Dasetta 1/35- Norethindrone And Ethinyl Estradiol prescribing information
WARING: CARDIOVASCULAR RISK ASSOCIATED WITH SMOKING
Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, combination oral contraceptives, including DASETTA TM 1/35, should not be used by women who are over 35 years of age and smoke.
INDICATIONS AND USAGE
DASETTA ™ 1/35 Tablets are indicated for the prevention of pregnancy in women who elect to use this product as a method of contraception.
Oral contraceptives are highly effective. Table 1 lists the typical accidental pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization, the IUD, and the NORPLANT ® System depends upon the reliability with which they are used. Correct and consistent use of methods can result in lower failure rates.
Adapted from Hatcher et al, 1998, Ref. #1. | |||
Emergency Contraceptive Pills: Treatment initiated within 72 hours after unprotected intercourse reduces the risk of pregnancy by at least 75%. 9 | |||
Lactational Amenorrhea Method: LAM is highly effective, temporary method of contraception. 1 0 | |||
Source: Trussell J, Contraceptive efficacy. In Hatcher RA, Trussell J, Stewart F, Cates W, Stewart GK, Kowal D, Guest F, Contraceptive Technology: Seventeenth Revised Edition. New York NY: Irvington Publishers, 1998. | |||
1 Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason. | |||
2 Among couples who initiate use of a method (not necessarily for the first time) and who use it perfectly (both consistently and correctly), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason. | |||
3 Among couples attempting to avoid pregnancy, the percentage who continue to use a method for one year. | |||
4 The percents becoming pregnant in columns (2) and (3) are based on data from populations where contraception is not used and from women who cease using contraception in order to become pregnant. Among such populations, about 89% become pregnant within one year. This estimate was lowered slightly (to 85%) to represent the percent who would become pregnant within one year among women now relying on reversible methods of contraception if they abandoned contraception altogether. | |||
5 Foams, creams, gels, vaginal suppositories, and vaginal film. | |||
6 Cervical mucus (ovulation) method supplemented by calendar in the pre-ovulatory and basal body temperature in the post-ovulatory phases. | |||
7 With spermicidal cream or jelly. | |||
8 Without spermicides. | |||
9 The treatment schedule is one dose within 72 hours after unprotected intercourse, and a second dose 12 hours after the first dose. The Food and Drug Administration has declared the following brands of oral contraceptives to be safe and effective for emergency contraception: Ovral ® (1 dose is 2 white pills), Alesse ® (1 dose is 5 pink pills), Nordette ® or Levlen ® (1 dose is 2 light-orange pills), Lo/Ovral ® (1 dose is 4 white pills), Triphasil ® or Tri-Levlen ® (1 dose is 4 yellow pills). | |||
1 0 However, to maintain effective protection against pregnancy, another method of contraception must be used as soon as menstruation resumes, the frequency or duration of breastfeeds is reduced, bottle feeds are introduced, or the baby reaches six months of age. | |||
| Table 1: Percentage of Women Experiencing an Unintended Pregnancy During the First Year of Typical Use and the First Year of Perfect Use of Contraception and the Percentage Continuing Use at the End of the First Year. United States. | |||
| % of Women Experiencing an Unintended Pregnancy within the First Year of Use | % of Women Continuing Use at One Year 3 | ||
| Method | Typical Use 1 | Perfect Use 2 | |
| (1) | (2) | (3) | (4) |
| Chance 4 | 85 | 85 | |
| Spermicides 5 | 26 | 6 | 40 |
| Periodic abstinence | 25 | 63 | |
| Calendar | 9 | ||
| Ovulation Method | 3 | ||
| Sympto-Thermal 6 | 2 | ||
| Post-Ovulation | 1 | ||
| Cap 7 | |||
| Parous Women | 40 | 26 | 42 |
| Nulliparous Women | 20 | 9 | 56 |
| Sponge | |||
| Parous Women | 40 | 20 | 42 |
| Nulliparous Women | 20 | 9 | 56 |
| Diaphragm 7 | 20 | 6 | 56 |
| Withdrawal | 19 | 4 | |
| Condom 8 | |||
| Female (Reality ® ) | 21 | 5 | 56 |
| Male | 14 | 3 | 61 |
| Pill | 5 | 71 | |
| Progestin Only | 0.5 | ||
| Combined | 0.1 | ||
| IUD | |||
| Progesterone T | 2.0 | 1.5 | 81 |
| Copper T380A | 0.8 | 0.6 | 78 |
| LNg 20 | 0.1 | 0.1 | 81 |
| Depo-Provera ® | 0.3 | 0.3 | 70 |
| Norplant ® and Norplant-2 ® | 0.05 | 0.05 | 88 |
| Female Sterilization | 0.5 | 0.5 | 100 |
| Male Sterilization | 0.15 | 0.10 | 100 |
DASETTA ™ 1/35 has not been studied for and is not indicated for use in emergency contraception.
DOSAGE AND ADMINISTRATION
To achieve maximum contraceptive effectiveness, DASETTA™ 1/35 Tablets must be taken exactly as directed and at intervals not exceeding 24 hours. DASETTA™ 1/35 Tablets are available in a compact blister card which is preset for a Sunday Start. Day 1 Start stickers are also provided.
Sunday Start
When taking DASETTA™ 1/35, the first "active" tablet should be taken on the first Sunday after menstruation begins. If period begins on Sunday, the first "active" tablet should be taken that day. Take one active tablet daily for 21 days followed by one white "reminder" tablet daily for 7 days. After 28 tablets have been taken, a new course is started the next day (Sunday). For the first cycle of a Sunday Start regimen, another method of contraception should be used until after the first 7 consecutive days of administration.
If the patient misses one (1) "active" tablet in Weeks 1, 2, or 3, the tablet should be taken as soon as she remembers. If the patient misses two (2) "active" tablets in Week 1 or Week 2, the patient should take two (2) tablets the day she remembers and two (2) tablets the next day; and then continue taking one (1) tablet a day until she finishes the pack. The patient should be instructed to use a back-up method of birth control such as condoms or spermicide if she has sex in the seven (7) days after missing pills. If the patient misses two (2) "active" tablets in the third week or misses three (3) or more "active" tablets in a row, the patient should continue taking one tablet every day until Sunday. On Sunday the patient should throw out the rest of the pack and start a new pack that same day. The patient should be instructed to use a back-up method of birth control if she has sex in the seven (7) days after missing pills.
Complete instructions to facilitate patient counseling on proper pill usage may be found in the Detailed Patient Labeling ("How to Take the Pill" section).
Day 1 Start
The dosage of DASETTA™ 1/35, for the initial cycle of therapy is one "active" tablet administered daily from the 1st through the 21st day of the menstrual cycle, counting the first day of menstrual flow as "Day 1" followed by one white "reminder" tablet daily for 7 days. Tablets are taken without interruption for 28 days. After 28 tablets have been taken, a new course is started the next day.
If the patient misses one (1) "active" tablet in Weeks 1, 2, or 3, the tablet should be taken as soon as she remembers. If the patient misses two (2) "active" tablets in Week 1 or Week 2, the patient should take two (2) tablets the day she remembers and two (2) tablets the next day; and then continue taking one (1) tablet a day until she finishes the pack. The patient should be instructed to use a back-up method of birth control such as condoms or spermicide if she has sex in the seven (7) days after missing pills. If the patient misses two (2) "active" tablets in the third week or misses three (3) or more "active" tablets in a row, the patient should throw out the rest of the pack and start a new pack that same day. The patient should be instructed to use a back-up method of birth control if she has sex in the seven (7) days after missing pills.
Complete instructions to facilitate patient counseling on proper pill usage may be found in the Detailed Patient Labeling ("How to Take the Pill" section).
The use of DASETTA™ 1/35 for contraception may be initiated 4 weeks postpartum in women who elect not to breast feed. When the tablets are administered during the postpartum period, the increased risk of thromboembolic disease associated with the postpartum period must be considered. (See CONTRAINDICATIONS and WARNINGS concerning thromboembolic disease. See also PRECAUTIONS for " Nursing Mothers .") The possibility of ovulation and conception prior to initiation of medication should be considered.
(See Discussion of Dose-Related Risk of Vascular Disease from Oral Contraceptives .)
ADDITIONAL INSTRUCTIONS
Breakthrough bleeding, spotting, and amenorrhea are frequent reasons for patients discontinuing oral contraceptives. In breakthrough bleeding, as in all cases of irregular bleeding from the vagina, nonfunctional causes should be borne in mind. In undiagnosed persistent or recurrent abnormal bleeding from the vagina, adequate diagnostic measures are indicated to rule out pregnancy or malignancy. If pathology has been excluded, time or a change to another formulation may solve the problem. Changing to an oral contraceptive with a higher estrogen content, while potentially useful in minimizing menstrual irregularity, should be done only if necessary since this may increase the risk of thromboembolic disease.
Use of oral contraceptives in the event of a missed menstrual period:
- If the patient has not adhered to the prescribed schedule, the possibility of pregnancy should be considered at the time of the first missed period and oral contraceptive use should be discontinued if pregnancy is confirmed.
- If the patient has adhered to the prescribed regimen and misses two consecutiveperiods, pregnancy should be ruled out.
CONTRAINDICATIONS
Oral contraceptives should not be used in women who currently have the following conditions:
- Thrombophlebitis or thromboembolic disorders
- A past history of deep vein thrombophlebitis or thromboembolic disorders
- Known thrombophilic conditions
- Cerebral vascular or coronary artery disease (current or history)
- Valvular heart disease with complications
- Persistent blood pressure values of 160 mm Hg systolic or 100 mg Hg diastolic 96
- Diabetes with vascular involvement
- Headaches with focal neurological symptoms
- Major surgery with prolonged immobilization
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive
- Carcinoma of the endometrium or other known or suspected estrogen-dependent neoplasia
- Undiagnosed abnormal genital bleeding
- Cholestatic jaundice of pregnancy or jaundice with prior pill use
- Acute or chronic hepatocellular disease with abnormal liver function
- Hepatic adenomas or carcinomas
- Known or suspected pregnancy
- Hypersensitivity to any component of this product
- Are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations (see WARNINGS , Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment )
ADVERSE REACTIONS
Post Marketing Experience:
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 2).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 2). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
Figure 2. Risk of Breast Cancer with Combined Oral Contraceptive Use
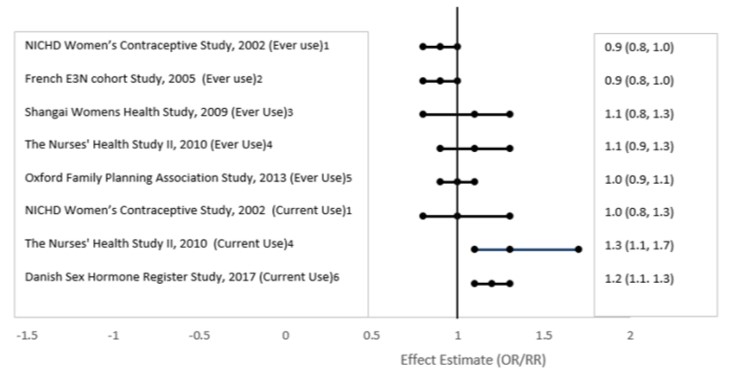
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs.
An increased risk of the following serious adverse reactions has been associated with the use of oral contraceptives (See WARNINGS Section).
●Thrombophlebitis and venous thrombosis with or without embolism
●Arterial thromboembolism
●Pulmonary embolism
●Myocardial infarction
●Cerebral hemorrhage
●Cerebral thrombosis
●Hypertension
●Gallbladder disease
●Hepatic adenomas or benign liver tumors
There is evidence of an association between the following conditions and the use of oral contraceptives:
●Mesenteric thrombosis
●Retinal thrombosis
The following adverse reactions have been reported in patients receiving oral contraceptives and are believed to be drug-related:
●Nausea
●Vomiting
●Gastrointestinal symptoms (such as abdominal cramps and bloating)
●Breakthrough bleeding
●Spotting
●Change in menstrual flow
●Amenorrhea
●Temporary infertility after discontinuation of treatment
●Edema
●Melasma which may persist
●Breast changes: tenderness, enlargement, secretion
●Change in weight (increase or decrease)
●Change in cervical erosion and secretion
●Diminution in lactation when given immediately postpartum
●Cholestatic jaundice
●Migraine
●Allergic reaction, including rash, urticaria, angioedema
●Mental depression
●Reduced tolerance to carbohydrates
●Vaginal candidiasis
●Change in corneal curvature (steepening)
●Intolerance to contact lenses
The following adverse reactions have been reported in users of oral contraceptives and a causal association has been neither confirmed nor refuted:
●Pre-menstrual syndrome
●Cataracts
●Changes in appetite
●Cystitis-like syndrome
●Headache
●Nervousness
●Dizziness
●Hirsutism
●Loss of scalp hair
●Erythema multiforme
●Erythema nodosum
●Hemorrhagic eruption
●Vaginitis
●Porphyria
●Impaired renal function
●Hemolytic uremic syndrome
●Acne
●Changes in libido
●Colitis
●Budd-Chiari Syndrome
The following adverse reactions were also reported in clinical trials or during post-marketing experience: Gastrointestinal Disorders: diarrhea, pancreatitis; Musculoskeletal and Connective Tissue Disorders: muscle spasms, back pain; Reproductive System and Breast Disorders: vulvovaginal pruritus, pelvic pain, dysmenorrhea, vulvovaginal dryness; Psychiatric Disorders: anxiety, mood swings, mood altered; Skin and Subcutaneous Tissue Disorders: pruritus, photosensitivity reaction; General Disorders and Administration Site Conditions: edema peripheral, fatigue, irritability, asthenia, malaise; Neoplasms Benign, Malignant, and Unspecified (Including Cysts and Polyps): breast cancer, breast mass, breast neoplasm, cervix carcinoma; Immune System Disorders: anaphylactic/anaphylactoid reaction; Hepatobiliary Disorders: hepatitis, cholelithiasis.
CLINICAL PHARMACOLOGY
COMBINED ORAL CONTRACEPTIVES
Combined oral contraceptives act by suppression of gonadotropins. Although the primary mechanism of this action is inhibition of ovulation, other alterations include changes in the cervical mucus (which increase the difficulty of sperm entry into the uterus) and the endometrium (which reduce the likelihood of implantation).
HOW SUPPLIED
DASETTA™ 1/35 Tablets are available in a compact blister card (NDC 16714-348-01) containing 28 tablets, as follows: 21 orange, biconvex round tablets with "D3" debossed on one side (1 mg norethindrone and 0.035 mg ethinyl estradiol) and 7 white, biconvex round tablets with "P" debossed on one side and the " N " on the other side containing inert ingredients.
DASETTA™ 1/35 Tablets are available in the following:
Carton of 1 NDC 16714-348-02
Carton of 3 NDC 16714-348-03
Carton of 6 NDC 16714-348-04
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature].
Rx Only