Get your patient on Clindamycin Phosphate - Clindamycin Phosphate cream (Clindamycin Phosphate)
Clindamycin Phosphate - Clindamycin Phosphate cream prescribing information
INDICATIONS AND USAGE
Clindamycin phosphate vaginal cream 2%, is indicated in the treatment of bacterial vaginosis (formerly referred to as Haemophilus vaginitis, Gardnerella vaginitis, nonspecific vaginitis, Corynebacterium vaginitis, or anaerobic vaginosis). Clindamycin phosphate vaginal cream 2%, can be used to treat non-pregnant women and pregnant women during the second and third trimester. (See CLINICAL STUDIES. )
NOTE: For purposes of this indication, a clinical diagnosis of bacterial vaginosis is usually defined by the presence of a homogeneous vaginal discharge that (a) has a pH of greater than 4.5, (b) emits a "fishy" amine odor when mixed with a 10% KOH solution, and (c) contains clue cells on microscopic examination. Gram's stain results consistent with a diagnosis of bacterial vaginosis include (a) markedly reduced or absent Lactobacillus morphology, (b) predominance of Gardnerella morphotype, and (c) absent or few white blood cells.
Other pathogens commonly associated with vulvovaginitis, eg, Trichomonas vaginalis , Chlamydia trachomatis, N. gonorrhoeae, Candida albicans , and Herpes simplex virus should be ruled out.
DOSAGE AND ADMINISTRATION
The recommended dose is one applicatorful of clindamycin phosphate vaginal cream 2%, (5 grams containing approximately 100 mg of clindamycin phosphate) intravaginally, preferably at bedtime, for 3 or 7 consecutive days in non-pregnant patients and for 7 consecutive days in pregnant patients. (See CLINICAL STUDIES. )
CONTRAINDICATIONS
Clindamycin phosphate vaginal cream 2%, is contraindicated in individuals with a history of hypersensitivity to clindamycin, lincomycin, or any of the components of this vaginal cream. Clindamycin phosphate vaginal cream 2%, is also contraindicated in individuals with a history of regional enteritis, ulcerative colitis, or a history of "antibiotic-associated" colitis.
ADVERSE REACTIONS
Clinical trials
Non-pregnant Women
In clinical trials involving non-pregnant women, 1.8% of 600 patients who received treatment with clindamycin phosphate vaginal cream 2% for 3 days and 2.7% of 1325 patients who received treatment for 7 days discontinued therapy due to drug-related adverse events. Medical events judged to be related, probably related, possibly related, or of unknown relationship to vaginally administered clindamycin phosphate vaginal cream 2%, were reported for 20.7% of the patients receiving treatment for 3 days and 21.3% of the patients receiving treatment for 7 days. Events occurring in ≥1% of patients receiving clindamycin phosphate vaginal cream 2% are shown in Table 1 .
| TABLE 1 – Events Occurring in ≥1% of Non-pregnant Patients Receiving Clindamycin Phosphate Vaginal Cream 2% | ||
|---|---|---|
| Event | Clindamycin Phosphate Vaginal Cream 2% | |
| 3 Day n=600 | 7 Day n=1325 | |
Urogenital | ||
Vaginal moniliasis | 7.7 | 10.4 |
Vulvovaginitis | 6.0 | 4.4 |
Vulvovaginal disorder | 3.2 | 5.3 |
Trichomonal vaginitis | 0 | 1.3 |
Body as a Whole | ||
Moniliasis (body) | 1.3 | 0.2 |
Other events occurring in <1% of the clindamycin vaginal cream 2% groups include:
Urogenital system : vaginal discharge, metrorrhagia, urinary tract infection, endometriosis, menstrual disorder, vaginitis/vaginal infection, and vaginal pain.
Body as a whole: localized abdominal pain, generalized abdominal pain, abdominal cramps, halitosis, headache, bacterial infection, inflammatory swelling, allergic reaction, and fungal infection.
Digestive system: nausea, vomiting, constipation, dyspepsia, flatulence, diarrhea, and gastrointestinal disorder.
Endocrine system : hyperthyroidism.
Central nervous system: dizziness and vertigo.
Respiratory system: epistaxis.
Skin: pruritus (non-application site), moniliasis, rash, maculopapular rash, erythema, and urticaria.
Special senses: taste perversion.
Pregnant Women
In a clinical trial involving pregnant women during the second trimester, 1.7% of 180 patients who received treatment for 7 days discontinued therapy due to drug-related adverse events. Medical events judged to be related, probably related, possibly related, or of unknown relationship to vaginally administered clindamycin phosphate vaginal cream 2%, were reported for 22.8% of pregnant patients. Events occurring in ≥1% of patients receiving either clindamycin phosphate vaginal cream 2% or placebo are shown in Table 2 .
| TABLE 2 - Events Occurring in ≥1% of Pregnant Patients Receiving Clindamycin Phosphate Vaginal Cream 2% or Placebo | ||
|---|---|---|
| Event | Clindamycin Phosphate Vaginal Cream 2% | Placebo |
| 7 DAY n=180 | 7 Day n=184 | |
Urogenital | ||
Vaginal moniliasis | 13.3 | 7.1 |
Vulvovaginal disorder | 6.7 | 7.1 |
Abnormal labor | 1.1 | 0.5 |
Body as a Whole | ||
Fungal infection | 1.7 | 0 |
Skin | ||
Pruritus, non-application site | 1.1 | 0 |
Other events occurring in <1% of the clindamycin vaginal cream 2% group include:
Urogenital system: dysuria, metrorrhagia, vaginal pain, and trichomonal vaginitis.
Body as a whole: upper respiratory infection.
Skin: pruritus (topical application site) and erythema.
Post-marketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In the post-marketing period, there have been case reports of pseudomembranous colitis with the use of clindamycin phosphate vaginal cream.
Other clindamycin formulations
Clindamycin vaginal cream affords minimal peak serum levels and systemic exposure (AUCs) of clindamycin compared to 100 mg oral clindamycin dosing. Although these lower levels of exposure are less likely to produce the common reactions seen with oral clindamycin, the possibility of these and other reactions cannot be excluded presently. Data from well-controlled trials directly comparing clindamycin administered orally to clindamycin administered vaginally are not available.
The following adverse reactions and altered laboratory tests have been reported with the oral or parenteral use of clindamycin:
Infections and Infestations: Clostridioides difficile coliti s
Gastrointestinal: Abdominal pain, esophagitis, nausea, vomiting, diarrhea and pseudomembranous colitis. (See WARNINGS. )
Hematopoietic: Transient neutropenia (leukopenia), eosinophilia, agranulocytosis, and thrombocytopenia have been reported. No direct etiologic relationship to concurrent clindamycin therapy could be made in any of these reports.
Hypersensitivity Reactions : Maculopapular rash and urticaria have been observed during drug therapy. Generalized mild to moderate morbilliform-like skin rashes are the most frequently reported of all adverse reactions. Cases of Acute Generalized Exanthematous Pustulosis (AGEP), erythema multiforme, some resembling Stevens-Johnson syndrome, have been associated with clindamycin. A few cases of anaphylactoid reactions have been reported. If a hypersensitivity reaction occurs, the drug should be discontinued.
Liver: Jaundice and abnormalities in liver function tests have been observed during clindamycin therapy.
Musculoskeletal : Cases of polyarthritis have been reported.
Renal: Acute kidney injury
Immune System : Drug reaction with eosinophilia and systemic symptoms (DRESS) cases have been reported.
Drug Interactions
Systemic clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, it should be used with caution in patients receiving such agents.
DESCRIPTION
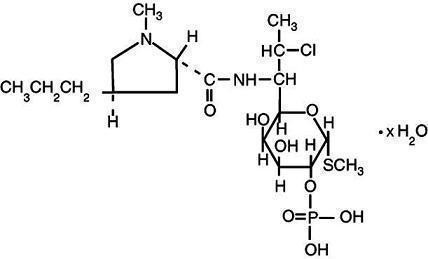
Clindamycin phosphate is a water soluble ester of the semi-synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent antibiotic lincomycin. The chemical name for clindamycin phosphate is methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl- trans -4-propyl-L-2-pyrrolidinecarboxamido)-1-thio-L- threo -α- D-galacto -octopyranoside 2-(dihydrogen phosphate). It has a molecular weight of 504.96, and the molecular formula is C 18 H 34 ClN 2 O 8 PS. The structural formula is represented below:
Clindamycin phosphate vaginal cream USP, 2%, is a semi-solid, white cream, which contains 2% clindamycin phosphate, USP, at a concentration equivalent to 20 mg clindamycin per gram. The pH of the cream is between 3.0 and 6.0. The cream also contains benzyl alcohol, cetostearyl alcohol, cetyl palmitate, mineral oil, polysorbate 60, propylene glycol, sorbitan monostearate, stearic acid and purified water.
Each applicatorful of 5 grams of vaginal cream contains approximately 100 mg of clindamycin phosphate.
CLINICAL PHARMACOLOGY
Mechanism of Action
Clindamycin is an antibacterial drug (See MICROBIOLOGY ).
Pharmacokinetics
Following a once a day intravaginal dose of 100 mg of clindamycin phosphate vaginal cream 2%, administered to 6 healthy female volunteers for 7 days, approximately 5% (range 0.6% to 11%) of the administered dose was absorbed systemically. The peak serum clindamycin concentration observed on the first day averaged 18 ng/mL (range 4 to 47 ng/mL) and on day 7 it averaged 25 ng/mL (range 6 to 61 ng/mL). These peak concentrations were attained approximately 10 hours post-dosing (range 4 to 24 hours).
Following a once a day intravaginal dose of 100 mg of clindamycin phosphate vaginal cream 2%, administered for 7 consecutive days to 5 women with bacterial vaginosis, absorption was slower and less variable than that observed in healthy females. Approximately 5% (range 2% to 8%) of the dose was absorbed systemically. The peak serum clindamycin concentration observed on the first day averaged 13 ng/mL (range 6 to 34 ng/mL) and on day 7 it averaged 16 ng/mL (range 7 to 26 ng/mL). These peak concentrations were attained approximately 14 hours post-dosing (range 4 to 24 hours).
There was little or no systemic accumulation of clindamycin after repeated vaginal dosing of clindamycin phosphate vaginal cream 2%. The systemic half-life was 1.5 to 2.6 hours.
CLINICAL STUDIES
In two clinical studies involving 674 evaluable non-pregnant women with bacterial vaginosis comparing clindamycin phosphate vaginal cream 2% for 3 or 7 days, the clinical cure rates, determined at 1 month posttherapy, ranged from 72% to 81% for the 3-day treatment and 84% to 86% for the 7-day treatment.
Clindamycin Phosphate Vaginal Cream 2% 3 Day | Clindamycin Phosphate Vaginal Cream 2% 7 Day | |||
US Study European Study | 94/131 161/199 | 72% 81% | 110/128 181/216 | 86% 84% |
In a clinical study involving 249 evaluable pregnant patients in the second and third trimester treated for 7 days, the clinical cure rate, determined at 1 month posttherapy, was 60% (77/129) in the clindamycin arm and 9% (11/120) for the vehicle arm. The determination of clinical cure was based on the absence of a "fishy" amine odor when the vaginal discharge was mixed with a 10% KOH solution and the absence of clue cells on microscopic examination.
Rx only
E. FOUGERA & CO. A division of Fougera PHARMACEUTICALS INC. Melville, New York 11747
46311926A
R06/2022
#116
HOW SUPPLIED
Clindamycin Phosphate Vaginal Cream USP, 2% is supplied as follows:
40 g tube (with 7 disposable applicators) NDC 0168-0277-40
Store at 20° to 25°C (68° to 77° F) [see USP Controlled Room Temperature]. Protect from freezing.