Get your patient on Clotic (clotrimazole) Otic Solution (Clotrimazole)
Clotic (Clotrimazole) Otic Solution prescribing information
INDICATIONS AND USAGE
CLOTIC is indicated for the treatment of fungal otitis externa (otomycosis) due to Aspergillus species and Candida species in patients 18 years of age and older.
Limitations of Use
CLOTIC has only been studied in patients with intact tympanic membranes. The use of CLOTIC is not recommended for the treatment of otomycosis in patients with perforated tympanic membranes.
DOSAGE AND ADMINISTRATION
Recommended Dosage and Administration
CLOTIC is for otic use only.
Instill the contents of one single-dose CLOTIC vial into the affected ear canal twice daily morning and evening, preferably 12 hours apart, for 14 consecutive days.
Preparation and Administration Instructions
- Warm the solution by holding the vial in the hand for 1 to 2 minutes. This is to avoid dizziness which may result from the instillation of a cold solution into the ear canal.
- The patient should lie with the affected ear upward, and then instill the medication.
- Gently pull the outer ear lobe upward and outward. This will allow the ear drops to flow down into the ear canal.
- Maintain this position for 1 minute after the drug administration.
- Repeat, if necessary, for the opposite ear [see Instructions for Use ].
DOSAGE FORMS AND STRENGTHS
Otic solution: Each single-dose vial of CLOTIC (clotrimazole) otic solution, 1%, delivers approximately 0.17 mL of solution equivalent to 1.7 mg of clotrimazole. The solution is clear colorless to slightly yellow.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Systemic absorption of clotrimazole following topical administration appears to be minimal, and maternal use of CLOTIC is not expected to result in fetal exposure to the drug.
The background risk of major birth defects and miscarriage for the indicated population is unknown All pregnancies have a background risk of major birth defects, loss, and other adverse outcomes. The background risk in the U.S. general population of major birth defects is 2% to 4% and of miscarriage is 15% to 20% of clinically recognized pregnancies.
Lactation
Risk Summary
Systemic absorption following topical administration of CLOTIC appears to be minimal, and breastfeeding is not expected to result in exposure of the child to CLOTIC. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for CLOTIC and any potential adverse effects on the breast-fed infant from CLOTIC or from the underlying maternal condition.
Pediatric Use
The safety and effectiveness of CLOTIC in pediatric patients less than 18 years of age have not been established.
Geriatric Use
Of the total number of patients with otomycosis who received CLOTIC in Trial 1 and Trial 2 (n=261), 78 (30%) were 65 years of age and older, while 23 (9%) were 75 years of age and older. No overall differences in safety or effectiveness were observed between subjects 65 years of age and older and younger adult patients, and other reported clinical experience has not identified differences in responses between the elderly and younger adult patients, but greater sensitivity of some older individuals cannot be ruled out.
CONTRAINDICATIONS
CLOTIC is contraindicated in patients with known hypersensitivity to clotrimazole.
WARNINGS AND PRECAUTIONS
None.
ADVERSE REACTIONS
The most common adverse reactions that occurred in ≥ 1 patient were headache, application site pain, tinnitus, tympanic membrane perforation, and paresthesia. (6 )
To report SUSPECTED ADVERSE REACTIONS, contact Carwin Pharmaceutical Associates, LLC at phone 1-844-700-5011 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In two randomized, placebo-controlled, phase 3 trials, 393 adult patients with otomycosis and intact tympanic membranes were treated with CLOTIC or placebo (Trial 1 and Trial 2) twice daily for a median duration of 14 days. There were 261 patients who received at least one dose of CLOTIC and 132 patients received at least one dose of placebo. These patients had a mean age of 51 years (range 19 to 89 years); 55% were male, 87% were White, and 26% were Hispanic or Latino ethnicity.
Adverse Reactions Leading to Discontinuation
Treatment discontinuation due to an adverse reaction occurred in 1% (3/261) of patients receiving CLOTIC and 3% (4/132) of patients receiving placebo. Adverse reactions leading to discontinuation of CLOTIC were application site pain in two patients and tympanic membrane perforation in one patient.
Most Common Adverse Reactions
Selected adverse reactions that occurred in 1 or more patients receiving CLOTIC are presented in Table 1.
| Adverse Reactions | CLOTIC N=261 n (%) | Placebo N=132 n (%) |
|---|---|---|
| Headache | 4 (1.5) | 2 (1.5) |
| Application site pain | 2 (0.8) | 0 |
| Tinnitus | 2 (0.8) | 0 |
| Tympanic membrane perforation | 2 (0.8) | 1 (0.8) |
| Paresthesia | 1 (0.4) | 0 |
DESCRIPTION
CLOTIC is a sterile preservative-free otic solution containing clotrimazole, an azole antifungal agent, at a concentration of 1% in polyethylene glycol (PEG) 400. The solution is clear colorless to slightly yellow. Each single-dose vial of CLOTIC delivers approximately 0.17 mL of solution equivalent to 1.7 mg of clotrimazole.
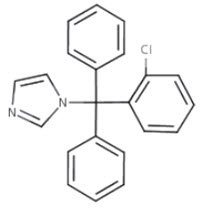
Chemically, clotrimazole is 1-(o-chloro-α,α-diphenylbenzyl)imidazole, with the empirical formula C 22 H 17 ClN 2 , a molecular weight of 344.84, and the following structural formula.
CLINICAL PHARMACOLOGY
Mechanism of Action
Clotrimazole is an azole antifungal agent [see Microbiology (12.4) ] .
Pharmacodynamics
Clotrimazole exposure-response relationships and the time course of pharmacodynamic response are unknown.
Pharmacokinetics
Systemic absorption following topical administration of CLOTIC is anticipated to be minimal. The plasma concentrations of clotrimazole were not measured following otic installation of 1.7 mg (0.17 mL) clotrimazole 1% solution twice daily.
Microbiology
Mechanism of Action
Clotrimazole, an azole antifungal agent, inhibits the cytochrome P450 dependent enzyme lanosterol 14-α-demethylase encoded by Erg11 in yeasts and Cyp51A in molds. Erg11/Cyp51A is necessary for the conversion of lanosterol to ergosterol, which is an essential sterol component of fungal cytoplasmic membrane. Disruption in ergosterol biosynthesis causes damage to the cell membrane by increasing its permeability, resulting in cell lysis and death. Clotrimazole inhibition of lanosterol 14-α-demethylase also leads to an accumulation of 14-α-methylsterols; the methylsterols may affect the electron transport system, thereby inhibiting growth of fungi.
Resistance
Resistance to azoles, including clotrimazole, has been associated with decreased affinity for the target enzyme (Erg11 in Candida spp. and Cyp51A in Aspergillus spp.), increase in target copy number, alteration of the ergosterol biosynthetic pathway and overexpression of efflux pumps.
Antimicrobial Activity
CLOTIC has been shown to be active against most isolates of the following microorganisms both in vitro and in clinical infections [see Indications and Usage (1) ] :
Fungal
Aspergillus spp. Candida spp.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No long-term studies of CLOTIC have been performed to evaluate carcinogenic potential. A 78-week oral (dietary) study of clotrimazole in rats did not show evidence of carcinogenicity at doses up to an equivalent of 150 mg/kg/day.
Mutagenesis
Clotrimazole has been studied in in vitro (bacterial) and in vivo (hamster spermatogonia) mutagenicity assays, and no evidence of mutagenic potential was found.
Impairment of Fertility
No reproduction toxicity studies were conducted with CLOTIC. Absorption of clotrimazole following topical administration is minimal.
CLINICAL STUDIES
Osteoarthritis
The effectiveness and safety of CLOTIC were evaluated in two phase 3, multicenter, randomized, double-blind, placebo-controlled trials in adult patients with unilateral or bilateral otomycosis and intact tympanic membranes (Trial 1, NCT03686384; Trial 2, NCT03686397). The treatment regimen consisted of instilling the contents of one single-dose vial of CLOTIC or placebo in the affected ear canal(s) twice daily for 14 consecutive days. Trial 1 was performed in the U.S. and Mexico; Trial 2 was performed in Europe.
Out of 394 patients randomized in Trial 1 and Trial 2, 228 had a positive baseline fungal culture for Aspergillus species and/or Candida species (mycological intent-to-treat [MITT] population). These patients had a median age of 53 years (range 19 to 89 years); 54% were male, 86% were White, and 28% were Hispanic or Latino ethnicity.
Therapeutic cure was defined as both clinical cure and mycological cure at the test of cure visit which occurred on Day 24 to 26. Clinical cure was defined as a total signs/symptoms score of zero on a 4 point scale for pruritis, otalgia, ear fullness, and otorrhea. Mycological cure was defined as eradication (culture does not show growth of any fungal pathogen) or presumed eradication (there is no material to culture and the overall clinical outcome is clinical cure). In both trials, CLOTIC demonstrated superiority to placebo in the MITT population for the proportion of patients with therapeutic cure.
The results for therapeutic cure (both clinical cure and mycological cure) for each trial are presented in Table 2:
| Trial 1 | Trial 2 | |||
|---|---|---|---|---|
| CLOTIC N = 82 n (%) | Placebo N = 36 n (%) | CLOTIC N = 75 n (%) | Placebo N = 35 n (%) | |
| Therapeutic Cure | 48 (58.5) | 10 (27.8) | 59 (78.7) | 8 (22.9) |
| Treatment Difference (95% CI) 95% confidence interval for between-treatment group difference is based on Wald method | 30.8 (12.7, 48.9) | 55.8 (39.1, 72.5) | ||
| p-value | 0.0021 | < 0.0001 | ||
HOW SUPPLIED/STORAGE AND HANDLING
How supplied
CLOTIC is a sterile otic solution of clotrimazole 1% packaged in low-density polyethylene (LDPE) single-dose vials with a deliverable volume of approximately 0.17 mL. The solution is clear colorless to slightly yellow. The vials are contained in an aluminum foil overwrap pouch and a carton box for protection from moisture. Each carton box contains 2 foil pouches. Each pouch contains two strips of 7 single-dose vials. There are a total of 28 vials (NDC 15370-220-28).
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F). Brief exposure to 15°C to 30°C (59°F to 86°F) permitted (see USP Controlled Room Temperature).
Store unused vials in the foil pouch to protect from humidity.
Do not open foil pouch until ready to use. Discard vial after use.
Discard unused vials 30 days after first opening the pouch.
INSTRUCTIONS FOR USE CLOTIC ['klↄtık] (clotrimazole) otic solution
This Instructions for Use contains information on how to use CLOTIC.
Important Information You Need to Know Before Using CLOTIC:
- For otic use only (use in the ear).
- Use CLOTIC exactly as your healthcare provider tells you to use it.
- Apply the entire dose of CLOTIC from 1 single-dose vial, into the affected ear 2 times a day (for a total of 2 single-dose vials a day) for 14 days. Each dose should be about 12 hours apart.
- Do not use CLOTIC after the expiration date (EXP) appearing on the outer carton, pouch and single-dose container. The expiration date is the last day of that month, for example, 2025-DEC means the medicine expires December 31, 2025.
Using CLOTIC
Follow these instructions ( Step 1 to Step 11 ) each time you use CLOTIC. Ask your healthcare provider if there is anything you do not understand.
| Step 1. | Wash your hands with soap and water (Figure 1). |  |
| Step 2. | Gently clean any fluid (discharge) from the outer ear using a clean cloth or tissue (Figure 2). Do not put a cotton swab or any other object in the ear canal. |  |
| Step 3. | Remove CLOTIC from the protective foil pouch. Pull apart 1 single-dose vial of CLOTIC as shown, by tearing along the dotted lines (perforations) until it is fully separated (Figure 3). Place the unused vials back into the protective foil pouch |  |
| Step 4. | Warm the dose of CLOTIC by holding the vial in your hand for 1 to 2 minutes (Figure 4). | 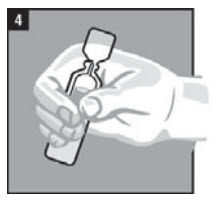 |
| Step 5. | Twist off the vial cap in the direction of the arrow as shown in Figure 5. | 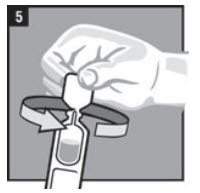 |
| Step 6. | The person receiving CLOTIC should be on his or her side with the affected ear up as shown in Figure 6. | 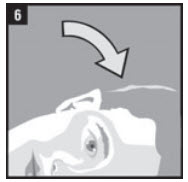 |
| Step 7. | Hold the vial of CLOTIC in your hand and place the vial close to the ear. Let the entire dose of CLOTIC fall into the affected ear (Figure 7). | 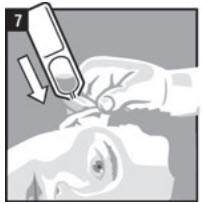 |
| Step 8. | Gently pull the outer ear lobe upward and outward (Figure 8). This will allow the ear drops to flow down into the ear canal. | 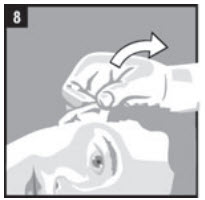 |
| Step 9. | Remain on your side with the affected ear facing upward for 1 minute (Figure 9). | 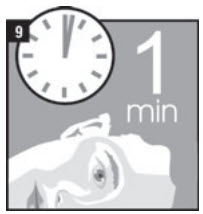 |
| Step 10. | If your healthcare provider has told you to use CLOTIC in both ears, repeat Steps 2 to 9 in the other ear. | |
| Step 11. | Safely throw away CLOTIC vials after use. |
How should I store CLOTIC?
- Store the carton at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not open the foil pouch until ready to use.
- Keep unused vials in the foil pouch to protect from humidity.
- Throw away unused vials 30 days after first opening the pouch.
- When a vial is opened, use it right away and throw away (discard) the vial after use.
Keep CLOTIC and all medicines out of the reach of children.
Manufactured by: PHARMALOOP, S.L., Pol. Industrial Azque – c/ Bolivia,15, Alcalá de Henares, 28806 MADRID
Distributed by: Carwin Pharmaceutical Associates, LLC 1301 Hwy 36 - Suite 11 Hazlet, NJ 07730 USA
Under license of Laboratorios Salvat, S.A.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Approved: October 2025
Mechanism of Action
Clotrimazole is an azole antifungal agent [see Microbiology (12.4) ] .