Get your patient on Cosopt Pf (Dorzolamide Hydrochloride And Timolol Maleate)
Cosopt PF prior authorization resources
Most recent state uniform prior authorization forms
Dosage & administration
DOSAGE AND ADMINISTRATION
The dose is one drop of COSOPT PF in the affected eye(s) two times daily.
If more than one topical ophthalmic drug is being used, the drugs should be administered at least five minutes apart [see Drug Interactions (7.3) ].
The solution from one individual unit is to be used immediately after opening for administration to one or both eyes. Since sterility cannot be maintained after the individual unit is opened, the remaining contents should be discarded immediately after administration.

Cosopt PF prescribing information
INDICATIONS AND USAGE
COSOPT® PF is indicated for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension who are insufficiently responsive to beta-blockers (failed to achieve target IOP determined after multiple measurements over time).
The IOP-lowering of COSOPT® administered twice a day was slightly less than that seen with the concomitant administration of 0.5% timolol administered twice a day and 2% dorzolamide administered three times a day [see Clinical Studies (14.1) ].
DOSAGE AND ADMINISTRATION
The dose is one drop of COSOPT PF in the affected eye(s) two times daily.
If more than one topical ophthalmic drug is being used, the drugs should be administered at least five minutes apart [see Drug Interactions (7.3) ].
The solution from one individual unit is to be used immediately after opening for administration to one or both eyes. Since sterility cannot be maintained after the individual unit is opened, the remaining contents should be discarded immediately after administration.
DOSAGE FORMS AND STRENGTHS
Ophthalmic solution containing 2% (20 mg/mL) dorzolamide (22.26 mg of dorzolamide hydrochloride) and 0.5% (5 mg/mL) timolol (6.83 mg of timolol maleate).
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary There are no adequate and well-controlled studies of COSOPT PF in pregnant women to inform a drug-associated risk. No evidence of teratogenicity has been observed in mice, rat, and rabbits with dorzolamide hydrochloride or timolol at clinically relevant doses [see Data].
Data Animal Data Oral administration of dorzolamide hydrochloride in pregnant rabbits at oral doses of ≥2.5 mg/kg/day (31 times the recommended human ophthalmic dose) revealed malformations of the vertebral bodies. These malformations occurred at doses that caused metabolic acidosis with decreased body weight gain in dams and decreased fetal weights. No treatment-related malformations were seen at 1 mg/kg/day (13 times the recommended human ophthalmic dose).
Oral administration of timolol in pregnant mice, rats and rabbits at does up to 50 mg/kg/day (7,000 times the systemic exposure following the maximum recommended human ophthalmic dose) demonstrated no evidence of fetal malformations. Although delayed fetal ossification was observed at this dose in rats, there were no adverse effects on postnatal development of offspring. Doses of 1,000 mg/kg/day (142,000 times the systemic exposure following the maximum recommended human ophthalmic dose) were maternotoxic in mice and resulted in an increased number of fetal resorptions. Increased fetal resorptions were also seen in rabbits at doses of 14,000 times the systemic exposure following the maximum recommended human ophthalmic dose, in this case without apparent maternotoxicity.
Lactation
Risk Summary It is not known whether dorzolamide hydrochloride and timolol are excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when COSOPT PF is administered to a nursing woman. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for COSOPT PF and any potential adverse effects on the breastfed child from COSOPT PF.
Pediatric Use
The safety and effectiveness of dorzolamide hydrochloride ophthalmic solution and timolol maleate ophthalmic solution have been established when administered individually in pediatric patients aged 2 years and older. Use of these drug products in these children is supported by evidence from adequate and well-controlled studies in children and adults. Safety and efficacy in pediatric patients below the age of 2 years have not been established.
Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
CONTRAINDICATIONS
COSOPT PF is contraindicated in patients with:
Asthma, COPD
COSOPT PF is contraindicated in patients with bronchial asthma, a history of bronchial asthma, or severe chronic obstructive pulmonary disease [see Warnings and Precautions (5.1) ].
Sinus Bradycardia, AV Block, Cardiac Failure, Cardiogenic Shock
COSOPT PF is contraindicated in patients with sinus bradycardia, second or third degree atrioventricular block, overt cardiac failure, and cardiogenic shock [see Warnings and Precautions (5.2) ].
Hypersensitivity
COSOPT PF is contraindicated in patients who are hypersensitive to any component of this product [see Warnings and Precautions (5.3) ].
WARNINGS AND PRECAUTIONS
Potentiation of Respiratory Reactions Including Asthma
COSOPT PF contains timolol maleate, a beta-adrenergic blocking agent; and although administered topically, is absorbed systemically. Therefore, the same types of adverse reactions that are attributable to systemic administration of beta-adrenergic blocking agents may occur with topical administration. For example, severe respiratory reactions, including death due to bronchospasm in patients with asthma, and rarely death in association with cardiac failure, have been reported following systemic or ophthalmic administration of timolol maleate [see Contraindications (4.1) and Patient Counseling Information (17.1) ].
Cardiac Failure
Sympathetic stimulation may be essential for support of the circulation in individuals with diminished myocardial contractility, and its inhibition by beta-adrenergic receptor blockade may precipitate more severe failure.
In patients without a history of cardiac failure continued depression of the myocardium with beta-blocking agents over a period of time can, in some cases, lead to cardiac failure. At the first sign or symptom of cardiac failure, COSOPT PF should be discontinued [see Contraindications (4.2) and Patient Counseling Information (17) ].
Sulfonamide Hypersensitivity
COSOPT PF contains dorzolamide, a sulfonamide; and although administered topically, it is absorbed systemically. Therefore, the same types of adverse reactions that are attributable to sulfonamides may occur with topical administration of COSOPT PF. Fatalities have occurred, although rarely, due to severe reactions to sulfonamides including Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, agranulocytosis, aplastic anemia, and other blood dyscrasias. Sensitization may recur when a sulfonamide is readministered irrespective of the route of administration. If signs of serious reactions or hypersensitivity occur, discontinue the use of this preparation [ see Contraindications (4.3) and Patient Counseling Information (17) ].
Obstructive Pulmonary Disease
Patients with chronic obstructive pulmonary disease (e.g., chronic bronchitis, emphysema) of mild or moderate severity, bronchospastic disease, or a history of bronchospastic disease (other than bronchial asthma or a history of bronchial asthma, in which COSOPT PF is contraindicated) should, in general, not receive beta-blocking agents, including COSOPT PF [see Contraindications (4.1) and Patient Counseling Information (17) ] .
Increased Reactivity to Allergens
While taking beta-blockers, patients with a history of atopy or a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated accidental, diagnostic, or therapeutic challenge with such allergens. Such patients may be unresponsive to the usual doses of epinephrine used to treat anaphylactic reactions.
Potentiation of Muscle Weakness
Beta-adrenergic blockade has been reported to potentiate muscle weakness consistent with certain myasthenic symptoms (e.g., diplopia, ptosis, and generalized weakness). Timolol has been reported rarely to increase muscle weakness in some patients with myasthenia gravis or myasthenic symptoms.
Masking of Hypoglycemic Symptoms in Patients with Diabetes Mellitus
Beta-adrenergic blocking agents should be administered with caution in patients subject to spontaneous hypoglycemia or to diabetic patients (especially those with labile diabetes) who are receiving insulin or oral hypoglycemic agents. Beta-adrenergic receptor blocking agents may mask the signs and symptoms of acute hypoglycemia.
Masking of Thyrotoxicosis
Beta-adrenergic blocking agents may mask certain clinical signs (e.g., tachycardia) of hyperthyroidism. Patients suspected of developing thyrotoxicosis should be managed carefully to avoid abrupt withdrawal of beta-adrenergic blocking agents that might precipitate a thyroid storm.
Renal and Hepatic Impairment
Dorzolamide has not been studied in patients with severe renal impairment (CrCl <30 mL/min). Because dorzolamide and its metabolite are excreted predominantly by the kidney, COSOPT PF is not recommended in such patients.
Dorzolamide has not been studied in patients with hepatic impairment and should therefore be used with caution in such patients.
Impairment of Beta-Adrenergically Mediated Reflexes During Surgery
The necessity or desirability of withdrawal of beta-adrenergic blocking agents prior to major surgery is controversial. Beta-adrenergic receptor blockade impairs the ability of the heart to respond to beta-adrenergically mediated reflex stimuli. This may augment the risk of general anesthesia in surgical procedures. Some patients receiving beta-adrenergic receptor blocking agents have experienced protracted severe hypotension during anesthesia. Difficulty in restarting and maintaining the heartbeat has also been reported. For these reasons, in patients undergoing elective surgery, some authorities recommend gradual withdrawal of beta-adrenergic receptor blocking agents.
If necessary during surgery, the effects of beta-adrenergic blocking agents may be reversed by sufficient doses of adrenergic agonists.
Corneal Endothelium
Carbonic anhydrase activity has been observed in both the cytoplasm and around the plasma membranes of the corneal endothelium. There is an increased potential for developing corneal edema in patients with low endothelial cell counts. Caution should be used when prescribing COSOPT PF to this group of patients.
ADVERSE REACTIONS
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
COSOPT and COSOPT PF COSOPT and COSOPT PF were evaluated in patients with elevated intraocular pressure treated for open-angle glaucoma or ocular hypertension for up to 15 months. Approximately 5% of all patients discontinued therapy because of adverse reactions.
The most frequently reported adverse reactions occurring in up to 30% of patients were taste perversion (bitter, sour, or unusual taste) or ocular burning and/or stinging. The following adverse reactions were reported in 5% to 15% of patients: conjunctival hyperemia, blurred vision, superficial punctate keratitis or eye itching.
The following adverse reactions were reported in 1% to 5% of patients: abdominal pain, back pain, blepharitis, bronchitis, cloudy vision, conjunctival discharge, conjunctival edema, conjunctival follicles, conjunctival injection, conjunctivitis, corneal erosion, corneal staining, cortical lens opacity, cough, dizziness, dryness of eyes, dyspepsia, eye debris, eye discharge, eye pain, eye tearing, eyelid edema, eyelid erythema, eyelid exudate/scales, eyelid pain or discomfort, foreign body sensation, glaucomatous cupping, headache, hypertension, influenza, lens nucleus coloration, lens opacity, nausea, nuclear lens opacity, pharyngitis, post-subcapsular cataract, sinusitis, upper respiratory infection, urinary tract infection, visual field defect, vitreous detachment. Other adverse reactions that have been reported with the individual components are listed below:
Dorzolamide 2%
Angioedema, asthenia/fatigue, bronchospasm, contact dermatitis, epistaxis, eyelid crusting, ocular discomfort, photophobia, signs and symptoms of ocular allergic reaction, transient myopia.
Timolol (ocular administration)
Body as a Whole: Asthenia/fatigue; Cardiovascular: Arrhythmia, syncope, cerebral ischemia, worsening of angina pectoris, palpitation, cardiac arrest, pulmonary edema, edema, claudication, Raynaud's phenomenon, and cold hands and feet; Digestive: Anorexia; Immunologic: Systemic lupus erythematosus; Nervous System/Psychiatric: Increase in signs and symptoms of myasthenia gravis, somnolence, insomnia, nightmares, behavioral changes and psychic disturbances including confusion, hallucinations, anxiety, disorientation, nervousness, and memory loss; Skin: Alopecia, psoriasiform rash or exacerbation of psoriasis; Hypersensitivity: Signs and symptoms of systemic allergic reactions, including anaphylaxis, angioedema, urticaria, and localized and generalized rash; Respiratory: Bronchospasm (predominantly in patients with pre-existing bronchospastic disease); Endocrine: Masked symptoms of hypoglycemia in diabetic patients; Special Senses: Ptosis, decreased corneal sensitivity, cystoid macular edema, visual disturbances including refractive changes and diplopia, pseudopemphigoid, and tinnitus; Urogenital: Retroperitoneal fibrosis, decreased libido, impotence, and Peyronie's disease.
Post-Marketing Experience
The following adverse reactions have been identified during postapproval use of COSOPT or COSOPT PF. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: bradycardia, cardiac failure, cerebral vascular accident, chest pain, choroidal detachment following filtration surgery, depression, diarrhea, dry mouth, dyspnea, heart block, hypotension, iridocyclitis, myocardial infarction, nasal congestion, Stevens-Johnson syndrome, toxic epidermal necrolysis, paresthesia, photophobia, respiratory failure, skin rashes, urolithiasis, and vomiting.
Timolol (oral administration) The following additional adverse reactions have been reported in clinical experience with ORAL timolol maleate or other ORAL beta-blocking agents and may be considered potential effects of ophthalmic timolol maleate: Allergic: Erythematous rash, fever combined with aching and sore throat, laryngospasm with respiratory distress; Body as a Whole: Extremity pain, decreased exercise tolerance, weight loss; Cardiovascular: Worsening of arterial insufficiency, vasodilatation; Digestive: Gastrointestinal pain, hepatomegaly, mesenteric arterial thrombosis, ischemic colitis; Hematologic: Nonthrombocytopenic purpura; thrombocytopenic purpura, agranulocytosis; Endocrine: Hyperglycemia, hypoglycemia; Skin: Pruritus, skin irritation, increased pigmentation, sweating; Musculoskeletal: Arthralgia; Nervous System/Psychiatric: Vertigo, local weakness, diminished concentration, reversible mental depression progressing to catatonia, an acute reversible syndrome characterized by disorientation for time and place, emotional lability, slightly clouded sensorium, and decreased performance on neuropsychometrics; Respiratory: Rales, bronchial obstruction; Urogenital: Urination difficulties.
DRUG INTERACTIONS
- Potential additive effect of oral carbonic anhydrase inhibitor with COSOPT PF. (7.1 )
- Potential acid-base and electrolyte disturbances. (7.2 )
- Concomitant use with systemic beta-blockers may potentiate systemic beta-blockade. (7.3 )
- Oral or intravenous calcium antagonists may cause atrioventricular conduction disturbances, left ventricular failure, and hypotension. (7.4 )
- Catecholamine-depleting drugs may have additive effects and produce hypotension and/or marked bradycardia. (7.5 )
- Digitalis and calcium antagonists may have additive effects in prolonging atrioventricular conduction time. (7.6 )
- CYP2D6 inhibitors may potentiate systemic beta-blockade. (7.7 )
Oral Carbonic Anhydrase Inhibitors
There is a potential for an additive effect on the known systemic effects of carbonic anhydrase inhibition in patients receiving an oral carbonic anhydrase inhibitor and COSOPT PF. The concomitant administration of COSOPT PF and oral carbonic anhydrase inhibitors is not recommended.
High-Dose Salicylate Therapy
Although acid-base and electrolyte disturbances were not reported in the clinical trials with dorzolamide hydrochloride ophthalmic solution, these disturbances have been reported with oral carbonic anhydrase inhibitors and have, in some instances, resulted in drug interactions (e.g., toxicity associated with high-dose salicylate therapy). Therefore, the potential for such drug interactions should be considered in patients receiving COSOPT PF.
Beta-Adrenergic Blocking Agents
Patients who are receiving a beta-adrenergic blocking agent orally and COSOPT PF should be observed for potential additive effects of beta-blockade, both systemic and on intraocular pressure. The concomitant use of two topical beta-adrenergic blocking agents is not recommended.
Calcium Antagonists
Caution should be used in the coadministration of beta-adrenergic blocking agents, such as COSOPT PF, and oral or intravenous calcium antagonists because of possible atrioventricular conduction disturbances, left ventricular failure, and hypotension. In patients with impaired cardiac function, coadministration should be avoided.
Catecholamine-Depleting Drugs
Close observation of the patient is recommended when a beta-blocker is administered to patients receiving catecholamine-depleting drugs such as reserpine, because of possible additive effects and the production of hypotension and/or marked bradycardia, which may result in vertigo, syncope, or postural hypotension.
Digitalis and Calcium Antagonists
The concomitant use of beta-adrenergic blocking agents with digitalis and calcium antagonists may have additive effects in prolonging atrioventricular conduction time.
CYP2D6 Inhibitors
Potentiated systemic beta-blockade (e.g., decreased heart rate, depression) has been reported during combined treatment with CYP2D6 inhibitors (e.g., quinidine, SSRIs) and timolol.
Clonidine
Oral beta-adrenergic blocking agents may exacerbate the rebound hypertension which can follow the withdrawal of clonidine. There have been no reports of exacerbation of rebound hypertension with ophthalmic timolol maleate.
DESCRIPTION
COSOPT PF (dorzolamide hydrochloride and timolol maleate ophthalmic solution) is a combination of dorzolamide, a topical carbonic anhydrase inhibitor, and timolol maleate, a topical beta-adrenergic receptor blocking agent.
Dorzolamide hydrochloride is described chemically as: (4 S-trans)-4-(ethylamino)-5,6-dihydro-6-methyl-4 H-thieno[2,3- b]thiopyran-2-sulfonamide 7,7-dioxide monohydrochloride. Dorzolamide hydrochloride is optically active. The specific rotation is:
| [α] | 25°C 405 nm | (C=1, water) = ~ -17°. |
Its empirical formula is C 10 H 16 N 2 O 4 S 3 ∙HCl and its structural formula is:

Dorzolamide hydrochloride has a molecular weight of 360.91. It is a white to off-white, crystalline powder, which is soluble in water and slightly soluble in methanol and ethanol.
Timolol maleate is described chemically as: (-)-1-( tert-butylamino)-3-[(4-morpholino-1,2,5-thiadiazol-3-yl)oxy]-2-propanol maleate (1:1) (salt). Timolol maleate possesses an asymmetric carbon atom in its structure and is provided as the levo-isomer. The optical rotation of timolol maleate is:
| [α] | 25°C 405 nm | in 1N HCl (C = 5) = -12.2° (-11.7° to -12.5°). |
Its molecular formula is C 13 H 24 N 4 O 3 S∙C 4 H 4 O 4 and its structural formula is:

Timolol maleate has a molecular weight of 432.50. It is a white, odorless, crystalline powder which is soluble in water, methanol, and alcohol. Timolol maleate is stable at room temperature.
COSOPT PF is supplied as a sterile, clear, colorless to nearly colorless, isotonic, buffered, slightly viscous, aqueous solution. The pH of the solution is approximately 5.65, and the osmolarity is 242-323 mOsM. Each mL of COSOPT PF contains 20 mg dorzolamide (22.26 mg of dorzolamide hydrochloride) and 5 mg timolol (6.83 mg timolol maleate).
Inactive ingredients are hydroxyethyl cellulose, mannitol, sodium citrate, sodium hydroxide, and water for injection.
COSOPT PF does not contain a preservative.
CLINICAL PHARMACOLOGY
Mechanism of Action
COSOPT PF is comprised of two components: dorzolamide hydrochloride and timolol maleate. Each of these two components decreases elevated intraocular pressure, whether or not associated with glaucoma, by reducing aqueous humor secretion. Elevated intraocular pressure is a major risk factor in the pathogenesis of optic nerve damage and glaucomatous visual field loss. The higher the level of intraocular pressure, the greater the likelihood of glaucomatous field loss and optic nerve damage.
Dorzolamide hydrochloride is an inhibitor of human carbonic anhydrase II. Inhibition of carbonic anhydrase in the ciliary processes of the eye decreases aqueous humor secretion, presumably by slowing the formation of bicarbonate ions with subsequent reduction in sodium and fluid transport. Timolol maleate is a beta 1 and beta 2 (non-selective) adrenergic receptor blocking agent that does not have significant intrinsic sympathomimetic, direct myocardial depressant, or local anesthetic (membrane-stabilizing) activity. The combined effect of these two agents administered as COSOPT PF administered twice daily results in additional intraocular pressure reduction compared to either component administered alone, but the reduction is not as much as when dorzolamide administered three times daily and timolol twice daily are administered concomitantly. [See Clinical Studies (14) .]
Pharmacokinetics
Dorzolamide Hydrochloride
When topically applied, dorzolamide reaches the systemic circulation. To assess the potential for systemic carbonic anhydrase inhibition following topical administration, drug and metabolite concentrations in RBCs and plasma and carbonic anhydrase inhibition in RBCs were measured. Dorzolamide accumulates in RBCs during chronic dosing as a result of binding to CA-II. The parent drug forms a single N-desethyl metabolite, which inhibits CA-II less potently than the parent drug but also inhibits CA-I. The metabolite also accumulates in RBCs where it binds primarily to CA-I. Plasma concentrations of dorzolamide and metabolite are generally below the assay limit of quantitation (15nM). Dorzolamide binds moderately to plasma proteins (approximately 33%).
Dorzolamide is primarily excreted unchanged in the urine; the metabolite also is excreted in urine. After dosing is stopped, dorzolamide washes out of RBCs nonlinearly, resulting in a rapid decline of drug concentration initially, followed by a slower elimination phase with a half-life of about four months.
To simulate the systemic exposure after long-term topical ocular administration, dorzolamide was given orally to eight healthy subjects for up to 20 weeks. The oral dose of 2 mg twice daily closely approximates the amount of drug delivered by topical ocular administration of dorzolamide 2% three times daily. Steady state was reached within 8 weeks. The inhibition of CA-II and total carbonic anhydrase activities was below the degree of inhibition anticipated to be necessary for a pharmacological effect on renal function and respiration in healthy individuals.
Timolol Maleate
In a study of plasma drug concentrations in six subjects, the systemic exposure to timolol was determined following twice daily topical administration of timolol maleate ophthalmic solution 0.5%. The mean peak plasma concentration following morning dosing was 0.46 ng/mL.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a two-year study of dorzolamide hydrochloride administered orally to male and female Sprague-Dawley rats, urinary bladder papillomas were seen in male rats in the highest dosage group of 20 mg/kg/day (250 times the recommended human ophthalmic dose). Papillomas were not seen in rats given oral doses equivalent to approximately 12 times the recommended human ophthalmic dose. No treatment-related tumors were seen in a 21-month study in female and male mice given oral doses up to 75 mg/kg/day (~900 times the recommended human ophthalmic dose).
The increased incidence of urinary bladder papillomas seen in the high-dose male rats is a class-effect of carbonic anhydrase inhibitors in rats. Rats are particularly prone to developing papillomas in response to foreign bodies, compounds causing crystalluria, and diverse sodium salts.
No changes in bladder urothelium were seen in dogs given oral dorzolamide hydrochloride for one year at 2 mg/kg/day (25 times the recommended human ophthalmic dose) or monkeys dosed topically to the eye at 0.4 mg/kg/day (~5 times the recommended human ophthalmic dose) for one year.
In a two-year study of timolol maleate administered orally to rats, there was a statistically significant increase in the incidence of adrenal pheochromocytomas in male rats administered 300 mg/kg/day (approximately 42,000 times the systemic exposure following the maximum recommended human ophthalmic dose). Similar differences were not observed in rats administered oral doses equivalent to approximately 14,000 times the maximum recommended human ophthalmic dose.
In a lifetime oral study of timolol maleate in mice, there were statistically significant increases in the incidence of benign and malignant pulmonary tumors, benign uterine polyps and mammary adenocarcinomas in female mice at 500 mg/kg/day, (approximately 71,000 times the systemic exposure following the maximum recommended human ophthalmic dose), but not at 5 or 50 mg/kg/day (approximately 700 or 7,000, respectively, times the systemic exposure following the maximum recommended human ophthalmic dose). In a subsequent study in female mice, in which post-mortem examinations were limited to the uterus and the lungs, a statistically significant increase in the incidence of pulmonary tumors was again observed at 500 mg/kg/day.
The increased occurrence of mammary adenocarcinomas was associated with elevations in serum prolactin which occurred in female mice administered oral timolol at 500 mg/kg/day, but not at doses of 5 or 50 mg/kg/day. An increased incidence of mammary adenocarcinomas in rodents has been associated with administration of several other therapeutic agents that elevate serum prolactin, but no correlation between serum prolactin levels and mammary tumors has been established in humans. Furthermore, in adult human female subjects who received oral dosages of up to 60 mg of timolol maleate (the maximum recommended human oral dosage), there were no clinically meaningful changes in serum prolactin.
Mutagenesis
The following tests for mutagenic potential were negative for dorzolamide: (1) in vivo (mouse) cytogenetic assay; (2) in vitro chromosomal aberration assay; (3) alkaline elution assay; (4) V-79 assay; and (5) Ames test.
Timolol maleate was devoid of mutagenic potential when tested in vivo (mouse) in the micronucleus test and cytogenetic assay (doses up to 800 mg/kg) and in vitro in a neoplastic cell transformation assay (up to 100 μg/mL). In Ames tests the highest concentrations of timolol employed, 5,000 or 10,000 μg/plate, were associated with statistically significant elevations of revertants observed with tester strain TA100 (in seven replicate assays), but not in the remaining three strains. In the assays with tester strain TA100, no consistent dose response relationship was observed, and the ratio of test to control revertants did not reach 2. A ratio of 2 is usually considered the criterion for a positive Ames test.
Impairment of Fertility
Reproduction and fertility studies in rats with either timolol maleate or dorzolamide hydrochloride demonstrated no adverse effect on male or female fertility at doses up to approximately 100 times the systemic exposure following the maximum recommended human ophthalmic dose.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
COSOPT and COSOPT PF COSOPT and COSOPT PF were evaluated in patients with elevated intraocular pressure treated for open-angle glaucoma or ocular hypertension for up to 15 months. Approximately 5% of all patients discontinued therapy because of adverse reactions.
The most frequently reported adverse reactions occurring in up to 30% of patients were taste perversion (bitter, sour, or unusual taste) or ocular burning and/or stinging. The following adverse reactions were reported in 5% to 15% of patients: conjunctival hyperemia, blurred vision, superficial punctate keratitis or eye itching.
The following adverse reactions were reported in 1% to 5% of patients: abdominal pain, back pain, blepharitis, bronchitis, cloudy vision, conjunctival discharge, conjunctival edema, conjunctival follicles, conjunctival injection, conjunctivitis, corneal erosion, corneal staining, cortical lens opacity, cough, dizziness, dryness of eyes, dyspepsia, eye debris, eye discharge, eye pain, eye tearing, eyelid edema, eyelid erythema, eyelid exudate/scales, eyelid pain or discomfort, foreign body sensation, glaucomatous cupping, headache, hypertension, influenza, lens nucleus coloration, lens opacity, nausea, nuclear lens opacity, pharyngitis, post-subcapsular cataract, sinusitis, upper respiratory infection, urinary tract infection, visual field defect, vitreous detachment. Other adverse reactions that have been reported with the individual components are listed below:
Dorzolamide 2%
Angioedema, asthenia/fatigue, bronchospasm, contact dermatitis, epistaxis, eyelid crusting, ocular discomfort, photophobia, signs and symptoms of ocular allergic reaction, transient myopia.
Timolol (ocular administration)
Body as a Whole: Asthenia/fatigue; Cardiovascular: Arrhythmia, syncope, cerebral ischemia, worsening of angina pectoris, palpitation, cardiac arrest, pulmonary edema, edema, claudication, Raynaud's phenomenon, and cold hands and feet; Digestive: Anorexia; Immunologic: Systemic lupus erythematosus; Nervous System/Psychiatric: Increase in signs and symptoms of myasthenia gravis, somnolence, insomnia, nightmares, behavioral changes and psychic disturbances including confusion, hallucinations, anxiety, disorientation, nervousness, and memory loss; Skin: Alopecia, psoriasiform rash or exacerbation of psoriasis; Hypersensitivity: Signs and symptoms of systemic allergic reactions, including anaphylaxis, angioedema, urticaria, and localized and generalized rash; Respiratory: Bronchospasm (predominantly in patients with pre-existing bronchospastic disease); Endocrine: Masked symptoms of hypoglycemia in diabetic patients; Special Senses: Ptosis, decreased corneal sensitivity, cystoid macular edema, visual disturbances including refractive changes and diplopia, pseudopemphigoid, and tinnitus; Urogenital: Retroperitoneal fibrosis, decreased libido, impotence, and Peyronie's disease.
HOW SUPPLIED/STORAGE AND HANDLING
COSOPT PF is supplied in a foil pouch containing 15 low density polyethylene 0.2 mL single-dose containers.
NDC 52584-604-30, package of 60 single-dose vials.
Store COSOPT PF at 20° to 25°C (68° to 77°F). Do not freeze.
Store in the original pouch. After the pouch is opened, store the remaining single-dose containers in the foil pouch to protect from light. Write down the date you open the foil pouch in the space provided on the pouch. Discard any unused containers 15 days after first opening the pouch.
Instructions for Use
COSOPT®PF (CO-sopt PEA EHF) (dorzolamide hydrochloride and timolol maleate ophthalmic solution) 2%/0.5% For topical ophthalmic use
Read these Instructions for Use before using your COSOPT PF and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your healthcare provider about your medical condition or your treatment.
Important: • COSOPT PF is for use in the eye. Do not swallow COSOPT PF. • COSOPT PF single-dose containers are packaged in a foil pouch. • Write down the date you open the foil pouch in the space provided on the pouch.
Every time you use COSOPT PF:
Step 1. Wash your hands.
Step 2. Take the strip of single-dose containers from the pouch.
Step 3. Pull off 1 single-dose container from the strip ( see Figure A ).

(Figure A)
Step 4. Put the remaining strip of single-dose containers back in the pouch and fold the edge to close the pouch.
Step 5. Hold the single-dose container upright. The container may appear empty if the solution is in the tip of the container. Make sure that the solution is in the bottom part of the single-dose container before opening ( see Figure B ).

(Figure B)
Step 6. Open the single-dose container by twisting off the tab (see Figure C and Figure D).

(Figure C)

(Figure D)
Step 7. Tilt your head backwards. If you are unable to tilt your head, lie down.
Step 8. Place the tip of the single-dose container close to your eye. Be careful not to touch your eye with the tip of the single-dose container.
Step 9. Pull the lower eyelid downwards and look up.
Step 10. Gently squeeze the container and let 1 drop of COSOPT PF fall into the space between your lower eyelid and your eye. If a drop misses your eye, try again (see Figure E).
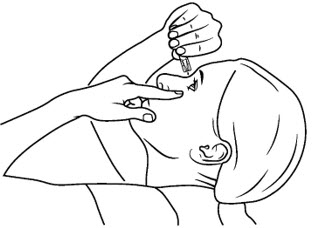
(Figure E)
Step 11. Blot any excess solution from the skin around the eye with a tissue.
• If your healthcare provider has told you to use drops in both eyes, repeat steps 7 to 11 for your other eye. • There is enough COSOPT PF in 1 single-dose container for 1 or both of your eyes. • Throw away the opened single-dose container with any remaining COSOPT PF right away.
Manufactured for: Thea Pharma Inc. Waltham, MA 02451
The COSOPT trademark is owned by Merck Sharp & Dohme Corp. and is used under license.
© 2026. Thea Pharma Inc. All rights reserved
This Instructions for Use have been approved by the U.S. Food and Drug Administration.
Revised: 4/2026
Mechanism of Action
COSOPT PF is comprised of two components: dorzolamide hydrochloride and timolol maleate. Each of these two components decreases elevated intraocular pressure, whether or not associated with glaucoma, by reducing aqueous humor secretion. Elevated intraocular pressure is a major risk factor in the pathogenesis of optic nerve damage and glaucomatous visual field loss. The higher the level of intraocular pressure, the greater the likelihood of glaucomatous field loss and optic nerve damage.
Dorzolamide hydrochloride is an inhibitor of human carbonic anhydrase II. Inhibition of carbonic anhydrase in the ciliary processes of the eye decreases aqueous humor secretion, presumably by slowing the formation of bicarbonate ions with subsequent reduction in sodium and fluid transport. Timolol maleate is a beta 1 and beta 2 (non-selective) adrenergic receptor blocking agent that does not have significant intrinsic sympathomimetic, direct myocardial depressant, or local anesthetic (membrane-stabilizing) activity. The combined effect of these two agents administered as COSOPT PF administered twice daily results in additional intraocular pressure reduction compared to either component administered alone, but the reduction is not as much as when dorzolamide administered three times daily and timolol twice daily are administered concomitantly. [See Clinical Studies (14) .]