Get your patient on Dapiprazole - Dapiprazole (Dapiprazole)
Dapiprazole - Dapiprazole prescribing information
INDICATIONS AND USAGE:
Dapiprazole hydrochloride ophthalmic solution is indicated in the treatment of iatrogenically induced mydriasis produced by adrenergic (phenylephrine) or parasympatholytic (tropicamide) agents. Dapiprazole hydrochloride ophthalmic solution is not indicated for the reduction of intraocular pressure or in the treatment of open angle glaucoma.
DOSAGE AND ADMINISTRATION:
Two drops followed 5 minutes later by an additional 2 drops applied topically to the conjunctiva of each eye should be administered after the ophthalmic examination to reverse the diagnostic mydriasis. Dapiprazole hydrochloride ophthalmic solution should not be used in the same patient more frequently than once per week.
Directions for Preparing Eyedrops:
- Use aseptic technique.
- Tear off aluminum seals, remove and discard rubber plugs from both drug and diluent vials
- Pour diluent into drug vial.
- Remove dropper assembly from its sterile wrapping and attach to the drug vial.
- Shake container for several minutes to ensure mixing.
CONTRAINDICATIONS:
Miotics are contraindicated where constriction is undesirable; such as acute iritis, and in those subjects showing hypersensitivity to any component of this preparation.
ADVERSE REACTIONS:
In controlled studies the most frequent reaction to dapiprazole was conjunctival injection lasting 20 minutes in over 80% of patients. Burning on instillation of dapiprazole hydrochloride ophthalmic solution was reported in approximately half of all patients. Reactions occurring in 10% to 40% of patients included ptosis, lid erythema, lid edema, chemosis, itching, punctate keratitis, corneal edema, browache, photophobia and headaches. Other reactions reported less frequently included dryness of eyes, tearing and blurring of vision.
DESCRIPTION:
For ophthalmic use only.
Dapiprazole hydrochloride is an alpha-adrenergic blocking agent.
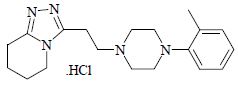
Dapiprazole hydrochloride is 5,6,7,8-tetrahydro-3-[2-(4- o .tolyl-1-piperazinyl)ethyl]- s -triazolo[4,3-a]pyridine hydrochloride.
Dapiprazole hydrochloride has the empirical formula C 19 H 27 N 5 • HCl and a molecular weight of 361.93.
The structural formula for dapiprazole hydrochloride is:
Dapiprazole hydrochloride is a sterile, white, lyophilized powder soluble in water.
Dapiprazole hydrochloride ophthalmic solution, 0.5% is a clear, colorless, slightly viscous solution for topical application. Each mL (when reconstituted as directed) contains 5 mg of dapiprazole hydrochloride as the active ingredient.
The reconstituted solution has a pH of approximately 6.6 and an osmolarity of approximately 415 mOsm.
The inactive ingredients include: mannitol (2%), sodium chloride, hydroxypropyl methylcellulose (0.4%), edetate sodium (0.01%), sodium phosphate dibasic, sodium phosphate monobasic, water for injection, and benzalkonium chloride (0.01%) as a preservative.
Dapiprazole hydrochloride ophthalmic solution, 0.5% is supplied in a kit consisting of one vial of dapiprazole hydrochloride (25 mg), one vial of diluent (5 mL) and one dropper for dispensing.
CLINICAL PHARMACOLOGY:
Dapiprazole hydrochloride ophthalmic solution acts through blocking the alpha-adrenergic receptors in smooth muscle. Dapiprazole hydrochloride ophthalmic solution produces miosis through an effect on the dilator muscle of the iris.
Dapiprazole hydrochloride ophthalmic solution does not have any significant activity on ciliary muscle contraction and, therefore does not induce a significant change in the anterior chamber depth or the thickness of the lens.
Dapiprazole hydrochloride ophthalmic solution has demonstrated safe and rapid reversal of mydriasis produced by phenylephrine and to a lesser degree tropicamide. In patients with decreased accommodative amplitude due to treatment with tropicamide, dapiprazole hydrochloride ophthalmic solution partially restores the accommodative amplitude. This activity is not only due to its miotic effect but also to a direct effect on accommodation.
Eye color affects the rate of pupillary constriction. In individuals with brown irides, the rate of pupillary constriction may be slightly slower than in individuals with blue or green irides. Eye color does not appear to affect the final pupil size.
Dapiprazole hydrochloride ophthalmic solution does not significantly alter intraocular pressure in normotensive eyes or in eyes with elevated intraocular pressure.
HOW SUPPLIED:
Dapiprazole hydrochloride ophthalmic solution, 0.5% - Sterile is supplied as an outer package (NDC 53020-265-01) containing:
NDC 53020-255-01 single vial of dapiprazole hydrochloride (25 mg) lyophilized powder
NDC 53020-245-01 single vial of diluent (5 mL)
Single dropper for dispensing