Get your patient on Cromolyn Sodium Inhalation Solution - Cromolyn Sodium Inhalation Solution solution (Cromolyn Sodium Inhalation Solution)
Cromolyn Sodium Inhalation Solution - Cromolyn Sodium Inhalation Solution solution prescribing information
Indications and Usage
Cromolyn sodium inhalation solution USP is a prophylactic agent indicated in the management of patients with bronchial asthma.
In patients whose symptoms are sufficiently frequent to require a continuous program of medication, cromolyn sodium inhalation solution USP is given by inhalation on a regular daily basis (see DOSAGE AND ADMINISTRATION ). The effect of cromolyn sodium is usually evident after several weeks of treatment, although some patients show an almost immediate response.
In patients who develop acute bronchoconstriction in response to exposure to exercise, toluene diisocyanate, environmental pollutants, etc., cromolyn sodium should be given shortly before exposure to the precipitating factor (see DOSAGE AND ADMINISTRATION ).
Dosage and Administration
For management of bronchial asthma in adults and pediatric patients (two years of age and over), the usual starting dosage is the contents of one vial administered by nebulization four times a day at regular intervals.
Drug stability and safety of cromolyn sodium inhalation solution when mixed with other drugs in a nebulizer have not been established.
Patients with chronic asthma should be advised that the effect of cromolyn sodium inhalation solution therapy is dependent upon its administration at regular intervals, as directed. Cromolyn sodium inhalation solution should be introduced into the patient's therapeutic regimen when the acute episode has been controlled, the airway has been cleared and the patient is able to inhale adequately.
For the prevention of acute bronchospasm which follows exercise or exposure to cold dry air, environmental agents (e.g., animal danders, toluene diisocyanate, pollutants), etc., the usual dose is the contents of one vial administered by nebulization shortly before exposure to the precipitating factor.
It should be emphasized to the patient that the drug is poorly absorbed when swallowed and is not effective by this route of administration.
For additional information, see the accompanying leaflet entitled “Living a Full Life with Asthma” .
Cromolyn Sodium Inhalation Solution Therapy in Relation to Other Treatments for Asthma:
Non-steroidal agents:
Cromolyn sodium inhalation solution should be added to the patient's existing treatment regimen (e.g., bronchodilators). When a clinical response to cromolyn sodium inhalation solution is evident, usually within two to four weeks, and if the asthma is under good control, an attempt may be made to decrease concomitant medication usage gradually.
If concomitant medications are eliminated or required on no more than a prn basis, the frequency of administration of cromolyn sodium inhalation solution may be titrated downward to the lowest level consistent with the desired effect. The usual decrease is from four to three vials per day. It is important that the dosage be reduced gradually to avoid exacerbation of asthma. It is emphasized that in patients whose dosage has been titrated to fewer than four vials per day, an increase in the dose of cromolyn sodium inhalation solution and the introduction of, or increase in, symptomatic medications may be needed if the patient's clinical condition deteriorates.
Corticosteroids:
In patients chronically receiving corticosteroids for the management of bronchial asthma, the dosage should be maintained following the introduction of cromolyn sodium inhalation solution. If the patient improves, an attempt to decrease corticosteroids should be made. Even if the corticosteroid-dependent patient fails to show symptomatic improvement following cromolyn sodium inhalation solution administration, the potential to reduce corticosteroids may nonetheless be present. Thus, gradual tapering of corticosteroid dosage may be attempted. It is important that the dose be reduced slowly, maintaining close supervision of the patient to avoid an exacerbation of asthma.
It should be borne in mind that prolonged corticosteroid therapy frequently causes an impairment in the activity of the hypothalamic-pituitary-adrenal axis and a reduction in the size of the adrenal cortex. A potentially critical degree of impairment or insufficiency may persist asymptomatically for some time even after gradual discontinuation of adrenocortical steroids. Therefore, if a patient is subjected to significant stress, such as a severe asthmatic attack, surgery, trauma or severe illness while being treated or within one year (occasionally up to two years) after corticosteroid treatment has been terminated, consideration should be given to reinstituting corticosteroid therapy. When respiratory function is impaired, as may occur in severe exacerbation of asthma, a temporary increase in the amount of corticosteroids may be required to regain control of the patient's asthma.
It is particularly important that great care be exercised if, for any reason, cromolyn sodium inhalation solution is withdrawn in cases where its use has permitted a reduction in the maintenance dose of corticosteroids. In such cases, continued close supervision of the patient is essential since there may be sudden reappearance of severe manifestations of asthma which will require immediate therapy and possible reintroduction of corticosteroids.
Contraindications
Cromolyn sodium inhalation solution is contraindicated in those patients who have shown hypersensitivity to cromolyn sodium.
Adverse Reactions
Clinical experience with the use of cromolyn sodium suggests that adverse reactions are rare events. The following adverse reactions have been associated with cromolyn sodium: cough, nasal congestion, nausea, sneezing, and wheezing.
Other reactions have been reported in clinical trials; however, a causal relationship could not be established: drowsiness, nasal itching, nose bleed, nose burning, serum sickness, and stomachache.
In addition, adverse reactions have been reported with cromolyn sodium for inhalation capsules. The most common side effects are associated with inhalation of the powder and include transient cough (1 in 5 patients) and mild wheezing (1 in 25 patients). These effects rarely require treatment or discontinuation of the drug.
Information on the incidence of adverse reactions to cromolyn sodium for inhalation capsules has been derived from U.S. postmarketing surveillance experience. The following adverse reactions attributed to cromolyn sodium, based upon recurrence following readministration, have been reported in less than 1 in 10,000 patients: laryngeal edema, swollen parotid gland, angioedema, bronchospasm, joint swelling and pain, dizziness, dysuria and urinary frequency, nausea, cough, wheezing, headache, nasal congestion, rash, urticaria and lacrimation.
Other adverse reactions have been reported in less than 1 in 100,000 patients, and it is unclear whether these are attributable to the drug: anaphylaxis, nephrosis, periarteritic vasculitis, pericarditis, peripheral neuritis, pulmonary infiltrates with eosinophilia, polymyositis, exfoliative dermatitis, hemoptysis, anemia, myalgia, hoarseness, photodermatitis, and vertigo.
Call your doctor for medical advice about side effects. You may report side effects to Ailex Pharmaceuticals, LLC at 1-888-514-4727 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Description
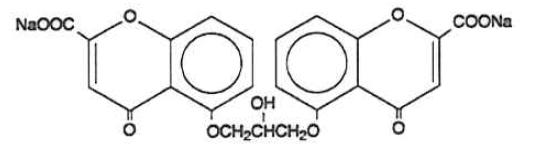
The active ingredient of cromolyn sodium inhalation solution USP is cromolyn sodium, USP. It is an inhaled anti-inflammatory agent for the preventive management of asthma. Cromolyn sodium, USP is chemically designated as disodium 5,5'- [(2-hydroxytrimethylene)dioxy] bis [4-oxo-4H-1-benzopyran-2-carboxylate]. The molecular formula is C23H14Na2O11; the molecular weight is 512.34. Cromolyn sodium, USP is a water-soluble, odorless, white, hydrated crystalline powder. It is tasteless at first, but leaves a slightly bitter aftertaste. Cromolyn sodium inhalation solution USP is clear, colorless to pale yellow, sterile and has a target pH of 5.5.
The structural formula is:
Each 2 mL vial for oral inhalation use only contains 20 mg cromolyn sodium, USP in water for injection, USP.
Clinical Pharmacology
In vitro and in vivo animal studies have shown that cromolyn sodium inhibits sensitized mast cell degranulation which occurs after exposure to specific antigens. Cromolyn sodium acts by inhibiting the release of mediators from mast cells. Studies show that cromolyn sodium indirectly blocks calcium ions from entering the mast cell, thereby preventing mediator release.
Cromolyn sodium inhibits both the immediate and non-immediate bronchoconstrictive reactions to inhaled antigen. Cromolyn sodium also attenuates bronchospasm caused by exercise, toluene diisocyanate, aspirin, cold air, sulfur dioxide, and environmental pollutants.
Cromolyn sodium has no intrinsic bronchodilator or antihistamine activity.
After administration by inhalation, approximately 8% of the total cromolyn sodium dose administered is absorbed and rapidly excreted unchanged, approximately equally divided between urine and bile. The remainder of the dose is either exhaled or deposited in the oropharynx, swallowed and excreted via the alimentary tract.
How Supplied
Cromolyn Sodium Inhalation Solution USP Unit-Dose 2 mL Vial is supplied as a colorless to pale yellow solution containing 20 mg cromolyn sodium, USP, in water for injection, USP, with 30 vials per foil pouch in a carton as listed below.
60 vials per carton (NDC 76204-028-60).
Each vial is made from a low density polyethylene (LDPE) resin.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Do not use if solution is discolored or contains a precipitate.
Retain in foil pouch until time of use.
PROTECT FROM LIGHT.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Manufactured for: Ritedose Pharmaceuticals, LLC Columbia, SC 29203 U.S.A.
Manufactured by:
The Ritedose Corporation
Columbia, SC 29203 U.S.A.
Instructions for the Use of Cromolyn Sodium Inhalation Solution, USP
An aqueous solution for nebulization
NOT FOR INJECTION
For best results, follow these instructions exactly and observe Care and Storage directions.
METHOD OF ADMINISTRATION
Cromolyn sodium inhalation solution is recommended for use in a power driven nebulizer operated at an airflow rate of 6-8 liters per minute and equipped with a suitable face mask. Hand-operated nebulizers are not suitable for the administration of cromolyn sodium inhalation solution. Your doctor will advise on the choice of a suitable nebulizer and how it should be used. Do not use any appliance without consulting your doctor.
Drug stability and safety of cromolyn sodium inhalation solution when mixed with other drugs in a nebulizer have not been established.
DOSAGE
Nebulization should be carried out four times a day at regular intervals, or as directed by your doctor. Use the contents of a fresh vial each time.
INHALATION
Once the nebulizer has been assembled and contains cromolyn sodium inhalation solution, hold the mask close to the patient’s face and switch on the device. The patient should breathe in through the mouth and out through the nose in a normal, relaxed manner. Nebulization should take approximately five to ten minutes.
1. Remove a single unit-dose vial from strip (Figure 1)
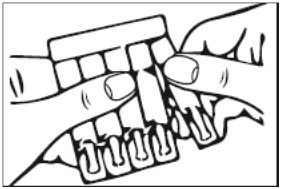
Figure 1
2. Open the unit-dose vial by twisting off the tabbed top section (Figure 2).
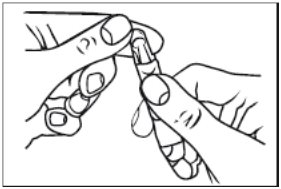
Figure 2
3. Squeeze the contents of the unit-dose vial into the solution container of your nebulizer (Figure 3). Discard the empty unit-dose vial.
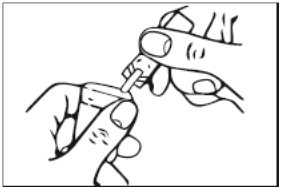
Figure 3