Get your patient on Clotrimazole - Clotrimazole solution (Clotrimazole)
Clotrimazole - Clotrimazole solution prescribing information
INDICATIONS AND USAGE
Prescription CLOTRIMAZOLE TOPICAL SOLUTION product is indicated for the topical treatment of candidiasis due to Candida albicans and tinea versicolor due to Malassezia furfur .
This formulation is also available as a nonprescription product which is indicated for the topical treatment of the following dermal infections: tinea pedis, tinea cruris, and tinea corporis due to Trichophyton rubrum, Trichophyton mentagrophytes , Epidermophyton fluoccosum , and Microsporum canis .
DOSAGE AND ADMINISTRATION
Gently massage sufficient CLOTRIMAZOLE TOPICAL SOLUTION USP, 1% into the affected and surrounding skin areas twice a day, in the morning and evening.
Clinical improvement, with relief of pruritus, usually occurs within the first week of treatment with CLOTRIMAZOLE TOPICAL SOLUTION USP, 1%. If the patient shows no clinical improvement after 4 weeks of treatment with CLOTRIMAZOLE TOPICAL SOLUTION USP, 1%, the diagnosis should be reviewed.
CONTRAINDICATIONS
Topical antifungal agents are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
ADVERSE REACTIONS
The following adverse reactions have been reported in connection with the use of clotrimazole: erythema, stinging, blistering, peeling, edema, pruritius, urticaria, burning, and general irritation of the skin.
Drug Interactions
Synergism or antagonism between clotrimazole and nystatin, or amphotericin B, or flucytosine against strains of C. albicans has not been reported.
DESCRIPTION
CLOTRIMAZOLE TOPICAL SOLUTION USP, 1% contains 10 mg clotrimazole USP, a synthetic antifungal agent having the chemical name 1-(o-Chloro-α,α-diphenylbenzyl) imidazole with the following structural formula:
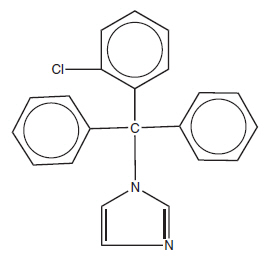
MOLECULAR FORMULA C 22 H 17 CIN 2 MOLECULAR WEIGHT 344.85
Clotrimazole is an odorless, white crystalline substance. It is practically insoluble in water, sparingly soluble in ether and very soluble in polyethylene glycol 400, ethanol, and chloroform.
Each mL of CLOTRIMAZOLE TOPICAL SOLUTION USP, 1% contains 10 mg clotrimazole USP in a nonaqueous vehicle of polyethylene glycol 400.
CLINICAL PHARMACOLOGY
Clotrimazole is a broad-spectrum antifungal agent that is used for the treatment of dermal infections caused by various species of pathogenic dermatophytes, yeasts, and Malassezia furfur . The primary action of clotrimazole is against dividing and growing organisms.
In vitro , clotrimazole exhibits fungistatic and fungicidal activity against isolates of Trichophyton rubrum, Trichophyton mentagrophytes, Epidermophyton floccosum, Microsporum canis , and Candida species including Candida albicans . In general, the in vitro activity of clotrimazole corresponds to that of tolnaftate and griseofulvin against the mycelia of dermatophytes ( Trichophyton, Microsporum , and Epidermophyton ), and to that of the polyenes (amphotericin B and nystatin) against budding fungi ( Candida ). Using an in vivo (mouse) and an in vitro (mouse kidney homogenate) testing system, clotrimazole and miconazole were equally effective in preventing the growth of the pseudomycelia and mycelia of Candida albicans .
Strains of fungi having a natural resistance to clotrimazole are rare. Only a single isolate of Candida guilliermondi has been reported to have primary resistance to clotrimazole.
No single-step or multiple-step resistance to clotrimazole has developed during successive passages of Candida albicans and Trichophyton mentagrophytes . No appreciable change in sensitivity was detected after successive passages of isolates of C. albicans, C. krusei , or C. pseudotropicalis in liquid or solid media containing clotrimazole. Also, resistance could not be developed in chemically induced mutant strains of polyene-resistant isolates of C. albicans .
Slight, reversible resistance was noted in three isolates of C. albicans tested by one investigator. There is a single report that records the clinical emergence of a C. albicans strain with considerable resistance to flucytosine and miconazole, and with cross-resistance to clotrimazole; the strain remained sensitive to nystatin and amphotericin B.
In studies of the mechanism of action, the minimum fungicidal concentration of clotrimazole caused leakage of intracellular phosphorus compounds into the ambient medium with concomitant breakdown of cellular nucleic acids and accelerated potassium efflux. Both these events began rapidly and extensively after addition of the drug.
Clotrimazole appears to be well absorbed in humans following oral administration and is eliminated mainly as inactive metabolites. Following topical and vaginal administration, however, clotrimazole appears to be minimally absorbed.
Six hours after the application of radioactive clotrimazole 1% cream and 1% solution onto intact and acutely inflamed skin, the concentration of clotrimazole varied from 100 mcg/cm 3 , in the stratum corneum to 0.5 to 1 mcg/cm 3 in the stratum reticulare, and 0.1 mcg/cm 3 in the subcutis.
No measurable amount of radioactivity (≤0.001 mcg/mL) was found in the serum within 48 hours after application under occlusive dressing of 0.5 mL of the solution or 0.8 g of the cream.
Only 0.5% or less of the applied radioactivity was excreted in the urine.
Following intravaginal administration of 100 mg 14 C-clotrimazole vaginal tablets to nine adult females, an average peak serum level, corresponding to only 0.03 µg equivalents/mL of clotrimazole, was reached 1 to 2 days after application. After intravaginal administration of 5 g of 1% 14 C-clotrimazole vaginal cream containing 50 mg active drug, to five subjects (one with candidal colpitis), serum levels corresponding to approximately 0.01 µg equivalents/mL were reached between 8 and 24 hours after application.
HOW SUPPLIED
CLOTRIMAZOLE TOPICAL SOLUTION USP, 1% is supplied in 30 mL plastic bottles (NDC 51672-1260-3); boxes of one.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].