Desipramine Hydrochloride - Desipramine Hydrochloride tablet, Film Coated prescribing information
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of desipramine hydrochloride tablets or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Desipramine hydrochloride tablets are not approved for use in pediatric patients (see WARNINGS: Clinical Worsening and Suicide Risk, PRECAUTIONS: Information for Patients, and PRECAUTIONS: Pediatric Use).
INDICATIONS AND USAGE
Desipramine hydrochloride tablets, USP are indicated for the treatment of depression.
DOSAGE AND ADMINISTRATION
Not recommended for use in children (see WARNINGS).
Lower dosages are recommended for elderly patients and adolescents. Lower dosages are also recommended for outpatients compared to hospitalized patients, who are closely supervised. Dosage should be initiated at a low level and increased according to clinical response and any evidence of intolerance. Following remission, maintenance medication may be required for a period of time and should be at the lowest dose that will maintain remission.
Usual Adult Dose
The usual adult dose is 100 mg to 200 mg per day. In more severely ill patients, dosage may be further increased gradually to 300 mg/day if necessary. Dosages above 300 mg/day are not recommended.
Dosage should be initiated at a lower level and increased according to tolerance and clinical response.
Treatment of patients requiring as much as 300 mg should generally be initiated in hospitals, where regular visits by the physician, skilled nursing care, and frequent electrocardiograms (ECGs) are available.
The best available evidence of impending toxicity from very high doses of desipramine hydrochloride tablets are prolongation of the QRS or QT intervals on the ECG. Prolongation of the PR interval is also significant, but less closely correlated with plasma levels. Clinical symptoms of intolerance, especially drowsiness, dizziness, and postural hypotension, should also alert the physician to the need for reduction in dosage.
Initial therapy may be administered in divided doses or a single daily dose.
Maintenance therapy may be given on a once-daily schedule for patient convenience and compliance.
Adolescent and Geriatric Dose
The usual adolescent and geriatric dose is 25 mg to 100 mg daily.
Dosage should be initiated at a lower level and increased according to tolerance and clinical response to a usual maximum of 100 mg daily. In more severely ill patients, dosage may be further increased to 150 mg/day. Doses above 150 mg/day are not recommended in these age groups.
Initial therapy may be administered in divided doses or a single daily dose.
Maintenance therapy may be given on a once-daily schedule for patient convenience and compliance.
Switching a Patient To or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders : At least 14 days should elapse between discontinuation of an MAOI intended to treat psychiatric disorders and initiation of therapy with desipramine hydrochloride tablets. Conversely, at least 14 days should be allowed after stopping desipramine hydrochloride tablets before starting an MAOI intended to treat psychiatric disorders (see CONTRAINDICATIONS).
Use of Desipramine Hydrochloride Tablets with Other MAOI’s Such as Linezolid or Methylene Blue:
Do not start desipramine hydrochloride tablets in a patient who is being treated with linezolid or intravenous methylene blue because there is increased risk of serotonin syndrome. In a patient who requires more urgent treatment of a psychiatric condition, other interventions, including hospitalization, should be considered (see CONTRAINDICATIONS).
In some cases, a patient already receiving desipramine hydrochloride tablets therapy may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of serotonin syndrome in a particular patient, desipramine hydrochloride tablets should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for symptoms of serotonin syndrome for 2 weeks or until 24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with desipramine hydrochloride tablets may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue (see WARNINGS). The risk of administering methylene blue by non-intravenous routes (such as oral tablets orby local injection) or in intravenous doses much lower than 1 mg/kg with desipramine hydrochloride tablet is unclear. The clinician should, nevertheless, be aware of the possibility of emergent symptoms of serotonin syndrome with such use (see WARNINGS).
CONTRAINDICATIONS
The use of MAOIs intended to treat psychiatric disorders with desipramine hydrochloride tablet or within 14 days of stopping treatment with desipramine hydrochloride tablet is contraindicated because of an increased risk of serotonin syndrome. The use of desipramine hydrochloride tablet within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated (see WARNINGS and DOSAGE AND ADMINISTRATION).
Starting desipramine hydrochloride tablets in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome (see WARNINGS and DOSAGE AND ADMINISTRATION).
Desipramine hydrochloride tablets are contraindicated in the acute recovery period following myocardial infarction. It should not be used in those who have shown prior hypersensitivity to the drug. Cross-sensitivity between this and other dibenzazepines is a possibility.
ADVERSE REACTIONS
Included in the following listing are a few adverse reactions that have not been reported with this specific drug. However, the pharmacologic similarities among the tricyclic antidepressant drugs require that each of the reactions be considered when desipramine hydrochloride tablets are given.
Cardiovascular: Hypotension, hypertension, palpitations, heart block, myocardial infarction, stroke, arrhythmias, premature ventricular contractions, tachycardia, ventricular tachycardia, ventricular fibrillation, sudden death
There has been a report of an “acute collapse” and “sudden death” in an 8-year-old (18 kg) male, treated for 2 years for hyperactivity.
There have been additional reports of sudden death in children (see PRECAUTIONS-Pediatric Use).
Psychiatric: Confusional states (especially in the elderly) with hallucinations, disorientation, delusions; anxiety, restlessness, agitation; insomnia and nightmares; hypomania; exacerbation of psychosis
Neurologic: Numbness, tingling, paresthesias of extremities; incoordination, ataxia, tremors; peripheral neuropathy; extrapyramidal symptoms; seizures; alterations in EEG patterns; tinnitus Symptoms attributed to Neuroleptic Malignant Syndrome have been reported during desipramine use with and without concomitant neuroleptic therapy.
Anticholinergic: Dry mouth, and rarely associated sublingual adenitis; blurred vision, disturbance of accommodation, mydriasis, increased intraocular pressure; constipation, paralytic ileus; urinary retention, delayed micturition, dilation of urinary tract
Allergic: Skin rash, petechiae, urticaria, itching, photosensitization (avoid excessive exposure to sunlight), edema (of face and tongue or general), drug fever, cross-sensitivity with other tricyclic drugs
Hematologic: Bone marrow depressions including agranulocytosis, eosinophilia, purpura, thrombocytopenia
Gastrointestinal: Anorexia, nausea and vomiting, epigastric distress, peculiar taste, abdominal cramps, diarrhea, stomatitis, black tongue, hepatitis, jaundice (simulating obstructive), altered liver function, elevated liver function tests, increased pancreatic enzymes
Endocrine: Gynecomastia in the male, breast enlargement and galactorrhea in the female; increased or decreased libido, impotence, painful ejaculation, testicular swelling; elevation or depression of blood sugar levels; syndrome of inappropriate antidiuretic hormone secretion (SIADH)
Other: Weight gain or loss; perspiration, flushing; urinary frequency, nocturia; parotid swelling; drowsiness, dizziness, proneness to falling, weakness and fatigue, headache; fever; alopecia; elevated alkaline phosphatase, hyponatremia.
Withdrawal Symptoms: Though not indicative of addiction, abrupt cessation of treatment after prolonged therapy may produce nausea, headache, and malaise.
To report SUSPECTED ADVERSE REACTIONS, contact Alembic Pharmaceuticals Limited at 1-866-210-9797 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Drugs Metabolized by P450 2D6.
The biochemical activity of the drug metabolizing isozyme cytochrome P450 2D6 (debrisoquin hydroxylase) is reduced in a subset of the Caucasian population (about 7% to 10% of Caucasians are so called “poor metabolizers”); reliable estimates of the prevalence of reduced P450 2D6 isozyme activity among Asian, African and other populations are not yet available. Poor metabolizers have higher than expected plasma concentrations of tricyclic antidepressants (TCAs) when given usual doses. Depending on the fraction of drug metabolized by P450 2D6, the increase in plasma concentration may be small, or quite large (8 fold increase in plasma AUC of the TCA).
In addition, certain drugs inhibit the activity of this isozyme and make normal metabolizers resemble poor metabolizers. An individual who is stable on a given dose of TCA may become abruptly toxic when given one of these inhibiting drugs as concomitant therapy. The drugs that inhibit cytochrome P450 2D6 include some that are not metabolized by the enzyme (quinidine; cimetidine) and many that are substrates for P450 2D6 (many other antidepressants, phenothiazines, and the Type IC antiarrhythmics propafenone and flecainide). While all the SSRIs, e.g., fluoxetine, sertraline, paroxetine, inhibit P450 2D6, they may vary in the extent of inhibition. The extent to which SSRI TCA interactions may pose clinical problems will depend on the degree of inhibition and the pharmacokinetics of the SSRI involved. Nevertheless, caution is indicated in the co-administration of TCAs with any of the SSRIs and also in switching from one class to the other. Of particular importance, sufficient time must elapse before initiating TCA treatment in a patient being withdrawn from fluoxetine, given the long half-life of the parent and active metabolite (at least 5 weeks may be necessary).
Concomitant use of tricyclic antidepressants with drugs that can inhibit cytochrome P450 2D6 may require lower doses than usually prescribed for either the tricyclic antidepressant or the other drug. Furthermore, whenever one of these other drugs is withdrawn from co-therapy, an increased dose of tricyclic antidepressant may be required. It is desirable to monitor TCA plasma levels whenever a TCA is going to be co-administered with another drug known to be an inhibitor of P450 2D6.
Close supervision and careful adjustment of dosage are required when this drug is given concomitantly with anticholinergic or sympathomimetic drugs.
Patients should be warned that while taking this drug their response to alcoholic beverages may be exaggerated.
If desipramine hydrochloride tablets are to be combined with other psychotropic agents such as tranquilizers or sedative/hypnotics, careful consideration should be given to the pharmacology of the agents employed since the sedative effects of desipramine hydrochloride tablets and benzodiazepines (e.g., chlordiazepoxide or diazepam) are additive. Both the sedative and anticholinergic effects of the major tranquilizers are also additive to those of desipramine hydrochloride tablets.
Concomitant use of Monoamine Oxidase Inhibitors (MAOIs) and serotonergic drugs may potentially cause life threatening adverse events (see CONTRAINDICATIONS, WARNINGS, and DOSAGE AND ADMINISTRATION).
DESCRIPTION
Desipramine hydrochloride, USP is an antidepressant drug of the tricyclic type, and is chemically: 5H-Dibenz [bƒ] azepine-5-propanamine, 10, 11-dihydro-N-methyl-, monohydrochloride.
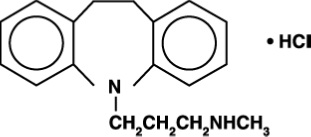
Each desipramine hydrochloride tablet, USP contains 10 mg, 25 mg, 50 mg, 75 mg, 100 mg, or 150 mg of desipramine hydrochloride, USP for oral administration. Inactive Ingredients
The following inactive ingredients are contained in all dosage strengths: citric acid monohydrate, croscarmellose sodium, hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol 6000 (only 150 mg), polyethylene glycol 8000 (10 mg, 25 mg, 50 mg, 75 mg and 100 mg), talc and titanium dioxide.
10 mg tablet also contains FD&C Blue No. 1 Aluminum Lake and iron oxide yellow.
25 mg tablet also contains D&C Yellow No. 10 Aluminum Lake, FD&C Blue No. 2 Aluminum Lake and FD&C Yellow No. 6 Aluminum Lake.
50 mg tablet also contains D&C Yellow No. 10 Aluminum Lake, FD&C Blue No.1 Aluminum Lake, and FD&C Yellow No. 6 Aluminum Lake.
75 mg tablet also contains FD&C Red No. 40 Aluminum Lake and FD&C Yellow No. 6 Aluminum Lake.
100 mg tablet also contains D&C Yellow No. 10 Aluminum Lake, FD&C Blue No. 2 Aluminum Lake and FD&C Yellow No. 6 Aluminum Lake.
CLINICAL PHARMACOLOGY
Mechanism of Action
Available evidence suggests that many depressions have a biochemical basis in the form of a relative deficiency of neurotransmitters such as norepinephrine and serotonin. Norepinephrine deficiency may be associated with relatively low urinary 3-methoxy-4-hydroxyphenyl glycol (MHPG) levels, while serotonin deficiencies may be associated with low spinal fluid levels of 5hydroxyindoleacetic acid.
While the precise mechanism of action of the tricyclic antidepressants is unknown, a leading theory suggests that they restore normal levels of neurotransmitters by blocking the re-uptake of these substances from the synapse in the central nervous system. Evidence indicates that the secondary amine tricyclic antidepressants, including desipramine hydrochloride tablets, may have greater activity in blocking the re-uptake of norepinephrine. Tertiary amine tricyclic antidepressants, such as amitriptyline, may have greater effect on serotonin re-uptake.
Desipramine hydrochloride tablet is not a monoamine oxidase inhibitor (MAOI) and does not act primarily as a central nervous system stimulant. It has been found in some studies to have a more rapid onset of action than imipramine. Earliest therapeutic effects may occasionally be seen in 2 to 5 days, but full treatment benefit usually requires 2 to 3 weeks to obtain.
Metabolism
Tricyclic antidepressants, such as desipramine hydrochloride, are rapidly absorbed from the gastrointestinal tract. Tricyclic antidepressants or their metabolites are to some extent excreted through the gastric mucosa and reabsorbed from the gastrointestinal tract. Desipramine is metabolized in the liver, and approximately 70% is excreted in the urine.
The rate of metabolism of tricyclic antidepressants varies widely from individual to individual, chiefly on a genetically determined basis. Up to a 36-fold difference in plasma level may be noted among individuals taking the same oral dose of desipramine. The ratio of 2-hydroxydesipramine to desipramine may be increased in the elderly, most likely due to decreased renal elimination with aging.
Certain drugs, particularly the psychostimulants and the phenothiazines, increase plasma levels of concomitantly administered tricyclic antidepressants through competition for the same metabolic enzyme systems. Concurrent administration of cimetidine and tricyclic antidepressants can produce clinically significant increases in the plasma concentrations of the tricyclic antidepressants. Conversely, decreases in plasma levels of the tricyclic antidepressants have been reported upon discontinuation of cimetidine, which may result in the loss of the therapeutic efficacy of the tricyclic antidepressant. Other substances, particularly barbiturates and alcohol, induce liver enzyme activity and thereby reduce tricyclic antidepressant plasma levels. Similar effects have been reported with tobacco smoke.
Research on the relationship of plasma level to therapeutic response with the tricyclic antidepressants has produced conflicting results. While some studies report no correlation, many studies cite therapeutic levels for most tricyclics in the range of 50 to 300 nanograms per milliliter. The therapeutic range is different for each tricyclic antidepressant. For desipramine, an optimal range of therapeutic plasma levels has not been established.
HOW SUPPLIED
Desipramine hydrochloride tablets USP, 10 mg are blue, round, biconvex film-coated tablets debossed with “L” on one side and “01”on other side. They are supplied as follows:
NDC 62332-315-30 bottle of 30 tablets with child-resistant closure
NDC 62332-315-31 bottle of 100 tablets with child-resistant closure
NDC 62332-315-91 bottle of 1000 tablets
Desipramine hydrochloride tablets USP, 25 mg are yellow, round, biconvex film-coated tablets debossed with “L” on one side and “02” on other side. They are supplied as follows:
NDC 62332-316-30 bottle of 30 tablets with child-resistant closure
NDC 62332-316-31 bottle of 100 tablets with child-resistant closure
NDC 62332-316-91 bottle of 1000 tablets
Desipramine hydrochloride tablets USP, 50 mg are green, round, biconvex film-coated tablets debossed with “L” on one side and “03”on other side. They are supplied as follows:
NDC 62332-317-30 bottle of 30 tablets with child-resistant closure
NDC 62332-317-31 bottle of 100 tablets with child-resistant closure
NDC 62332-317-91 bottle of 1000 tablets
Desipramine hydrochloride tablets USP, 75 mg are orange, round, biconvex film-coated tablets debossed with “566” on one side plain on other side. They are supplied as follows:
NDC 62332-318-30 bottle of 30 tablets with child-resistant closure
NDC 62332-318-31 bottle of 100 tablets with child-resistant closure
NDC 62332-318-91 bottle of 1000 tablets
Desipramine hydrochloride tablets USP, 100 mg are peach, round, biconvex film-coated tablets debossed with “567” on one side and plain on other side. They are supplied as follows:
NDC 62332-319-30 bottle of 30 tablets with child-resistant closure
NDC 62332-319-31 bottle of 100 tablets with child-resistant closure
NDC 62332-319-91 bottle of 1000 tablets
Desipramine hydrochloride tablets USP, 150 mg are white, round, biconvex film-coated tablets debossed with “568” on one side and plain on other side. They are supplied as follows:
NDC 62332-320-30 bottle of 30 tablets with child-resistant closure
NDC 62332-320-50 bottle of 50 tablets with child-resistant closure
NDC 62332-320-31 bottle of 100 tablets with child-resistant closure
NDC 62332-320-91 bottle of 1000 tablets
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Dispense in a tight container. Protect from excessive heat.
Rx Only
Manufactured by:
Alembic Pharmaceuticals Limited
(Formulation Division),
Panelav 389350, Gujarat, India
Manufactured for:
Alembic Pharmaceuticals, Inc.
Bedminster, NJ 07921, USA
Revised: 07/2025