Get your patient on Dermacinrx Clorhexacin (Mupirocin Ointment)
Dermacinrx Clorhexacin prescribing information
INDICATIONS AND USAGE
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to: S. aureus and S. pyogenes .
DOSAGE AND ADMINISTRATION
A small amount of Mupirocin Ointment USP, 2% should be applied to the affected area 3 times daily. The area treated may be covered with a gauze dressing if desired. Patients not showing a clinical response within 3 to 5 days should be re-evaluated.
CONTRAINDICATIONS
This drug is contraindicated in individuals with a history of sensitivity reactions to any of its components.
ADVERSE REACTIONS
The following local adverse reactions have been reported in connection with the use of mupirocin ointment USP, 2%: Burning, stinging, or pain in 1.5% of patients; itching in 1% of patients; rash, nausea, erythema, dry skin, tenderness, swelling, contact dermatitis, and increased exudate in less than 1% of patients. Systemic reactions to mupirocin ointment USP, 2% have occurred rarely.
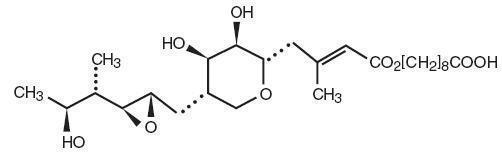
DESCRIPTION
Each gram of Mupirocin Ointment USP, 2% contains 20 mg mupirocin in a bland water miscible ointment base (polyethylene glycol ointment, NF) consisting of polyethylene glycol 400 and polyethylene glycol 3350. Mupirocin is a naturally occurring antibiotic. The chemical name is ( E )-(2 S ,3 R ,4 R ,5 S )-5-[(2 S ,3 S ,4 S ,5 S )-2,3-Epoxy-5-hydroxy-4-methylhexyl]tetrahydro-3,4-dihydroxy-β-methyl-2H-pyran-2-crotonic acid, ester with 9-hydroxynonanoic acid. The molecular formula of mupirocin is C 26 H 44 O 9 and the molecular weight is 500.62. The chemical structure is:
CLINICAL PHARMACOLOGY
Application of 14 C-labeled mupirocin ointment to the lower arm of normal male subjects followed by occlusion for 24 hours showed no measurable systemic absorption (<1.1 nanogram mupirocin per milliliter of whole blood). Measurable radioactivity was present in the stratum corneum of these subjects 72 hours after application.
Following intravenous or oral administration, mupirocin is rapidly metabolized. The principal metabolite, monic acid, is eliminated by renal excretion, and demonstrates no antibacterial activity. In a study conducted in 7 healthy adult male subjects, the elimination half-life after intravenous administration of mupirocin was 20 to 40 minutes for mupirocin and 30 to 80 minutes for monic acid. The pharmacokinetics of mupirocin has not been studied in individuals with renal insufficiency.
Microbiology - Mupirocin is an antibacterial agent produced by fermentation using the organism Pseudomonas fluorescens . It is active against a wide range of gram-positive bacteria including methicillin-resistant Staphylococcus aureus (MRSA). It is also active against certain gram-negative bacteria. Mupirocin inhibits bacterial protein synthesis by reversibly and specifically binding to bacterial isoleucyl transfer-RNA synthetase. Due to this unique mode of action, mupirocin demonstrates no in vitro cross-resistance with other classes of antimicrobial agents.
Resistance occurs rarely. However, when mupirocin resistance does occur, it appears to result from the production of a modified isoleucyl-tRNA synthetase. High-level plasmid-mediated resistance (MIC > 1024 mcg/mL) has been reported in some strains of S. aureus and coagulase-negative staphylococci.
Mupirocin is bactericidal at concentrations achieved by topical administration. However, the minimum bactericidal concentration (MBC) against relevant pathogens is generally 8-fold to 30-fold higher than the minimum inhibitory concentration (MIC). In addition, mupirocin is highly protein bound (>97%), and the effect of wound secretions on the MICs of mupirocin has not been determined.
Mupirocin has been shown to be active against most strains of S. aureus and Streptococcus pyogenes , both in vitro and in clinical studies (see INDICATIONS AND USAGE ). The following in vitro data are available, BUT THEIR CLINICAL SIGNIFICANCE IS UNKNOWN. Mupirocin is active against most strains of Staphylococcus epidermidis and Staphylococcus saprophyticus .
CLINICAL STUDIES
The efficacy of topical mupirocin ointment USP, 2% in impetigo was tested in two studies. In the first, patients with impetigo were randomized to receive either mupirocin ointment USP, 2% or vehicle placebo three times daily for 8 to 12 days. Clinical efficacy rates at end of therapy in the evaluable populations (adults and pediatric patients included) were 71% for mupirocin ointment USP, 2% (n=49) and 35% for vehicle placebo (n=51). Pathogen eradication rates in the evaluable populations were 94% for mupirocin ointment USP, 2% and 62% for vehicle placebo. There were no side effects reported in the group receiving mupirocin ointment USP, 2%.
In the second study, patients with impetigo were randomized to receive either mupirocin ointment USP, 2% three times daily or 30 to 40 mg/kg oral erythromycin ethylsuccinate per day (this was an unblinded study) for 8 days. There was a follow-up visit 1 week after treatment ended. Clinical efficacy rates at the follow-up visit in the evaluable populations (adults and pediatric patients included) were 93% for mupirocin ointment USP, 2% (n=29) and 78.5% for erythromycin (n=28). Pathogen eradication rates in the evaluable patient populations were 100% for both test groups. There were no side effects reported in the group receiving mupirocin ointment.
Pediatrics - There were 91 pediatric patients aged 2 months to 15 years in the first study described above. Clinical efficacy rates at end of therapy in the evaluable populations were 78% for mupirocin ointment USP, 2% (n=42) and 36% for vehicle placebo (n=49). In the second study described above, all patients were pediatric except two adults in the group receiving mupirocin ointment USP, 2%. The age range of the pediatric patients was 7 months to 13 years. The clinical efficacy rate for mupirocin ointment USP, 2% (n=27) was 96%, and for erythromycin it was unchanged (78.5%).
HOW SUPPLIED
Mupirocin Ointment USP, 2% is available as follows: 22 gram tube (NDC 45802-112-22)
Store at 20-25°C (68-77°F) [see USP Controlled Room Temperature].
•Bactroban Nasal ® is a registered trademark of GlaxoSmithKline.