Get your patient on Dexamethasone Sodium Phosphate - Dexamethasone Sodium Phosphate injection, Emulsion (Dexamethasone Sodium Phosphate)
Dexamethasone Sodium Phosphate - Dexamethasone Sodium Phosphate injection, Emulsion prescribing information
DOSAGE AND ADMINISTRATION
A. Intravenous or intramuscular administration.
The initial dosage of dexamethasone sodium phosphate injection may vary from 0.50 mg/day to 9.0 mg/day depending on the specific disease entity being treated. In situations of less severity, lower doses will generally suffice while in selected patients higher initial doses may be required. Usually the parenteral dosage ranges are one-third to one-half the oral dose given every 12 hours. However, in certain overwhelming, acute, life-threatening situations, administration of dosages exceeding the usual dosages may be justified and may be in multiples of the oral dosages.
For the treatment of unresponsive shock high pharmacologic doses of this product are currently recommended. Reported regimens range from 1 to 6 mg/kg of body weight as a single intravenous injection to 40 mg initially followed by repeat intravenous injection every 2 to 6 hours while shock persists.
For the treatment of cerebral edema in adults an initial intravenous dose of 10 mg is recommended followed by 4 mg intramuscularly every six hours until maximum response has been noted. This regimen may be continued for several days postoperatively in patients requiring brain surgery. Oral dexamethasone, 1 to 3 mg t.i.d., should be given as soon as possible and dosage tapered off over a period of five to seven days. Nonoperative cases may require continuous therapy to remain free of symptoms of increased intracranial pressure. The smallest effective dose should be used in children, preferably orally. This may approximate 0.2 mg/kg/24 hours in divided doses.
In treatment of acute exacerbations of multiple sclerosis daily doses of 200 mg of prednisolone for a week followed by 80 mg every other day or 4-8 mg dexamethasone every other day for 1 month have been shown to be effective.
The initial dosage should be maintained or adjusted until a satisfactory response is noted. If after a reasonable period of time there is a lack of satisfactory clinical response, dexamethasone sodium phosphate injection should be discontinued and the patient transferred to other appropriate therapy. It should be emphasized that dosage requirements are variable and must be individualized on the basis of the disease under treatment and the response of the patient.
After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage which will maintain an adequate clinical response is reached. It should be kept in mind that constant monitoring is needed in regard to drug dosage. Included in the situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient's individual drug responsiveness and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this later situation it may be necessary to increase the dosage of dexamethasone sodium phosphate injection for a period of time consistent with the patient's condition. If after a long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly.
B. Intra-articular, soft tissue or intralesional administration.
The dose for intrasynovial administration is usually 2 to 4 mg for large joints and 0.8 to 1 mg for small joints. For soft tissue and bursal injections a dose of 2 to 4 mg is recommended. Ganglia require a dose of 1 to 2 mg. A dose of 0.4 to 1 mg is used for injection into tendon sheaths. Injection into intervertebral joints should not be attempted at any time and hip joint injection cannot be recommended as an office procedure.
Intrasynovial and soft tissue injections should be employed only when affected areas are limited to 1 or 2 sites. It should be remembered that corticoids provide palliation only and that other conventional or curative methods of therapy should be employed when indicated.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Frequency of injection usually ranges from once every 3 to 5 days to once every 2 to 3 weeks. Frequent intra-articular injection may cause damage to joint tissue.
CONTRAINDICATIONS
Systemic fungal infection.
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Fluid and electrolyte disturbances:
- Sodium retention
- Fluid retention
- Congestive heart failure in susceptible patients
- Potassium loss
- Hypokalemic alkalosis
- Hypertension
Musculoskeletal:
- Muscle weakness
- Steroid myopathy
- Loss of muscle mass
- Osteoporosis
- Vertebral compression fractures
- Aseptic necrosis of femoral and humeral heads
- Pathologic fracture of long bones
Gastrointestinal:
- Peptic ulcer with possible subsequent perforation and hemorrhage
- Pancreatitis
- Abdominal distention
- Ulcerative esophagitis
Dermatological:
- Impaired wound healing
- Thin fragile skin
- Facial erythema
- Increased sweating
- May suppress reactions to skin tests
- Petechiae and ecchymoses
Neurological:
- Convulsions
- Increased intracranial pressure with papilledema (pseudotumor cerebri) usually after treatment
- Vertigo
- Headache
Ophthalmic:
- Posterior subcapsular cataracts
- Increased intraocular pressure
- Glaucoma
Endocrine:
- Menstrual irregularities
- Development of cushingoid state
- Suppression of growth in children
- Secondary adrenocortical and pituitary unresponsiveness, particularly in times of stress, as in trauma, surgery, or illness
- Decreased carbohydrate tolerance
- Manifestations of latent diabetes mellitus
- Increased requirements for insulin or oral hypoglycemic agents in diabetics
Metabolic:
- Negative nitrogen balance due to protein catabolism
Miscellaneous:
- Hyperpigmentation or hypopigmentation
- Subcutaneous and cutaneous atrophy
- Sterile abscess
- Post-injection flare, following intra-articular use
- Charcot-like arthropathy
- Itching, burning, tingling in the ano-genital region
DESCRIPTION
Dexamethasone Sodium Phosphate Injection, USP, is a water-soluble inorganic ester of dexamethasone which produces a rapid response even when injected intramuscularly.
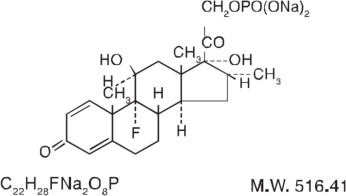
Dexamethasone Sodium Phosphate, USP has a molecular weight of 516.41 and chemically is Pregna-1,4-diene-3,20-dione, 9-fluoro-11,17-dihydroxy-16-methyl-21-(phosphonooxy)-, disodium salt, (11 ß , 16 α ).
It occurs as a white to creamy white powder, is exceedingly hygroscopic, is soluble in water and its solutions have a pH between 7.0 and 8.5. It has the following structural formula:
Each mL of Dexamethasone Sodium Phosphate Injection, USP (Preservative Free) contains
Dexamethasone Sodium Phosphate, USP equivalent to 10 mg dexamethasone phosphate; 25.80 mg sodium citrate dihydrate; and water for injection, q.s. pH adjusted with citric acid or sodium hydroxide, if necessary. pH: 7.0 to 8.5.
ACTIONS
Naturally occurring glucocorticoids (hydrocortisone), which also have salt-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs are primarily used for their potent anti-inflammatory effects in disorders of many organ systems.
Glucocorticoids cause profound and varied metabolic effects. In addition, they modify the body's immune responses to diverse stimuli.
HOW SUPPLIED
Product Code | Unit of Sale | Strength | Each |
RF727910 | NDC 76045-212-10 Unit of 24 | 10 mg/mL | NDC 76045-212-00 1 mL Single-Dose Prefilled Syringe This product is RFID enabled. |
727910 | NDC 76045-109-10 Unit of 24 | 10 mg/mL | NDC 76045-109-01 1 mL Single-Dose Prefilled Syringe |
Dexamethasone Sodium Phosphate Injection, USP (Preservative Free) equivalent to 10 mg dexamethasone phosphate, is supplied as:
Storage
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Sensitive to heat. Do not autoclave.
Protect from freezing.
Protect from light.
Do NOT place syringe on a Sterile Field.
Discard unused portion.
INSTRUCTIONS FOR USE
Figure 1: Outer Packaging and Prefilled Syringe
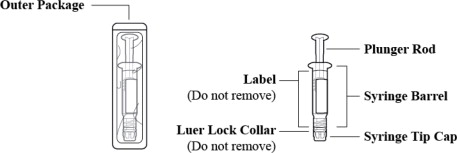
NOTES:
- Do not introduce any other fluid into the syringe at any time.
- Do not dilute for IV push.
- Do not re-sterilize the syringe.
- Do not use this product on a sterile field.
- This product is for single dose only.
1. Inspect the outer packaging (blister pack) to confirm the integrity of the packaging. Do not use if the blister pack or the prefilled syringe has been damaged.
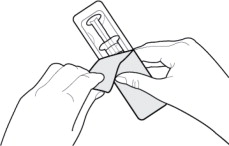
2. Remove the syringe from the outer packaging. (See Figure 2 )
- Figure 2
3. Visually inspect the syringe. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
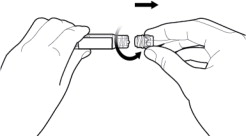
4. Twist off the syringe tip cap. Do not remove the label around the luer lock collar. (See Figure 3 )
- Figure 3
5. Expel air bubble(s). Adjust the dose (if applicable).
6. Administer the dose ensuring that pressure is maintained on the plunger rod during the entire administration.
7. Discard the used syringe into an appropriate receptacle.
For more information concerning this drug, please call Fresenius Kabi USA, LLC at 1-800-551-7176.
The brand names mentioned in this document are the trademarks of their respective owners.
U.S. Patents 9731082, 10661018 and 11426522

www.fresenius-kabi.com/us 451518B Revised: October 2025