Get your patient on Clindacin P - Clindamycin Phosphate solution (Clindamycin Phosphate)
Clindacin P - Clindamycin Phosphate solution prescribing information
INDICATIONS AND USAGE
Clindacin•P ® is indicated in the treatment of acne vulgaris. In view of the potential for diarrhea, bloody diarrhea and pseudo-membranous colitis, the physician should consider whether other agents are more appropriate (see CONTRAINDICATIONS , WARNINGS and ADVERSE REACTIONS ).
DOSAGE AND ADMINISTRATION
Do not use if the seal on jar is broken. Remove pledget from jar just before use. Use a pledget to apply a thin film of Clindamycin Phosphate Topical Solution to the affected area twice daily. More than one pledget may be used. Each pledget should be used only once and then discarded. Keep jar tightly closed when not in use.
CONTRAINDICATIONS
Clindacin•P ® is contraindicated in individuals with a history of hypersensitivity to preparations containing clindamycin or lincomycin, a history of regional enteritis or ulcerative colitis, or a history of antibiotic-associated colitis.
ADVERSE REACTIONS
In 18 clinical studies of various formulations of topical clindamycin phosphate using placebo vehicle and/or active comparator drugs as controls, patients experienced a number of treatment emergent adverse dermatologic events [see table below].
| Number of Patients Reporting Events | |||
|---|---|---|---|
| Treatment Emergent Adverse Event | Solution n=553 (%) | Gel n=148 (%) | Lotion n=160 (%) |
| Burning | 62 (11) | 15 (10) | 17 (11) |
| Itching | 36 (7) | 15 (10) | 17 (11) |
| Burning/Itching | 60 (11) | not recorded ( – ) | ( – ) |
| Dryness | 105 (19) | 34 (23) | 29 (18) |
| Erythema | 86 (16) | 10 (7) | 22 (14) |
| Oiliness/Oily Skin | 8 (1) | 26 (18) | 12 of 126 subjects (10) |
| Peeling | 61 (11) | ( – ) | 11 (7) |
Orally and parenterally administered clindamycin has been associated with severe colitis which may end fatally. Cases of diarrhea, bloody diarrhea and colitis (including pseudomembranous colitis) have been reported as adverse reactions in patients treated with oral and parenteral formulations of clindamycin and rarely with topical clindamycin (see WARNINGS ).
Abdominal pain, gastrointestinal disturbances, gram-negative folliculitis, eye pain and contact dermatitis have also been reported in association with the use of topical formulations of clindamycin.
To report SUSPECTED ADVERSE REACTIONS, contact Medimetriks Pharmaceuticals, Inc., at 1-973-882-7512 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore it should be used with caution in patients receiving such agents.
DESCRIPTION
Clindacin•P ® contains clindamycin phosphate, USP, at a concentration equivalent to 10 mg clindamycin per milliliter.
Each Clindacin•P ® pledget applicator contains approximately 1 mL of topical solution.
Clindamycin phosphate is a water soluble ester of the semi-synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent antibiotic lincomycin.
The solution contains isopropyl alcohol 50% v/v, propylene glycol, purified water, and sodium hydroxide (to adjust the pH to between 4.0 - 7.0).
The structural formula is represented below:
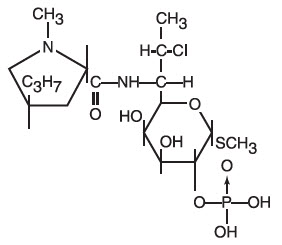
The chemical name for clindamycin phosphate is Methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl- trans -4-propyl-L-2-pyrrolidine-carboxamido)-1-thio-L- threo -α-D- galacto -octopyranoside 2-(dihydrogen phosphate).
CLINICAL PHARMACOLOGY
Although clindamycin phosphate is inactive in vitro , rapid in vivo hydrolysis converts this compound to the antibacterially active clindamycin.
Cross resistance has been demonstrated between clindamycin and lincomycin. Antagonism has been demonstrated between clindamycin and erythromycin.
Following multiple topical applications of clindamycin phosphate at a concentration equivalent to 10 mg clindamycin per mL in an isopropyl alcohol and water solution, very low levels of clindamycin are present in the serum (0–3 ng/mL) and less than 0.2% of the dose is recovered in urine as clindamycin.
Clindamycin activity has been demonstrated in comedones from acne patients. The mean concentration of antibiotic activity in extracted comedones after application of clindamycin phosphate topical solution for 4 weeks was 597 mcg/g of comedonal material (range 0–1490). Clindamycin in vitro inhibits all Propionibacterium acnes cultures tested (MICs 0.4 mcg/mL). Free fatty acids on the skin surface have been decreased from approximately 14% to 2% following application of clindamycin.
HOW SUPPLIED
Clindacin•P ® Clindamycin Phosphate Topical Solution USP, 1% (Pledgets) is available as follows:
A jar containing 69 single-use pledget applicators (NDC 43538-170-69)
STORAGE
Store at 20-25°C (68-77°F) [see USP Controlled Room Temperature]. Protect from freezing.