Get your patient on Clomipramine Hydrochloride - Clomipramine Hydrochloride capsule (Clomipramine Hydrochloride)
Clomipramine Hydrochloride - Clomipramine Hydrochloride capsule prescribing information
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of clomipramine hydrochloride or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Clomipramine hydrochloride is not approved for use in pediatric patients except for patients with obsessive compulsive disorder (OCD) ( see WARNINGS, Clinical Worsening and Suicide Risk; PRECAUTIONS, Information for Patients; and PRECAUTIONS, Pediatric Use).
INDICATIONS AND USAGE
Clomipramine hydrochloride capsules USP are indicated for the treatment of obsessions and compulsions in patients with Obsessive-Compulsive Disorder (OCD). The obsessions or compulsions must cause marked distress, be time-consuming, or significantly interfere with social or occupational functioning, in order to meet the DSM-III-R (circa 1989) diagnosis of OCD.
Obsessions are recurrent, persistent ideas, thoughts, images, or impulses that are ego-dystonic. Compulsions are repetitive, purposeful, and intentional behaviors performed in response to an obsession or in a stereotyped fashion, and are recognized by the person as excessive or unreasonable.
The effectiveness of clomipramine hydrochloride capsules for the treatment of OCD was demonstrated in multicenter, placebo-controlled, parallel-group studies, including two 10-week studies in adults and one 8-week study in children and adolescents 10 to 17 years of age. Patients in all studies had moderate-to-severe OCD (DSM-III), with mean baseline ratings on the Yale-Brown Obsessive Compulsive Scale (YBOCS) ranging from 26 to 28 and a mean baseline rating of 10 on the NIMH Clinical Global Obsessive Compulsive Scale (NIMH-OC). Patients taking CMI experienced a mean reduction of approximately 10 on the YBOCS, representing an average improvement on this scale of 35% to 42% among adults and 37% among children and adolescents. CMI-treated patients experienced a 3.5 unit decrement on the NIMH-OC. Patients on placebo showed no important clinical response on either scale. The maximum dose was 250 mg/day for most adults and 3 mg/kg/day (up to 200 mg) for all children and adolescents.
The effectiveness of clomipramine hydrochloride capsules for long-term use (i.e., for more than 10 weeks) has not been systematically evaluated in placebo-controlled trials. The physician who elects to use clomipramine hydrochloride capsules for extended periods should periodically reevaluate the long-term usefulness of the drug for the individual patient ( see DOSAGE AND ADMINISTRATION ).
DOSAGE AND ADMINISTRATION
The treatment regimens described below are based on those used in controlled clinical trials of clomipramine hydrochloride capsules in 520 adults, and 91 children and adolescents with OCD. During initial titration, clomipramine hydrochloride capsules should be given in divided doses with meals to reduce gastrointestinal side effects. The goal of this initial titration phase is to minimize side effects by permitting tolerance to side effects to develop or allowing the patient time to adapt if tolerance does not develop.
Because both CMI and its active metabolite, DMI, have long elimination half-lives, the prescriber should take into consideration the fact that steady-state plasma levels may not be achieved until 2 to 3 weeks after dosage change ( see CLINICAL PHARMACOLOGY ). Therefore, after initial titration, it may be appropriate to wait 2 to 3 weeks between further dosage adjustments.
Initial Treatment/Dose Adjustment (Adults)
Treatment with clomipramine hydrochloride capsules should be initiated at a dosage of 25 mg daily and gradually increased, as tolerated, to approximately 100 mg during the first 2 weeks. During initial titration, clomipramine hydrochloride capsules should be given in divided doses with meals to reduce gastrointestinal side effects. Thereafter, the dosage may be increased gradually over the next several weeks, up to a maximum of 250 mg daily. After titration, the total daily dose may be given once daily at bedtime to minimize daytime sedation.
Initial Treatment/Dose Adjustment (Children and Adolescents)
As with adults, the starting dose is 25 mg daily and should be gradually increased (also given in divided doses with meals to reduce gastrointestinal side effects) during the first 2 weeks, as tolerated, up to a daily maximum of 3 mg/kg or 100 mg, whichever is smaller. Thereafter, the dosage may be increased gradually over the next several weeks up to a daily maximum of 3 mg/kg or 200 mg, whichever is smaller ( see PRECAUTIONS, Pediatric Use ). As with adults, after titration, the total daily dose may be given once daily at bedtime to minimize daytime sedation.
Maintenance/Continuation Treatment (Adults, Children, and Adolescents)
While there are no systematic studies that answer the question of how long to continue clomipramine hydrochloride capsules, OCD is a chronic condition and it is reasonable to consider continuation for a responding patient. Although the efficacy of clomipramine hydrochloride capsules after 10 weeks has not been documented in controlled trials, patients have been continued in therapy under double-blind conditions for up to 1 year without loss of benefit. However, dosage adjustments should be made to maintain the patient on the lowest effective dosage, and patients should be periodically reassessed to determine the need for treatment. During maintenance, the total daily dose may be given once daily at bedtime.
Switching a Patient To or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders
At least 14 days should elapse between discontinuation of an MAOI intended to treat psychiatric disorders and initiation of therapy with clomipramine hydrochloride capsules. Conversely, at least 14 days should be allowed after stopping clomipramine hydrochloride capsules before starting an MAOI intended to treat psychiatric disorders ( see CONTRAINDICATIONS ).
Use of Clomipramine Hydrochloride Capsules With Other MAOIs, Such as Linezolid or Methylene Blue
Do not start clomipramine hydrochloride capsules in a patient who is being treated with linezolid or intravenous methylene blue because there is increased risk of serotonin syndrome. In a patient who requires more urgent treatment of a psychiatric condition, other interventions, including hospitalization, should be considered ( see CONTRAINDICATIONS ).
In some cases, a patient already receiving clomipramine hydrochloride capsules therapy may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of serotonin syndrome in a particular patient, clomipramine hydrochloride capsules should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for symptoms of serotonin syndrome for two weeks or until 24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with clomipramine hydrochloride capsules may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue ( see WARNINGS ).
The risk of administering methylene blue by non-intravenous routes (such as oral tablets or by local injection) or in intravenous doses much lower than 1 mg/kg with clomipramine hydrochloride capsules is unclear. The clinician should, nevertheless, be aware of the possibility of emergent symptoms of serotonin syndrome with such use ( see WARNINGS ).
CONTRAINDICATIONS
Clomipramine hydrochloride capsules are contraindicated in patients with a history of hypersensitivity to clomipramine hydrochloride capsules or other tricyclic antidepressants.
Monoamine Oxidase Inhibitors (MAOIs)
The use of MAOIs intended to treat psychiatric disorders with clomipramine hydrochloride capsules or within 14 days of stopping treatment with clomipramine hydrochloride capsules is contraindicated because of an increased risk of serotonin syndrome. The use of clomipramine hydrochloride capsules within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated ( see WARNINGS and DOSAGE AND ADMINISTRATION ).
Starting clomipramine hydrochloride capsules in a patient who is being treated with linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome ( see WARNINGS and DOSAGE AND ADMINISTRATION ).
Myocardial Infarction
Clomipramine hydrochloride capsules are contraindicated during the acute recovery period after a myocardial infarction.
ADVERSE REACTIONS
Commonly Observed
The most commonly observed adverse events associated with the use of clomipramine hydrochloride capsules and not seen at an equivalent incidence among placebo-treated patients were gastrointestinal complaints, including dry mouth, constipation, nausea, dyspepsia, and anorexia; nervous system complaints, including somnolence, tremor, dizziness, nervousness, and myoclonus; genitourinary complaints, including changed libido, ejaculatory failure, impotence, and micturition disorder; and other miscellaneous complaints, including fatigue, sweating, increased appetite, weight gain, and visual changes.
Leading to Discontinuation of Treatment
Approximately 20% of 3,616 patients who received clomipramine hydrochloride capsules in U.S. premarketing clinical trials discontinued treatment because of an adverse event. Approximately one-half of the patients who discontinued (9% of the total) had multiple complaints, none of which could be classified as primary. Where a primary reason for discontinuation could be identified, most patients discontinued because of nervous system complaints (5.4%), primarily somnolence. The second-most-frequent reason for discontinuation was digestive system complaints (1.3%), primarily vomiting and nausea.
There was no apparent relationship between the adverse events and elevated plasma drug concentrations.
Incidence in Controlled Clinical Trials
The following table enumerates adverse events that occurred at an incidence of 1% or greater among patients with OCD who received clomipramine hydrochloride capsules in adult or pediatric placebo-controlled clinical trials. The frequencies were obtained from pooled data of clinical trials involving either adults receiving clomipramine hydrochloride capsules (N=322) or placebo (N=319) or children treated with clomipramine hydrochloride capsules (N=46) or placebo (N=44). The prescriber should be aware that these figures cannot be used to predict the incidence of side effects in the course of usual medical practice, in which patient characteristics and other factors differ from those that prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators. The cited figures, however, provide the physician with a basis for estimating the relative contribution of drug and nondrug factors to the incidence of side effects in the populations studied.
| •Events reported by at least 1% of clomipramine hydrochloride capsules patients are included. | |||||
| Adults | Children and Adolescents | ||||
| Body System/ Adverse Event• | Clomipramine Hydrochloride Capsules (N=322) | Placebo (N=319) | Clomipramine Hydrochloride Capsules (N=46) | Placebo (N=44) | |
| Nervous System | |||||
| Somnolence | 54 | 16 | 46 | 11 | |
| Tremor | 54 | 2 | 33 | 2 | |
| Dizziness | 54 | 14 | 41 | 14 | |
| Headache | 52 | 41 | 28 | 34 | |
| Insomnia | 25 | 15 | 11 | 7 | |
| Libido change | 21 | 3 | - | - | |
| Nervousness | 18 | 2 | 4 | 2 | |
| Myoclonus | 13 | - | 2 | - | |
| Increased appetite | 11 | 2 | - | 2 | |
| Paresthesia | 9 | 3 | 2 | 2 | |
| Memory impairment | 9 | 1 | 7 | 2 | |
| Anxiety | 9 | 4 | 2 | - | |
| Twitching | 7 | 1 | 4 | 5 | |
| Impaired concentration | 5 | 2 | - | - | |
| Depression | 5 | 1 | - | - | |
| Hypertonia | 4 | 1 | 2 | - | |
| Sleep disorder | 4 | - | 9 | 5 | |
| Psychosomatic disorder | 3 | - | - | - | |
| Yawning | 3 | - | - | - | |
| Confusion | 3 | - | 2 | - | |
| Speech disorder | 3 | - | - | - | |
| Abnormal dreaming | 3 | - | - | 2 | |
| Agitation | 3 | - | - | - | |
| Migraine | 3 | - | - | - | |
| Depersonalization | 2 | - | 2 | - | |
| Irritability | 2 | 2 | 2 | - | |
| Emotional lability | 2 | - | - | 2 | |
| Panic reaction | 1 | - | 2 | - | |
| Aggressive reaction | - | - | 2 | - | |
| Paresis | - | - | 2 | - | |
| Skin and Appendages | |||||
| Increased sweating | 29 | 3 | 9 | - | |
| Rash | 8 | 1 | 4 | 2 | |
| Pruritus | 6 | - | 2 | 2 | |
| Dermatitis | 2 | - | - | 2 | |
| Acne | 2 | 2 | - | 5 | |
| Dry skin | 2 | - | - | 5 | |
| Urticaria | 1 | - | - | - | |
| Abnormal skin odor | - | - | 2 | - | |
| Digestive System | |||||
| Dry mouth | 84 | 17 | 63 | 16 | |
| Constipation | 47 | 11 | 22 | 9 | |
| Nausea | 33 | 14 | 9 | 11 | |
| Dyspepsia | 22 | 10 | 13 | 2 | |
| Diarrhea | 13 | 9 | 7 | 5 | |
| Anorexia | 12 | - | 22 | 2 | |
| Abdominal pain | 11 | 9 | 13 | 16 | |
| Vomiting | 7 | 2 | 7 | - | |
| Flatulence | 6 | 3 | - | 2 | |
| Tooth disorder | 5 | - | - | - | |
| Gastrointestinal disorder | 2 | - | - | 2 | |
| Dysphagia | 2 | - | - | - | |
| Esophagitis | 1 | - | - | - | |
| Eructation | - | - | 2 | 2 | |
| Ulcerative stomatitis | - | - | 2 | - | |
| Body as a Whole | |||||
| Fatigue | 39 | 18 | 35 | 9 | |
| Weight increase | 18 | 1 | 2 | - | |
| Flushing | 8 | - | 7 | - | |
| Hot flushes | 5 | - | 2 | - | |
| Chest pain | 4 | 4 | 7 | - | |
| Fever | 4 | - | 2 | 7 | |
| Allergy | 3 | 3 | 7 | 5 | |
| Pain | 3 | 2 | 4 | 2 | |
| Local edema | 2 | 4 | - | - | |
| Chills | 2 | 1 | - | - | |
| Weight decrease | - | - | 7 | - | |
| Otitis media | - | - | 4 | 5 | |
| Asthenia | - | - | 2 | - | |
| Halitosis | - | - | 2 | - | |
| Cardiovascular System | |||||
| Postural hypotension | 6 | - | 4 | - | |
| Palpitation | 4 | 2 | 4 | - | |
| Tachycardia | 4 | - | 2 | - | |
| Syncope | - | - | 2 | - | |
| Respiratory System | |||||
| Pharyngitis | 14 | 9 | - | 5 | |
| Rhinitis | 12 | 10 | 7 | 9 | |
| Sinusitis | 6 | 4 | 2 | 5 | |
| Coughing | 6 | 6 | 4 | 5 | |
| Bronchospasm | 2 | - | 7 | 2 | |
| Epistaxis | 2 | - | - | 2 | |
| Dyspnea | - | - | 2 | - | |
| Laryngitis | - | 1 | 2 | - | |
| Urogenital System | |||||
| Male and Female Patients Combined | |||||
| Micturition disorder | 14 | 2 | 4 | 2 | |
| Urinary tract infection | 6 | 1 | - | - | |
| Micturition frequency | 5 | 3 | - | - | |
| Urinary retention | 2 | - | 7 | - | |
| Dysuria | 2 | 2 | - | - | |
| Cystitis | 2 | - | - | - | |
| Female Patients Only | (N=182) | (N=167) | (N=10) | (N=21) | |
| Dysmenorrhea | 12 | 14 | 10 | 10 | |
| Lactation (nonpuerperal) | 4 | - | - | - | |
| Menstrual disorder | 4 | 2 | - | - | |
| Vaginitis | 2 | - | - | - | |
| Leukorrhea | 2 | - | - | - | |
| Breast enlargement | 2 | - | - | - | |
| Breast pain | 1 | - | - | - | |
| Amenorrhea | 1 | - | - | - | |
| Male Patients Only | (N=140) | (N=152) | (N=36) | (N=23) | |
| Ejaculation failure | 42 | 2 | 6 | - | |
| Impotence | 20 | 3 | - | - | |
| Special Senses | |||||
| Abnormal vision | 18 | 4 | 7 | 2 | |
| Taste perversion | 8 | - | 4 | - | |
| Tinnitus | 6 | - | 4 | - | |
| Abnormal lacrimation | 3 | 2 | - | - | |
| Mydriasis | 2 | - | - | - | |
| Conjunctivitis | 1 | - | - | - | |
| Anisocoria | - | - | 2 | - | |
| Blepharospasm | - | - | 2 | - | |
| Ocular allergy | - | - | 2 | - | |
| Vestibular disorder | - | - | 2 | 2 | |
| Musculoskeletal | |||||
| Myalgia | 13 | 9 | - | - | |
| Back pain | 6 | 6 | - | - | |
| Arthralgia | 3 | 5 | - | - | |
| Muscle weakness | 1 | - | 2 | - | |
| Hemic and Lymphatic | |||||
| Purpura | 3 | - | - | - | |
| Anemia | - | - | 2 | 2 | |
| Metabolic and Nutritional | |||||
| Thirst | 2 | 2 | - | 2 | |
Other Events Observed During the Premarketing Evaluation of Clomipramine Hydrochloride Capsules
During clinical testing in the U.S., multiple doses of clomipramine hydrochloride capsules were administered to approximately 3,600 subjects. Untoward events associated with this exposure were recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse events without first grouping similar types of untoward events into a smaller number of standardized event categories.
In the tabulations that follow, a modified World Health Organization dictionary of terminology has been used to classify reported adverse events. The frequencies presented, therefore, represent the proportion of the 3,525 individuals exposed to clomipramine hydrochloride capsules who experienced an event of the type cited on at least one occasion while receiving clomipramine hydrochloride capsules. All events are included except those already listed in the previous table, those reported in terms so general as to be uninformative, and those in which an association with the drug was remote. It is important to emphasize that although the events reported occurred during treatment with clomipramine hydrochloride capsules, they were not necessarily caused by it.
Events are further categorized by body system and listed in order of decreasing frequency according to the following definitions: frequent adverse events are those occurring on one or more occasions in at least 1/100 patients; infrequent adverse events are those occurring in 1/100 to 1/1,000 patients; rare events are those occurring in less than 1/1,000 patients.
Body as a Whole – Infrequent - general edema, increased susceptibility to infection, malaise. Rare - dependent edema, withdrawal syndrome.
Cardiovascular System – Infrequent - abnormal ECG, arrhythmia, bradycardia, cardiac arrest, extrasystoles, pallor. Rare - aneurysm, atrial flutter, bundle branch block, cardiac failure, cerebral hemorrhage, heart block, myocardial infarction, myocardial ischemia, peripheral ischemia, thrombophlebitis, vasospasm, ventricular tachycardia.
Digestive System – Infrequent - abnormal hepatic function, blood in stool, colitis, duodenitis, gastric ulcer, gastritis, gastroesophageal reflux, gingivitis, glossitis, hemorrhoids, hepatitis, increased saliva, irritable bowel syndrome, peptic ulcer, rectal hemorrhage, tongue ulceration, tooth caries. Rare - cheilitis, chronic enteritis, discolored feces, gastric dilatation, gingival bleeding, hiccup, intestinal obstruction, oral/pharyngeal edema, paralytic ileus, salivary gland enlargement.
Endocrine System – Infrequent - hypothyroidism. Rare - goiter, gynecomastia, hyperthyroidism.
Hemic and Lymphatic System – Infrequent - lymphadenopathy. Rare - leukemoid reaction, lymphoma-like disorder, marrow depression.
Metabolic and Nutritional Disorder – Infrequent - dehydration, diabetes mellitus, gout, hypercholesterolemia, hyperglycemia, hyperuricemia, hypokalemia. Rare - fat intolerance, glycosuria.
Musculoskeletal System – Infrequent - arthrosis. Rare - dystonia, exostosis, lupus erythematosus rash, bruising, myopathy, myositis, polyarteritis nodosa, torticollis.
Nervous System – Frequent - abnormal thinking, vertigo. Infrequent - abnormal coordination, abnormal EEG, abnormal gait, apathy, ataxia, coma, convulsions, delirium, delusion, dyskinesia, dysphonia, encephalopathy, euphoria, extrapyramidal disorder, hallucinations, hostility, hyperkinesia, hypnagogic hallucinations, hypokinesia, leg cramps, manic reaction, neuralgia, paranoia, phobic disorder, psychosis, sensory disturbance, somnambulism, stimulation, suicidal ideation, suicide attempt, teeth-grinding. Rare - anticholinergic syndrome, aphasia, apraxia, catalepsy, cholinergic syndrome, choreoathetosis, generalized spasm, hemiparesis, hyperesthesia, hyperreflexia, hypoesthesia, illusion, impaired impulse control, indecisiveness, mutism, neuropathy, nystagmus, oculogyric crisis, oculomotor nerve paralysis, schizophrenic reaction, stupor, suicide.
Respiratory System – Infrequent - bronchitis, hyperventilation, increased sputum, pneumonia. Rare - cyanosis, hemoptysis, hypoventilation, laryngismus.
Skin and Appendages – Infrequent - alopecia, cellulitis, cyst, eczema, erythematous rash, genital pruritus, maculopapular rash, photosensitivity reaction, psoriasis, pustular rash, skin discoloration. Rare - chloasma, folliculitis, hypertrichosis, piloerection, seborrhea, skin hypertrophy, skin ulceration.
Special Senses – Infrequent - abnormal accommodation, deafness, diplopia, earache, eye pain, foreign body sensation, hyperacusis, parosmia, photophobia, scleritis, taste loss. Rare - blepharitis, chromatopsia, conjunctival hemorrhage, exophthalmos, glaucoma, keratitis, labyrinth disorder, night blindness, retinal disorder, strabismus, visual field defect.
Urogenital System – Infrequent - endometriosis, epididymitis, hematuria, nocturia, oliguria, ovarian cyst, perineal pain, polyuria, prostatic disorder, renal calculus, renal pain, urethral disorder, urinary incontinence, uterine hemorrhage, vaginal hemorrhage. Rare - albuminuria, anorgasmy, breast engorgement, breast fibroadenosis, cervical dysplasia, endometrial hyperplasia, premature ejaculation, pyelonephritis, pyuria, renal cyst, uterine inflammation, vulvar disorder.
Postmarketing Experience
The following adverse drug reaction has been reported during post-approval use of clomipramine hydrochloride capsules. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate frequency.
Eye Disorders – A ngle-closure glaucoma.
Immune System Disorders – Drug Rash with Eosinophilia and Systemic Symptoms (DRESS)
Metabolism and Nutrition Disorder - Hyponatremia
Endocrine Disorders - Syndrome of inappropriate antidiuretic hormone section (SIADH)
Drug Interactions
The risks of using clomipramine hydrochloride capsules in combination with other drugs have not been systematically evaluated. Given the primary CNS effects of clomipramine hydrochloride capsules, caution is advised in using them concomitantly with other CNS-active drugs ( see Information for Patients ). Clomipramine hydrochloride capsules should not be used with MAO inhibitors ( see CONTRAINDICATIONS ).
Close supervision and careful adjustment of dosage are required when clomipramine hydrochloride capsules are administered with anticholinergic or sympathomimetic drugs.
Several tricyclic antidepressants have been reported to block the pharmacologic effects of guanethidine, clonidine, or similar agents, and such an effect may be anticipated with CMI because of its structural similarity to other tricyclic antidepressants.
The plasma concentration of CMI has been reported to be increased by the concomitant administration of haloperidol; plasma levels of several closely related tricyclic antidepressants have been reported to be increased by the concomitant administration of methylphenidate or hepatic enzyme inhibitors (e.g., cimetidine, fluoxetine) and decreased by the concomitant administration of hepatic enzyme inducers (e.g., barbiturates, phenytoin), and such an effect may be anticipated with CMI as well. Administration of CMI has been reported to increase the plasma levels of phenobarbital, if given concomitantly ( see CLINICAL PHARMACOLOGY, Interactions ).
Drugs Metabolized by P450 2D6 – The biochemical activity of the drug metabolizing isozyme cytochrome P450 2D6 (debrisoquin hydroxylase) is reduced in a subset of the Caucasian population (about 7% to 10% of Caucasians are so-called “poor metabolizers”); reliable estimates of the prevalence of reduced P450 2D6 isozyme activity among Asian, African and other populations are not yet available. Poor metabolizers have higher than expected plasma concentrations of tricyclic antidepressants (TCAs) when given usual doses. Depending on the fraction of drug metabolized by P450 2D6, the increase in plasma concentration may be small, or quite large (8 fold increase in plasma AUC of the TCA). In addition, certain drugs inhibit the activity of this isozyme and make normal metabolizers resemble poor metabolizers. An individual who is stable on a given dose of TCA may become abruptly toxic when given one of these inhibiting drugs as concomitant therapy. The drugs that inhibit cytochrome P450 2D6 include some that are not metabolized by the enzyme (quinidine; cimetidine) and many that are substrates for P450 2D6 (many other antidepressants, phenothiazines, and the Type 1C antiarrhythmics propafenone and flecainide). While all the selective serotonin reuptake inhibitors (SSRIs), e.g., fluoxetine, sertraline, paroxetine, and fluvoxamine, inhibit P450 2D6, they may vary in the extent of inhibition. Fluvoxamine has also been shown to inhibit P450 1A2, an isoform also involved in TCA metabolism. The extent to which SSRI-TCA interactions may pose clinical problems will depend on the degree of inhibition and the pharmacokinetics of the SSRI involved. Nevertheless, caution is indicated in the co-administration of TCAs with any of the SSRIs and also in switching from one class to the other. Of particular importance, sufficient time must elapse before initiating TCA treatment in a patient being withdrawn from fluoxetine, given the long half-life of the parent and active metabolite (at least 5 weeks may be necessary). Concomitant use of agents in the tricyclic antidepressant class (which includes clomipramine hydrochloride capsules) with drugs that can inhibit cytochrome P450 2D6 may require lower doses than usually prescribed for either the tricyclic antidepressant agent or the other drug. Furthermore, whenever one of these drugs is withdrawn from co-therapy, an increased dose of tricyclic antidepressant agent may be required. It is desirable to monitor TCA plasma levels whenever an agent of the tricyclic antidepressant class including clomipramine hydrochloride capsules is going to be co-administered with another drug known to be an inhibitor of P450 2D6 (and/or P450 1A2).
Because clomipramine hydrochloride capsules are highly bound to serum protein, the administration of clomipramine hydrochloride capsules to patients taking other drugs that are highly bound to protein (e.g., warfarin, digoxin) may cause an increase in plasma concentrations of these drugs, potentially resulting in adverse effects. Conversely, adverse effects may result from displacement of protein-bound clomipramine hydrochloride capsules by other highly bound drugs ( see CLINICAL PHARMACOLOGY, Distribution ).
DESCRIPTION
Clomipramine hydrochloride capsules USP is an antiobsessional drug that belongs to the class (dibenzazepine) of pharmacologic agents known as tricyclic antidepressants. Clomipramine hydrochloride capsules are available as capsules of 25, 50, and 75 mg for oral administration.
Clomipramine hydrochloride USP is 3-chloro-5-[3-(dimethylamino)propyl]-10,11-dihydro- 5H -dibenz[ b,f ] azepine monohydrochloride, and its structural formula is:
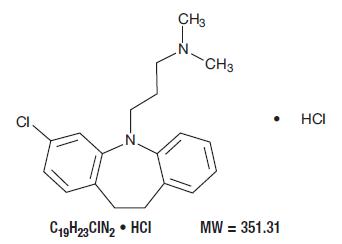
Clomipramine hydrochloride USP is a white to off-white crystalline powder. It is freely soluble in water, in methanol, and in methylene chloride, and insoluble in ethyl ether and in hexane.
Inactive Ingredients . colloidal silicon dioxide, D&C Yellow No. 10, FD&C Blue No. 1 (25 mg and 50 mg strengths only), FD&C Red No. 3 (25 mg and 50 mg strengths only), FD&C Red No. 40 (50 mg strength only), FD&C Yellow No. 6 (25 mg and 75 mg strength only), gelatin , magnesium stearate, pregelatinized starch, and titanium dioxide.
In addition, the black imprinting ink for the 25 mg, 50 mg and 75 mg capsules contains black iron oxide, propylene glycol, shellac, strong ammonia solution.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Clomipramine (CMI) is presumed to influence obsessive and compulsive behaviors through its effects on serotonergic neuronal transmission. The actual neurochemical mechanism is unknown, but CMI's capacity to inhibit the reuptake of serotonin (5-HT) is thought to be important.
Pharmacokinetics
Absorption/Bioavailability – CMI from clomipramine hydrochloride capsules is as bioavailable as CMI from a solution. The bioavailability of CMI from capsules is not significantly affected by food.
In a dose proportionality study involving multiple CMI doses, steady-state plasma concentrations (C ss ) and area-under-plasma-concentration-time curves (AUC) of CMI and CMI's major active metabolite, desmethylclomipramine (DMI), were not proportional to dose over the ranges evaluated, i.e., between 25 to 100 mg/day and between 25 to 150 mg/day, although C ss and AUC are approximately linearly related to dose between 100 to 150 mg/day. The relationship between dose and CMI/DMI concentrations at higher daily doses has not been systematically assessed, but if there is significant dose dependency at doses above 150 mg/day, there is the potential for dramatically higher C ss and AUC even for patients dosed within the recommended range. This may pose a potential risk to some patients ( see WARNINGS and PRECAUTIONS, Drug Interactions ).
After a single 50 mg oral dose, maximum plasma concentrations of CMI occur within 2 to 6 hours (mean, 4.7 hr) and range from 56 ng/mL to 154 ng/mL (mean, 92 ng/mL). After multiple daily doses of 150 mg of clomipramine hydrochloride capsules, steady-state maximum plasma concentrations range from 94 ng/mL to 339 ng/mL (mean, 218 ng/mL) for CMI and from 134 ng/mL to 532 ng/mL (mean, 274 ng/mL) for DMI. Additional information from a rising dose study of doses up to 250 mg suggests that DMI may exhibit nonlinear pharmacokinetics over the usual dosing range. At a dose of clomipramine hydrochloride capsules 200 mg, subjects who had a single blood sample taken approximately 9 to 22 hours, (median 16 hours), after the dose had plasma concentrations of up to 605 ng/mL for CMI, 781 ng/mL for DMI, and 1,386 ng/mL for both.
Distribution – CMI distributes into cerebrospinal fluid (CSF) and brain and into breast milk. DMI also distributes into CSF, with a mean CSF/plasma ratio of 2.6. The protein binding of CMI is approximately 97%, principally to albumin, and is independent of CMI concentration. The interaction between CMI and other highly protein-bound drugs has not been fully evaluated, but may be important ( see PRECAUTIONS, Drug Interactions ).
Metabolism – CMI is extensively biotransformed to DMI and other metabolites and their glucuronide conjugates. DMI is pharmacologically active, but its effects on OCD behaviors are unknown. These metabolites are excreted in urine and feces, following biliary elimination. After a 25 mg radiolabeled dose of CMI in two subjects, 60% and 51%, respectively, of the dose were recovered in the urine and 32% and 24%, respectively, in feces. In the same study, the combined urinary recoveries of CMI and DMI were only about 0.8% to 1.3% of the dose administered. CMI does not induce drug-metabolizing enzymes, as measured by antipyrine half-life.
Elimination – Evidence that the C ss and AUC for CMI and DMI may increase disproportionately with increasing oral doses suggests that the metabolism of CMI and DMI may be capacity limited. This fact must be considered in assessing the estimates of the pharmacokinetic parameters presented below, as these were obtained in individuals exposed to doses of 150 mg. If the pharmacokinetics of CMI and DMI are nonlinear at doses above 150 mg, their elimination half-lives may be considerably lengthened at doses near the upper end of the recommended dosing range (i.e., 200 mg/day to 250 mg/day). Consequently, CMI and DMI may accumulate, and this accumulation may increase the incidence of any dose- or plasma-concentration-dependent adverse reactions, in particular seizures ( see WARNINGS ).
After a 150 mg dose, the half-life of CMI ranges from 19 hours to 37 hours (mean, 32 hr) and that of DMI ranges from 54 hours to 77 hours (mean, 69 hr). Steady-state levels after multiple dosing are typically reached within 7 to 14 days for CMI. Plasma concentrations of the metabolite exceed the parent drug on multiple dosing. After multiple dosing with 150 mg/day, the accumulation factor for CMI is approximately 2.5 and for DMI is 4.6. Importantly, it may take two weeks or longer to achieve this extent of accumulation at constant dosing because of the relatively long elimination half-lives of CMI and DMI ( see DOSAGE AND ADMINISTRATION ). The effects of hepatic and renal impairment on the disposition of clomipramine hydrochloride capsules have not been determined.
Interactions – Co-administration of haloperidol with CMI increases plasma concentrations of CMI. Co-administration of CMI with phenobarbital increases plasma concentrations of phenobarbital ( see PRECAUTIONS, Drug Interactions ). Younger subjects (18 to 40 years of age) tolerated CMI better and had significantly lower steady-state plasma concentrations, compared with subjects over 65 years of age. Children under 15 years of age had significantly lower plasma concentration/dose ratios, compared with adults. Plasma concentrations of CMI were significantly lower in smokers than in nonsmokers.
HOW SUPPLIED
Clomipramine Hydrochloride Capsules USP
Capsules 25 mg – the body is opaque ivory with "T" and the cap is opaque orange with "280" imprinted on it
Bottles of 100…………………………………...NDC 52817-280-10
Capsules 50 mg – the body is opaque ivory with "T" and the cap is opaque light blue with "281" imprinted on it
Bottles of 100…………………………………..NDC 52817-281-10
Capsules 75 mg – the body is opaque ivory with "T" and the cap is opaque yellow with "282" imprinted on it
Bottles of 100…………………………………..NDC 52817-282-10
Clomipramine Hydrochloride - Clomipramine Hydrochloride capsule PubMed™ news
- Journal Article • 2026 MayEmpagliflozin mitigates depression-like behavior in diabetic mice via putative CRF(1) receptor modulation: behavioral, biochemical and in silico studies.
- Journal Article • 2026 AprUltrasound model of depression in rodents: A comprehensive analysis of validity, pathogenic mechanisms, and potential for translation to humans.
- Journal Article • 2026 AprPalliative psychiatry and euthanasia in refractory obsessive-compulsive disorder: A case report.
- Journal Article • 2026 AprParenteral clomipramine for depression or obsessive-compulsive disorder: a systematic review and meta-analysis.
- Journal Article • 2026 AprMulti-criteria risk assessment for prioritising contaminants of emerging concern in treated wastewater and surface waters.