Get your patient on Cortifoam - Hydrocortisone Acetate aerosol, Foam (Hydrocortisone Acetate)
Cortifoam - Hydrocortisone Acetate aerosol, Foam prescribing information
INDICATIONS AND USAGE
Cortifoam ® is indicated as adjunctive therapy in the topical treatment of ulcerative proctitis of the distal portion of the rectum in patients who cannot retain hydrocortisone or other corticosteroid enemas.
DOSAGE AND ADMINISTRATION
The usual dose is one applicatorful once or twice daily for two or three weeks, and every second day thereafter, administered rectally. Directions for use, below and on the carton, describe how to use the aerosol container and applicator. Satisfactory response usually occurs within five to seven days marked by a decrease in symptoms. Symptomatic improvement in ulcerative proctitis should not be used as the sole criterion for evaluating efficacy. Sigmoidoscopy is also recommended to judge dosage adjustment, duration of therapy, and rate of improvement.
It Should Be Emphasized that Dosage Requirements are Variable and Must Be Individualized on the Basis of the Disease Under Treatment and the Response of the Patient. After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage which will maintain an adequate clinical response is reached. Situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient's individual drug responsiveness, and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this latter situation it may be necessary to increase the dosage of the corticosteroid for a period of time consistent with the patient's condition. If after long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly.
Directions For Use
(1) Shake foam container vigorously for 5-10 seconds before each use. Do not remove container cap during use of the product.
(2) Hold container upright on a level surface and gently place the tip of the applicator onto the nose of the container cap. CONTAINER MUST BE HELD UPRIGHT TO OBTAIN PROPER FLOW OF MEDICATION.
(3) Pull plunger past the fill line on the applicator barrel.
(4) To fill applicator barrel, press down firmly on cap flanges, hold for 1 – 2 seconds and release. Pause 5 – 10 seconds to allow foam to expand in applicator barrel. Repeat until foam reaches fill line. Remove applicator from container cap. Allow some foam to remain on the applicator tip. A burst of air may come out of container with first pump.
(5) Hold applicator firmly by barrel, making sure thumb and middle finger are positioned securely underneath and resting against barrel wings. Place index finger over the plunger. Gently insert tip into anus. Once in place, push plunger to expel foam, then withdraw applicator.
CAUTION: Do not insert any part of the aerosol container directly into the anus. Apply to anus only with enclosed applicator.
(6) After each use, applicator parts should be pulled apart for thorough cleaning with warm water. The container cap and underlying tip should also be pulled apart and rinsed to help prevent build-up of foam and possible blockage.
CONTRAINDICATIONS
Cortifoam ® is contraindicated in patients who are hypersensitive to any components of this product.
Local contraindications to the use of intrarectal steroids include obstruction, abscess, perforation, peritonitis, fresh intestinal anastomoses, extensive fistulas and sinus tracts.
ADVERSE REACTIONS
(listed alphabetically, under each subsection)
To report SUSPECTED ADVERSE REACTIONS, contact Meda Pharmaceuticals Inc. at 1-877-848-6608 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Allergic reactions: Anaphylactoid reaction, anaphylaxis, angioedema.
Cardiovascular: Bradycardia, cardiac arrest, cardiac arrhythmias, cardiac enlargement, circulatory collapse, congestive heart failure, fat embolism, hypertension, hypertrophic cardiomyopathy in premature infants, myocardial rupture following recent myocardial infarction (see WARNINGS ), pulmonary edema, syncope, tachycardia, thromboembolism, thrombophlebitis, vasculitis.
Dermatologic: Acne, allergic dermatitis, cutaneous and subcutaneous atrophy, dry scaly skin, ecchymoses and petechiae, edema, erythema, hyperpigmentation, hypopigmentation, impaired wound healing, increased sweating, rash, sterile abscess, striae, suppressed reactions to skin tests, thin fragile skin, thinning scalp hair, urticaria.
Endocrine: Decreased carbohydrate and glucose tolerance, development of cushingoid state, glycosuria, hirsutism, hypertrichosis, increased requirements for insulin or oral hypoglycemic agents in diabetes, manifestations of latent diabetes mellitus, menstrual irregularities, secondary adrenocortical and pituitary unresponsiveness (particularly in times of stress, as in trauma, surgery, or illness), suppression of growth in pediatric patients.
Fluid and electrolyte disturbances: Congestive heart failure in susceptible patients, fluid retention, hypokalemic alkalosis, potassium loss, sodium retention.
Where hypokalemia and other symptoms associated with fluid and electrolyte imbalance call for potassium supplementation and salt poor or salt-free diets, these may be instituted and are compatible with diet requirements for ulcerative proctitis.
Gastrointestinal: Abdominal distention, elevation in serum liver enzyme levels (usually reversible upon discontinuation), hepatomegaly, increased appetite, nausea, pancreatitis, peptic ulcer with possible perforation and hemorrhage, perforation of the small and large intestine (particularly in patients with inflammatory bowel disease), ulcerative esophagitis.
Metabolic: Negative nitrogen balance due to protein catabolism.
Musculoskeletal: Aseptic necrosis of femoral and humeral heads, Charcot-like arthropathy, loss of muscle mass, muscle weakness, osteoporosis, pathologic fracture of long bones, steroid myopathy, tendon rupture, vertebral compression fractures.
Neurologic/Psychiatric: Convulsions, depression, emotional instability, euphoria, headache, increased intracranial pressure with papilledema (pseudotumor cerebri) usually following discontinuation of treatment, insomnia, mood swings, neuritis, neuropathy, paresthesia, personality changes, psychic disorders, vertigo.
Ophthalmic: Blurred vision, exophthalmos, glaucoma, increased intraocular pressure, posterior subcapsular cataracts, rare instances of blindness associated with periocular injections.
Other: Abnormal fat deposits, decreased resistance to infection, hiccups, increased or decreased motility and number of spermatozoa, malaise, moon face, weight gain.
Drug Interactions
Aminoglutethimide: Aminoglutethimide may lead to a loss of corticosteroid-induced adrenal suppression.
Amphotericin B injection and potassium-depleting agents: When corticosteroids are administered concomitantly with potassium-depleting agents (i.e., amphotericin B, diuretics), patients should be observed closely for development of hypokalemia. There have been cases reported in which concomitant use of amphotericin B and hydrocortisone was followed by cardiac enlargement and congestive heart failure.
Antibiotics: Macrolide antibiotics have been reported to cause a significant decrease in corticosteroid clearance.
Anticholinesterases: Concomitant use of anticholinesterase agents and corticosteroids may produce severe weakness in patients with myasthenia gravis. If possible, anticholinesterase agents should be withdrawn at least 24 hours before initiating corticosteroid therapy.
Anticoagulants, oral: Coadministration of corticosteroids and warfarin result in inhibition of response to warfarin. Therefore, coagulation indices should be monitored to maintain the desired anticoagulant effect.
Antidiabetics: Because corticosteroids may increase blood glucose concentrations, dosage adjustments of antidiabetic agents may be required.
Antitubercular drugs: Serum concentrations of isoniazid may be decreased.
Cholestyramine: Cholestyramine may increase the clearance of corticosteroids.
Cyclosporine: Increased activity of both cyclosporine and corticosteroids may occur when the two are used concurrently. Convulsions have been reported with this concurrent use.
Digitalis glycosides: Patients on digitalis glycosides may be at increased risk of arrhythmias due to hypokalemia.
Estrogens, including oral contraceptives: Estrogens may decrease the hepatic metabolism of certain corticosteroids, thereby increasing their effect.
Hepatic Enzyme Inducers (e.g., barbiturates, phenytoin, carbamazepine, rifampin): Drugs which induce hepatic microsomal drug metabolizing enzyme activity may enhance the metabolism of corticosteroids and require that the dosage of the corticosteroid be increased.
Ketoconazole: Ketoconazole has been reported to decrease the metabolism of certain corticosteroids by up to 60%, leading to an increased risk of corticosteroids side effects.
Nonsteroidal anti-inflammatory agents (NSAIDS): Concomitant use of aspirin (or other nonsteroidal anti-inflammatory agents) and corticosteroids increases the risk of gastrointestinal side effects. Aspirin should be used cautiously in conjunction with corticosteroids in hypoprothrombinemia. The clearance of salicylates may be increased with concurrent use of corticosteroids.
Skin tests: Corticosteroids may suppress reactions to skin tests.
Vaccines: Patients on prolonged corticosteroid therapy may exhibit a diminished response to toxoids and live or inactivated vaccines due to inhibition of antibody response. Corticosteroids may also potentiate the replication of some organisms contained in live attenuated vaccines. Routine administration of vaccines or toxoids should be deferred until corticosteroid therapy is discontinued if possible (see WARNINGS, Infections, Vaccination ).
DESCRIPTION
Cortifoam ® (hydrocortisone acetate rectal aerosol) 10% Rectal Foam contains hydrocortisone acetate 10% in a base containing propylene glycol, emulsifying wax, polyoxyethylene-10-stearyl ether, cetyl alcohol, methylparaben, propylparaben, trolamine, purified water and inert propellants: isobutane and propane.
Each application delivers approximately 900 mg of foam containing 80 mg of hydrocortisone (90 mg of hydrocortisone acetate).
The molecular weight of hydrocortisone acetate is 404.50. It is designated chemically as pregn-4-ene-3,20-dione,21-(acetyloxy)-11,17-dihydroxy-,(11β)-. The empirical formula is C 23 H 32 O 6 and the structural formula is:
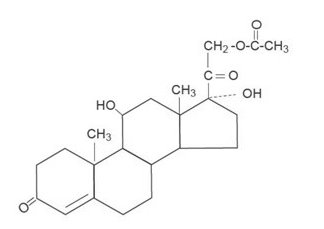
Hydrocortisone acetate, a synthetic adrenocortical steroid, is a white to practically white, odorless, crystalline powder. It is insoluble in water (1 mg/100 mL) and slightly soluble in alcohol and chloroform.
CLINICAL PHARMACOLOGY
Cortifoam ® provides effective topical administration of an anti-inflammatory corticosteroid as adjunctive therapy of ulcerative proctitis. Direct observations of methylene blue-containing foam have shown staining about 10 centimeters into the rectum.
HOW SUPPLIED
Cortifoam ® is supplied in an aerosol container with a special rectal applicator. Each applicator delivers approximately 900 mg of foam containing approximately 80 mg of hydrocortisone as 90 mg of hydrocortisone acetate. When used correctly, the aerosol container will deliver a minimum of 14 applications.
NDC 0037-6830-15 15 g
Store at controlled room temperature, 20°-25°C (68°-77°F).
DO NOT REFRIGERATE.
Rx Only
Distributed by: Meda Pharmaceuticals Inc. Canonsburg, PA 15317 U.S.A.
© 2024 Mylan Specialty L.P., a Viatris Company
For Medical Inquiries, call toll-free 1-877-848-6608
CORTIFOAM is a registered trademark of Alaven Pharmaceutical LLC, a Viatris Company.
IN-683015-05 141280-0324