Get your patient on Cyclopentolate Hydrochloride - Cyclopentolate Hydrochloride solution/ Drops (Cyclopentolate Hydrochloride)
Cyclopentolate Hydrochloride - Cyclopentolate Hydrochloride solution/ Drops prescribing information
INDICATIONS AND USAGE
Cyclopentolate hydrochloride ophthalmic solution is used to produce mydriasis and cycloplegia.
DOSAGE AND ADMINISTRATION
Adults: Instill one or two drops of 1% solution in the eye which may be repeated in five to ten minutes if necessary. Complete recovery usually occurs in 24 hours. Complete recovery from mydriasis in some individuals may require several days.
Children: Instill one or two drops of 1% solution in the eye, which may be repeated five to ten minutes later by a second application of 1% solution if necessary.
Individuals with heavily pigmented irides may require higher strengths.
CONTRAINDICATIONS
Hypersensitive to any component of this preparation.
ADVERSE REACTIONS
Ocular: Increased intraocular pressure, burning, photophobia, blurred vision, irritation, hyperemia, conjunctivitis, blepharoconjunctivitis, punctate keratitis, synechiae have been reported.
Non-ocular: Use of cyclopentolate has been associated with psychotic reactions and behavioral disturbances, usually in children, especially with 2% concentration. These disturbances include ataxia, incoherent speech, restlessness, hallucinations, hyperactivity, seizures, disorientation as to time and place, and failure to recognize people. This drug produces reactions similar to those of other anticholinergic drugs, but the CNS manifestations as noted above are more common. Other toxic manifestations of anticholinergic drugs are skin rash, abdominal distention in infants, necrotizing enterocolitis (in preterm infants), unusual drowsiness, tachycardia, hyperpyrexia, vasodilation, urinary retention, diminished gastrointestinal motility and decreased secretion in salivary and sweat glands, pharynx, bronchi and nasal passages. Severe manifestations of toxicity include coma, medullary paralysis and death.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions:
Cyclopentolate may interfere with the ocular anti-hypertensive action of carbachol, pilocarpine, or ophthalmic cholinesterase inhibitors.
DESCRIPTION
Cyclopentolate hydrochloride ophthalmic solution USP, 1% is an anticholinergic prepared as a sterile, borate buffered, solution for topical ocular use. The active ingredient is represented by the structural formula:
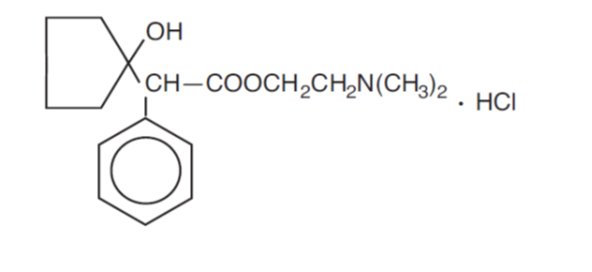
Molecular Weight = 327.85 Molecular Formula: C 17 H 25 NO 3 • HCl
Established name: cyclopentolate hydrochloride Chemical name: 2-(Dimethylamino) ethyl 1-hydroxy-α-phenylcyclopentaneacetate hydrochloride
Each mL contains: Active: cyclopentolate hydrochloride, 10 mg (1%)
Inactives: boric acid, edetate disodium, potassium chloride, purified water. Sodium carbonate and/or hydrochloric acid (to adjust pH). The pH range is between 3.0 and 5.5.
Preservative: benzalkonium chloride 0.01%.
CLINICAL PHARMACOLOGY
This anticholinergic preparation blocks the responses of the sphincter muscle of the iris and the accommodative muscle of the ciliary body to cholinergic stimulation, producing pupillary dilation (mydriasis) and paralysis of accommodation (cycloplegia). It acts rapidly, but has a shorter duration than atropine. Maximal cycloplegia occurs within 25 to 75 minutes after instillation. Complete recovery of accommodation usually takes 6 to 24 hours. Complete recovery from mydriasis in some individuals may require several days. Heavily pigmented irides may require more doses than lightly pigmented irides.
HOW SUPPLIED
Cyclopentolate hydrochloride ophthalmic solution USP, 1% is available in multidose plastic squeeze bottles with a controlled drop tip and red cap in the following sizes:
2 mL bottle - NDC 24208-735-01 15 mL bottle - NDC 24208-735-06
Storage:
Store between 15°C to 25°C (59°F to 77°F).

KEEP OUT OF REACH OF CHILDREN.
Distributed by: Bausch & Lomb Americas Inc. Bridgewater, NJ 08807 USA Manufactured by: Bausch & Lomb Incorporated Tampa, FL 33637 USA © 2022 Bausch & Lomb Incorporated or its affiliates Rev. 09/2022
9114607 (Folded) 9114707 (Flat)