Get your patient on Dextrose - Dextrose Monohydrate injection, Solution, Concentrate (Dextrose Monohydrate)
Dextrose - Dextrose Monohydrate injection, Solution, Concentrate prescribing information
INDICATIONS AND USAGE
10% Dextrose Injection, USP (concentrated dextrose in water) in a partial-fill container is indicated for admixture with amino acids or dilution with other compatible IV fluids to provide a 5% final dextrose concentration for intravenous infusion in patients whose condition requires parenteral nutrition.
DOSAGE AND ADMINISTRATION
Concentrated Dextrose in Water is administered by slow intravenous infusion (a) after admixture with amino acid solutions or (b) after dilution with other compatible IV fluids. Dosage should be adjusted to meet the requirements of each individual patient.
The maximum rate at which dextrose can be infused without producing glycosuria is 0.5 g/kg of body weight /hr. About 95% of the dextrose is retained when infused at a rate of 0.8 g/kg/hr.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
A list of nutritional admixture values is appended.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. (See PRECAUTIONS ).
Drug Interaction
Additives may be incompatible. Consult with pharmacist, if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
Some opacity of the plastic due to moisture absorption during sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
WARNING: Do not use flexible container in series connections.
CONTRAINDICATIONS
A concentrated dextrose solution should not be used when intracranial or intraspinal hemorrhage is present nor in the presence of delirium tremens if the patient is already dehydrated.
Dextrose Injection, USP without electrolytes should not be administered simultaneously with blood through the same infusion set because of the possibility that pseudoagglutination of red cells may occur.
ADVERSE REACTIONS
Hyperosmolar syndrome, resulting from excessively rapid administration of concentrated dextrose may cause hypovolemia, dehydration, mental confusion and/or loss of consciousness.
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
Drug Interaction
Additives may be incompatible. Consult with pharmacist, if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
Some opacity of the plastic due to moisture absorption during sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
WARNING: Do not use flexible container in series connections.
DESCRIPTION
10% Dextrose Injection, USP (concentrated dextrose in water) is a sterile, nonpyrogenic, hypertonic solution of Dextrose, USP in water for injection for intravenous administration after appropriate admixture or dilution.
10% Dextrose Injection, USP is provided as a 500 mL volume in a 1000 mL partial-fill container. The container is designed to facilitate admixture or dilution.
See table under HOW SUPPLIED for summary of content and characteristics of this concentrated solution.
The solution contains no bacteriostat, antimicrobial agent or added buffer and is intended only for use as a single-dose injection following admixture or dilution.
The flexible plastic container is fabricated from a specially formulated polyvinyl chloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the plastic container materials. Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
Dextrose Injection, USP is a parenteral fluid and nutrient replenisher.
Dextrose Injection, USP is chemically designated D-glucose monohydrate (C 6 H 12 O 6 • H 2 O), a hexose sugar freely soluble in water. It has the following structural formula:
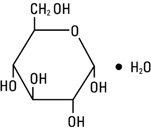
Water for Injection, USP is chemically designated H 2 O.
CLINICAL PHARMACOLOGY
When administered intravenously, solutions containing carbohydrate in the form of dextrose restore blood glucose levels and provide calories. Carbohydrate in the form of dextrose may aid in minimizing liver glycogen depletion and exerts a protein sparing action. Dextrose Injection, USP undergoes oxidation to carbon dioxide and water.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirement ranges from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production, respectively).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments, and sodium (Na + ) plays a major role in maintaining physiologic equilibrium.
HOW SUPPLIED
10% Dextrose Injection, USP is supplied in single-dose, partial-fill, flexible containers as follows: a 500 mL volume in a 1000 mL container. See the following table.
NDC No. | % Conc. | Fill Volume (mL) | Total Grams of Dextrose Hydrous Per Container | kcal Caloric value calculated on the basis of 3.4 kcal/g of dextrose, hydrous. /100 mL (Per Container) | mOsmol/liter (calc.) | pH (range) |
0409-7938-19 | 10 | 500 | 50 | 34 (170) | 505 | 4.3 (3.2 to 6.5) |
| 0990-7938-19 | 10 | 500 | 50 | 34 (170) | 505 | 4.3 (3.2 to 6.5) |
ICU Medical is transitioning NDC codes from the "0409" to a "0990" labeler code. Both NDC codes are expected to be in the market for a period of time.
Dextrose Pre-Dilution Concentration | Admixture Non-Protein kcal/g N | Admixture Non-Protein kcal/liter | Admixture g N/liter | Admixture Dextrose Concentration |
10% | 31 | 170 | 5.5 | 5% |
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
Revised: November, 2018
EN-4694
ICU Medical, Inc., Lake Forest, Illinois, 60045, USA