Get your patient on Dextroamphetamine - Dextroamphetamine solution (Dextroamphetamine)
Dextroamphetamine - Dextroamphetamine solution prescribing information
WARNING: ABUSE, MISUSE, AND ADDICTION
INDICATIONS AND USAGE
Dextroamphetamine sulfate oral solution is indicated in:
Narcolepsy
Attention Deficit Disorder with Hyperactivity: As an integral part of a total treatment program that typically includes other remedial measures (psychological, educational, social) for a stabilizing effect in pediatric patients (ages 3 years to 16 years) with a behavioral syndrome characterized by the following group of developmentally inappropriate symptoms: Moderate to severe distractibility, short attention span, hyperactivity, emotional lability, and impulsivity. The diagnosis of this syndrome should not be made with finality when these symptoms are only of comparatively recent origin. Nonlocalizing (soft) neurological signs, learning disability, and abnormal EEG may or may not be present, and a diagnosis of central nervous system dysfunction may or may not be warranted.
DOSAGE AND ADMINISTRATION
Amphetamines should be administered at the lowest effective dosage and dosage should be individually adjusted. Late evening doses should be avoided because of the resulting insomnia.
Narcolepsy: Usual dose is 5 mg to 60 mg per day in divided doses, depending on the individual patient response.
Narcolepsy seldom occurs in children under 12 years of age; however, when it does, dextroamphetamine sulfate oral solution may be used. The suggested initial dose for patients aged 6 to 12 is 5 mg daily; daily dose may be raised in increments of 5 mg at weekly intervals until optimal response is obtained. In patients 12 years of age and older, start with 10 mg daily; daily dosage may be raised in increments of 10 mg at weekly intervals until optimal response is obtained. If bothersome adverse reactions appear (e.g., insomnia or anorexia), dosage should be reduced. Give first dose on awakening; additional doses (1 or 2) at intervals of 4 to 6 hours.
Attention Deficit Disorder with Hyperactivity: Not recommended for pediatric patients under 3 years of age.
In pediatric patients from 3 to 5 years of age, start with 2.5 mg daily; daily dosage may be raised in increments of 2.5 mg at weekly intervals until optimal response is obtained.
In pediatric patients 6 years of age and older, start with 5 mg once or twice daily; daily dosage may be raised in increments of 5 mg at weekly intervals until optimal response is obtained. Only in rare cases will it be necessary to exceed a total of 40 mg per day.
Give first dose on awakening; additional doses (1 or 2) at intervals of 4 to 6 hours.
Where possible, drug administration should be interrupted occasionally to determine if there is a recurrence of behavioral symptoms sufficient to require continued therapy.
Prior to treating patients with dextroamphetamine sulfate assess:
CONTRAINDICATIONS
Known hypersensitivity to amphetamine products. During or within 14 days following the administration of monoamine oxidase inhibitors (hypertensive crisis may result).
ADVERSE REACTIONS
Cardiovascular: Palpitations, tachycardia, elevation of blood pressure. There have been isolated reports of cardiomyopathy associated with chronic amphetamine use.
Central Nervous System: Psychotic episodes at recommended doses (rare), overstimulation, restlessness, dizziness, insomnia, euphoria, dyskinesia, dysphoria, tremor, headache, exacerbation of motor and verbal tics and Tourette’s syndrome.
Gastrointestinal: Dryness of the mouth, unpleasant taste, diarrhea, constipation, intestinal ischemia and other gastrointestinal disturbances. Anorexia and weight loss may occur as undesirable effects.
Allergic: Urticaria.
Endocrine: Impotence, changes in libido.
Musculoskeletal: Rhabdomyolysis.
To report SUSPECTED ADVERSE REACTIONS, contact Cranbury Pharmaceuticals, LLC at 1-732-940-0358 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
MAO Inhibitors - MAOI antidepressants, as well as a metabolite of furazolidone, slow amphetamine metabolism. This slowing potentiates amphetamines, increasing their effect on the release of norepinephrine and other monoamines from adrenergic nerve endings; this can cause headaches and other signs of hypertensive crisis. A variety of neurological toxic effects and malignant hyperpyrexia can occur, sometimes with fatal results.
Serotonergic Drugs - The concomitant use of dextroamphetamine sulfate oral solution and serotonergic drugs increases the risk of serotonin syndrome. Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during dextroamphetamine sulfate oral solution initiation or dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine sulfate oral solution and the concomitant serotonergic drug(s) (see WARNINGS , PRECAUTIONS ) . Examples of serotonergic drugs include selective serotonin reuptake inhibitors (SSRI), serotonin norepinephrine reuptake inhibitors (SNRI), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John’s Wort.
CYP2D6 Inhibitors - The concomitant use of dextroamphetamine sulfate oral solution and CYP2D6 inhibitors may increase the exposure of dextroamphetamine sulfate oral solution compared to the use of the drug alone and increase the risk of serotonin syndrome. Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during dextroamphetamine sulfate oral solution initiation and after a dosage increase. If serotonin syndrome occurs, discontinue dextroamphetamine sulfate oral solution and the CYP2D6 inhibitor (see WARNINGS , OVERDOSAGE ) . Examples of CYP2D6 Inhibitors include paroxetine and fluoxetine (also serotonergic drugs), quinidine, ritonavir.
Acidifying Agents – Gastrointestinal acidifying agents (guanethidine, reserpine, glutamic acid HCl, ascorbic acid, fruit juices, etc.) lower absorption of amphetamines. Urinary acidifying agents (ammonium chloride, sodium acid phosphate, etc.) increase the concentration of the ionized species of the amphetamine molecule, thereby increasing urinary excretion. Both groups of agents lower blood levels and efficacy of amphetamines.
Adrenergic Blockers – Adrenergic blockers are inhibited by amphetamines.
Alkalinizing Agents – Gastrointestinal alkalinizing agents (sodium bicarbonate, etc.) increase absorption of amphetamines. Urinary alkalinizing agents (acetazolamide, some thiazides) increase the concentration of the non-ionized species of the amphetamine molecule, thereby decreasing urinary excretion. Both groups of agents increase blood levels and therefore potentiate the actions of amphetamines.
Antidepressants, Tricyclic – Amphetamines may enhance the activity of tricyclic or sympathomimetic agents; d-amphetamine with desipramine or protriptyline and possibly other tricyclics cause striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated.
Antihistamines – Amphetamines may counteract the sedative effect of antihistamines.
Antihypertensives – Amphetamines may antagonize the hypotensive effects of antihypertensives.
Chlorpromazine – Chlorpromazine blocks dopamine and norepinephrine reuptake, thus inhibiting the central stimulant effects of amphetamines, and can be used to treat amphetamine poisoning.
Ethosuximide – Amphetamines may delay intestinal absorption of ethosuximide.
Haloperidol – Haloperidol blocks dopamine and norepinephrine reuptake, thus inhibiting the central stimulant effects of amphetamines.
Lithium Carbonate – The stimulatory effects of amphetamines may be inhibited by lithium carbonate.
Meperidine – Amphetamines potentiate the analgesic effect of meperidine.
Methenamine Therapy – Urinary excretion of amphetamines is increased, and efficacy is reduced, by acidifying agents used in methenamine therapy.
Norepinephrine – Amphetamines enhance the adrenergic effect of norepinephrine.
Phenobarbital – Amphetamines may delay intestinal absorption of phenobarbital; co-administration of phenobarbital may produce a synergistic anticonvulsant action.
Phenytoin – Amphetamines may delay intestinal absorption of phenytoin; co-administration of phenytoin may produce a synergistic anticonvulsant action.
Propoxyphene – In cases of propoxyphene overdosage, amphetamine CNS stimulation is potentiated and fatal convulsions can occur.
Veratrum Alkaloids – Amphetamines inhibit the hypotensive effect of veratrum alkaloids.
Drug/Laboratory Test Interactions
Amphetamines can cause a significant elevation in plasma corticosteroid levels. This increase is greatest in the evening.
Amphetamines may interfere with urinary steroid determinations.
Carcinogenesis/Mutagenesis
Mutagenicity studies and long-term studies in animals to determine the carcinogenic potential of dextroamphetamine sulfate have not been performed.
Pregnancy
Teratogenic Effects
Pregnancy Category C
Dextroamphetamine sulfate has been shown to have embryotoxic and teratogenic effects when administered to A/Jax mice and C57BL mice in doses approximately 41 times the maximum human dose. Embryotoxic effects were not seen in New Zealand white rabbits given the drug in doses 7 times the human dose nor in rats given 12.5 times the maximum human dose. While there are no adequate and well-controlled studies in pregnant women, there has been one report of severe congenital bony deformity, tracheoesophageal fistula, and anal atresia (VATER association) in a baby born to a woman who took dextroamphetamine sulfate with lovastatin during the first trimester of pregnancy. Dextroamphetamine sulfate should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects
Infants born to mothers dependent on amphetamines have an increased risk of premature delivery and low birth weight. Also, these infants may experience symptoms of withdrawal as demonstrated by dysphoria, including agitation, and significant lassitude.
Nursing Mothers
Amphetamines are excreted in human milk. Mothers taking amphetamines should be advised to refrain from nursing.
Pediatric Use
Long-term effects of amphetamines in pediatric patients have not been well established.
Amphetamines are not recommended for use in pediatric patients under 3 years of age with Attention Deficit Disorder with Hyperactivity described under INDICATIONS AND USAGE .
Clinical experience suggests that in psychotic children, administration of amphetamines may exacerbate symptoms of behavior disturbance and thought disorder.
Amphetamines have been reported to exacerbate motor and phonic tics and Tourette’s syndrome. Therefore, clinical evaluation for tics and Tourette’s syndrome in children and their families should precede use of stimulant medications.
Data are inadequate to determine whether chronic administration of amphetamines may be associated with growth inhibition; therefore, growth should be monitored during treatment.
Drug treatment is not indicated in all cases of Attention Deficit Disorder with Hyperactivity and should be considered only in light of the complete history and evaluation of the child. The decision to prescribe amphetamines should depend on the physician’s assessment of the chronicity and severity of the child’s symptoms and their appropriateness for his or her age. Prescription should not depend solely on the presence of one or more of the behavioral characteristics.
When these symptoms are associated with acute stress reactions, treatment with amphetamines is usually not indicated.
DESCRIPTION
Dextroamphetamine sulfate, USP is the dextro isomer of the compound d,l -amphetamine sulfate, a sympathomimetic amine of the amphetamine group. Chemically, dextroamphetamine is d -alpha-methylphenethylamine, and is present in all forms of dextroamphetamine sulfate as the neutral sulfate.
Structural Formula:
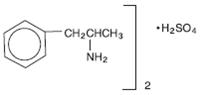
Dextroamphetamine sulfate, USP is a colorless, bubble gum flavored oral solution. Each 5 mL (1 teaspoonful) of dextroamphetamine sulfate oral solution contains 5 mg of dextroamphetamine sulfate. Inactive ingredients consist of anhydrous citric acid, benzoic acid, bubble gum flavor, purified water, sodium citrate anhydrous, sodium saccharin, and sorbitol solution.
CLINICAL PHARMACOLOGY
Amphetamines are noncatecholamine, sympathomimetic amines with CNS stimulant activity. Peripheral actions include elevations of systolic and diastolic blood pressures and weak bronchodilator and respiratory stimulant action.
There is neither specific evidence that clearly establishes the mechanism whereby amphetamines produce mental and behavioral effects in children, nor conclusive evidence regarding how these effects relate to the condition of the central nervous system.
Pharmacokinetics
Ingestion of 10 mg of dextroamphetamine sulfate in oral solution form by healthy volunteers produced an average peak dextroamphetamine blood level of 33.2 ng/mL. The half-life was 11.75 hours. The average urinary recovery was 38% in 48 hours.
In 12 healthy subjects, the rate and extent of dextroamphetamine absorption were similar following administration of the sustained release capsule formulation in the fed (58 to 75 gm fat) and fasted state.
HOW SUPPLIED
Dextroamphetamine Sulfate Oral Solution 5 mg/5 mL is a colorless, bubble gum flavored oral solution. It is available in 16 fluid ounces (473 mL) bottles, NDC 27808-085-01.
Store at 20° to 25°C (68° to 77°F); excursions permitted from 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Dispense in a tight, light-resistant container with a child-resistant closure.
Manufactured by
Tris Pharma, Inc.
Monmouth Junction, NJ 08852
For:
Cranbury Pharmaceuticals, LLC
Monmouth Junction, NJ 08852
LB8255
Rev.11
Revised: 05/2025
Dextroamphetamine - Dextroamphetamine solution PubMed™ news
- Journal Article • 2026 MayEffects of stimulant medication on neuropsychological functioning in college students without ADHD.
- Journal Article • 2026 MayUptake and Retention on Prescribed Psychostimulants for Harm Reduction: A Canadian Cohort Study.
- Journal Article • 2026 AprSympathomimetic Overdose With Lisdexamfetamine in a Pediatric Patient Resulting in Stress Cardiomyopathy-A Case Report.
- Journal Article • 2026 AprComparing daytime pharmacokinetic and pharmacodynamic profiles of once-daily lisdexamfetamine versus twice-daily dexamfetamine in adult ADHD.
- Journal Article • 2026 AprExpert meeting report: epidemiology and management of acquired hypothalamic obesity.