Cortisporin Tc - Colistin Sulfate, Neomycin Sulfate, Thonzonium Bromide And Hydrocortisone Acetate suspension prescribing information
INDICATIONS AND USAGE
Cortisporin ® TC Otic is indicated for the treatment of superficial bacterial infections of the external auditory canal, caused by organisms susceptible to the action of the antibiotics; and for the treatment of infections of mastoidectomy and fenestration cavities, caused by organisms susceptible to the antibiotics.
DOSAGE AND ADMINISTRATION
Therapy with this product should be limited to 10 days (See WARNINGS ).
The external auditory canal should be thoroughly cleansed and dried with a sterile cotton applicator.
When using the calibrated dropper:
For adults, 5 drops of the suspension should be instilled into the affected ear 3 or 4 times daily. For pediatric patients, 4 drops are suggested because of the smaller capacity of the ear canal.
The patient should lie with the affected ear upward and then the drops should be instilled. This position should be maintained for 5 minutes to facilitate penetration of the drops into the ear canal. Repeat, if necessary, for the opposite ear.
If preferred, a cotton wick may be inserted into the canal and then the cotton may be saturated with the suspension. This wick should be kept moist by adding further solution every 4 hours. The wick should be replaced at least once every 24 hours.
CONTRAINDICATIONS
This product is contraindicated in those individuals who have shown hypersensitivity to any of its components.
This product should not be used if the external auditory canal disorder is suspected or known to be due to cutaneous viral infection (e.g., herpes simplex virus or varicella zoster virus).
ADVERSE REACTIONS
Neomycin occasionally causes skin sensitization.
Ototoxicity (see WARNINGS section) and nephrotoxicity have also been reported.
Adverse reactions have occurred with topical use of antibiotic combinations. Exact incidence figures are not available since no denominator of treated patients is available. The reaction occurring most often is allergic sensitization. In one clinical study, using a 20% neomycin patch, neomycin-induced allergic skin reactions occurred in two of 2,175 (0.09%) individuals in the general population. In another study the incidence was found to be approximately 1%.
The following local adverse events have been reported with topical corticosteroids, especially under occlusive dressings: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, and miliaria.
For medical advice about adverse reactions contact your medical professional. To report SUSPECTED ADVERSE REACTIONS, contact Endo at 1-800-462-3636 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Cortisporin ® TC Otic with Neomycin and Hydrocortisone (colistin sulfate—neomycin sulfate—thonzonium bromide—hydrocortisone acetate otic suspension) is a sterile antibacterial and anti-inflammatory aqueous suspension containing in each mL: Colistin base activity, 3 mg (as the sulfate); Neomycin base activity, 3.3 mg (as the sulfate); Hydrocortisone acetate, 10 mg (1%); Thonzonium bromide, 0.5 mg (0.05%); Polysorbate 80, acetic acid, and sodium acetate in a buffered aqueous vehicle. Thimerosal (mercury derivative), 0.002%, is added as a preservative. It is a nonviscous liquid, buffered at pH 5, for instillation into the canal of the external ear or direct application to the affected aural skin.
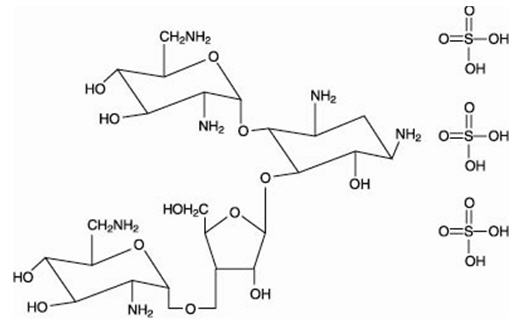
The structural formulas of colistin sulfate (mixture of Colistin A & B), neomycin sulfate (mixture of neomycin A, B & C), hydrocortisone acetate ((11β)-21-(acetyloxy)-11,17-dihydroxypregn) methyl]-2 pyrimidinylamino] ethyl]-N,N-dimethyl-1-hexadecanaminium, bromide) are represented below:
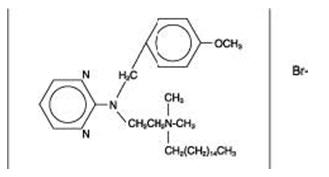
Thonzonium Bromide

Colistin sulfate
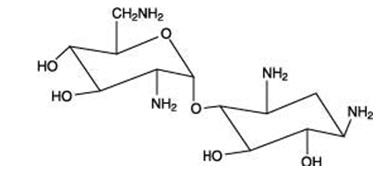
Neomycin A
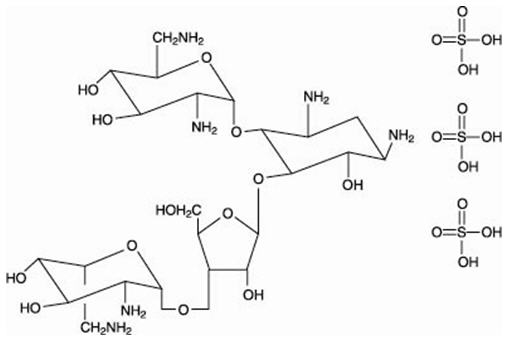
Neomycin B Sulfate
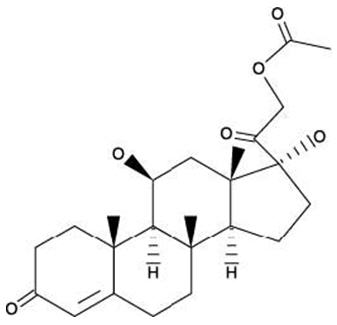
Hydrocortisone Acetate
Neomycin C Sulfate
CLINICAL PHARMACOLOGY
Colistin sulfate is a polypeptide antibiotic which penetrates into and disrupts the bacterial cell membrane. Neomycin sulfate is an aminoglycoside antibiotic which inhibits protein synthesis, disrupting the normal cycle of ribosomal function. Hydrocortisone acetate is a corticosteroid hormone which is thought to act by regulating the rate of protein synthesis; it controls inflammation, edema, pruritus and other dermal reactions. Corticosteroids suppress the inflammatory response to a variety of agents and they may delay healing. Since corticoids may inhibit the body's defense mechanism against infection, a concomitant antimicrobial drug may be used when this inhibition is considered to be clinically significant in a particular case.
The relative potency of corticosteroids depends on the molecular structure, concentration, and release from the vehicle.
Thonzonium bromide is a surface-active agent that promotes tissue contact by dispersion and penetration of the cellular debris and exudate.
Microbiology
Together, colistin sulfate and neomycin sulfate have bactericidal activity against most strains of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Aerobic gram-positive microorganisms:
Staphylococcus aureus.
Aerobic gram-negative microorganisms:
Enterobacter aerogenes
Escherichia coli
Klebsiella pneumoniae
Pseudomonas aeruginosa.
Susceptibility Tests:
It is not recommended that colistin sulfate or neomycin sulfate be routinely tested and reported by clinical microbiology laboratories.
HOW SUPPLIED
Cortisporin ® TC Otic is supplied as:
NDC 63481-529-10 10 mL bottle with dropper
Each mL contains: Colistin sulfate equivalent to 3 mg of colistin base activity, Neomycin sulfate equivalent to 3.3 mg neomycin base activity, Hydrocortisone acetate 10 mg (1%), Thonzonium bromide 0.5 mg (0.05%), and Polysorbate 80 in an aqueous vehicle buffered with acetic acid and sodium acetate. Thimerosal (mercury derivative) 0.002% is added as a preservative.
A sterilized dropper-cap assembly for use on the bottle of suspension is included in the package.
Shake well before using.
Store at 20° to 25°C (68° to 77°F).
Rx only.