Get your patient on Cyproheptadine Hydrochloride - Cyproheptadine Hydrochloride tablet (Cyproheptadine Hydrochloride)
Cyproheptadine Hydrochloride - Cyproheptadine Hydrochloride tablet prescribing information
INDICATIONS AND USAGE
Perennial and seasonal allergic rhinitis Vasomotor rhinitis Allergic conjunctivitis due to inhalant allergens and foods Mild, uncomplicated allergic skin manifestations of urticaria and angioedema. Amelioration of allergic reactions to blood or plasma Cold urticaria Dermatographism
As therapy for anaphylactic reactions adjunctive to epinephrine and other standard measures after the acute manifestations have been controlled.
DOSAGE AND ADMINISTRATION
DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND THE RESPONSE OF THE PATIENT.
Each tablet contains 4 mg of cyproheptadine hydrochloride.
Pediatric Patients
Age 2 to 6 years
The total daily dosage for pediatric patients may be calculated on the basis of body weight or body area using approximately 0.25 mg/kg/day or 8 mg per square meter of body surface (8 mg/m 2 ).
The usual dose is 2 mg (½ tablet) two or three times a day, adjusted as necessary to the size and response of the patient. The dose is not to exceed 12 mg a day.
Age 7 to 14 years
The usual dose is 4 mg (1 tablet) two or three times a day adjusted as necessary to the size and response of the patient. The dose is not to exceed 16 mg a day.
Adults
The total daily dose for adults should not exceed 0.5 mg/kg/day. The therapeutic range is 4 to 20 mg a day, with the majority of patients requiring 12 to 16 mg a day. An occasional patient may require as much as 32 mg a day for adequate relief. It is suggested that dosage be initiated with 4 mg (1 tablet) three times a day and adjusted according to the size and response of the patient.
CONTRAINDICATIONS
Newborn or Premature Infants
This drug should not be used in newborn or premature infants.
Nursing Mothers
Because of the higher risk of antihistamines for infants generally and for newborns and prematures in particular, antihistamine therapy is contraindicated in nursing mothers.
Other Conditions
Hypersensitivity to cyproheptadine and other drugs of similar chemical structure. Monoamine oxidase inhibitor therapy (See DRUG INTERACTIONS .) Angle-closure glaucoma Stenosing peptic ulcer Symptomatic prostatic hypertrophy Bladder neck obstruction Pyloroduodenal obstruction Elderly, debilitated patients
ADVERSE REACTIONS
Adverse reactions which have been reported with the use of antihistamines are as follows:
Central Nervous System
Sedation and sleepiness (often transient), dizziness, disturbed coordination, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, paresthesias, neuritis, convulsions, euphoria, hallucinations, hysteria, faintness.
Integumentary
Allergic manifestation of rash and edema, excessive perspiration, urticaria, photosensitivity.
Special Senses
Acute labyrinthitis, blurred vision, diplopia, vertigo, tinnitus.
Cardiovascular
Hypotension, palpitation, tachycardia, extrasystoles, anaphylactic shock.
Hematologic
Hemolytic anemia, leukopenia, agranulocytosis, thrombocytopenia.
Digestive System
Cholestasis, hepatic failure, hepatitis, hepatic function abnormality, dryness of mouth, epigastric distress, anorexia, nausea, vomiting, diarrhea, constipation, jaundice.
Genitourinary
Urinary frequency, difficult urination, urinary retention, early menses.
Respiratory
Dryness of nose and throat, thickening of bronchial secretions, tightness of chest and wheezing, nasal stuffiness.
Miscellaneous
Fatigue, chills, headache, increased appetite/weight gain.
Drug Interactions
MAO inhibitors prolong and intensify the anticholinergic effects of antihistamines.
Antihistamines may have additive effects with alcohol and other CNS depressants, e.g., hypnotics, sedatives, tranquilizers, antianxiety agents.
DESCRIPTION
Cyproheptadine HCl, is an antihistaminic and antiserotonergic agent. Cyproheptadine hydrochloride, USP is a white to slightly yellowish crystalline solid, with a molecular weight of 350.89, which is soluble in water, freely soluble in methanol, sparingly soluble in ethanol, soluble in chloroform, and practically insoluble in ether. It is the sesquihydrate 4-(5H-dibenzo[a,d]cyclohepten-5-ylidene)-1-methylpiperidine hydrochloride. The molecular formula of the anhydrous salt is C 21 H 21 N•HCl and the structural formula of the anhydrous salt is as follows:
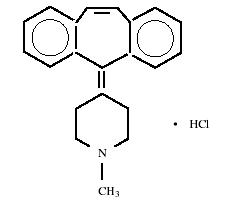
C 21 H 21 N• HCl M.W. 350.89
Cyproheptadine hydrochloride, USP is available for oral administration in 4 mg tablets. Each tablet contains: Inactives: Lactose Monohydrate, Microcrystalline Cellulose, Sodium Starch Glycolate and Magnesium Stearate.
CLINICAL PHARMACOLOGY
Cyproheptadine is a serotonin and histamine antagonist with anticholinergic and sedative effects. Antiserotonin and antihistamine drugs appear to compete with serotonin and histamine, respectively, for receptor sites.
Pharmacokinetics and Metabolism
After a single 4 mg oral dose of 14 C-labelled cyproheptadine HCl in normal subjects, given as tablets, 2 to 20% of the radioactivity was excreted in the stools. Only about 34% of the stool radioactivity was unchanged drug, corresponding to less than 5.7% of the dose. At least 40% of the administered radioactivity was excreted in the urine. No detectable amounts of unchanged drug were present in the urine of patients on chronic 12 to 20 mg daily doses. The principle metabolite found in human urine has been identified as a quaternary ammonium glucuronide conjugate of cyproheptadine. Elimination is diminished in renal insufficiency.
HOW SUPPLIED
Cyproheptadine hydrochloride tablets USP, 4 mg are available as white to off-white color, round flat face tablet with debossed “B137” on one side and scored on the other, containing 4 mg of cyproheptadine HCl packaged in HDPE bottles of 100 and 1000 tablets.
PHARMACIST: Dispense in a well-closed container as defined in the USP, with a child-resistant closure (as required).
Bottles of 100 tablets: NDC 76385- 130 -01 Bottles of 1000 tablets: NDC 76385- 130 -10