 (85%) | Cefdinir equivalent to control | ||
| Pediatric Patients | Eradication of S. pyogenes Clinical Cure Rates | 176/196 (90%) | 135/193 (70%) | Cefdinir superior to control |
| 179/196 (91%) | 173/193 (90%) | Cefdinir equivalent to control |
HOW SUPPLIED
Cefdinir for Oral Suspension USP is a off white to yellow free flowing powder filled in HDPE bottle that, when reconstituted as directed, contains 125 mg cefdinir/5 mL or 250 mg cefdinir/5 mL. The reconstituted suspensions have a off white to yellow colour and strawberry flavor. The powder is available as follows: Cefdinir for Oral Suspension USP 125 mg/5 mL 60 mL bottle: NDC 67877-547-98 100 mL bottle: NDC 67877-547-88 Cefdinir for Oral Suspension USP 250 mg/5 mL 60 mL bottle: NDC 67877-548-98 100 mL bottle: NDC 67877-548-88 Store unsuspended powder at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Once reconstituted, the oral suspension can be stored at controlled room temperature for 10 days.