Get your patient on Diazepam - Diazepam injection, Solution (Diazepam)
Diazepam - Diazepam injection, Solution prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death (see WARNINGS ).
- Reserve concomitant prescribing of these drugs in patients for whom alternative treatment options are inadequate.
- Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS and PRECAUTIONS ).
- The use of benzodiazepines, including diazepam, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing diazepam and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (see WARNINGS ).
- The continued use of benzodiazepines may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Although diazepam is indicated only for intermittent use (see INDICATIONS AND USAGE and DOSAGE AND ADMINISTRATION ), if used more frequently than recommended, abrupt discontinuation or rapid dosage reduction of diazepam may precipitate acute withdrawal reactions, which can be life-threatening. For patients using diazepam more frequently than recommended, to reduce the risk of withdrawal reactions, use a gradual taper to discontinue diazepam (see WARNINGS ).
INDICATIONS AND USAGE
Diazepam is indicated for the management of anxiety disorders or for the short-term relief of the symptoms of anxiety. Anxiety or tension associated with the stress of everyday life usually does not require treatment with an anxiolytic. In acute alcohol withdrawal, diazepam may be useful in the symptomatic relief of acute agitation, tremor, impending or acute delirium tremens and hallucinosis. As an adjunct prior to endoscopic procedures if apprehension, anxiety or acute stress reactions are present, and to diminish the patient's recall of the procedures (See WARNINGS ).
Diazepam is a useful adjunct for the relief of skeletal muscle spasm due to reflex spasm to local pathology (such as inflammation of the muscles or joints, or secondary to trauma); spasticity caused by upper motor neuron disorders (such as cerebral palsy and paraplegia); athetosis; stiff-man syndrome; and tetanus. Diazepam injection is a useful adjunct in status epilepticus. Diazepam is a useful premedication (the intramuscular route is preferred) for relief of anxiety and tension in patients who are to undergo surgical procedures. Intravenously, prior to cardioversion for the relief of anxiety and tension and to diminish the patient's recall of the procedure.
DOSAGE AND ADMINISTRATION
Dosage should be individualized for maximum beneficial effect. The usual recommended dose in older children and adults ranges from 2 mg to 20 mg intramuscular or intravenous, depending on the indication and its severity. In some conditions, e.g., tetanus, larger doses may be required. (See dosage for specific indications.) In acute conditions the injection may be repeated within one hour although an interval of 3 to 4 hours is usually satisfactory. Lower doses (usually 2 mg to 5 mg) and slow increase in dosage should be used for elderly or debilitated patients and when other sedative drugs are administered.(See WARNINGS and ADVERSE REACTIONS ).
For dosage in infants above the age of 30 days and children, see the specific indications below. When intravenous use is indicated, facilities for respiratory assistance should be readily available. Intramuscular: Diazepam injection should be injected deeply into the muscle. Intravenous Use: For the treatment of status epilepticus in children, the solution should be injected slowly, taking one minute for administration (See WARNINGS , particularly for use in children). For other indications, the solution should be injected slowly taking at least one minute for each 5 mg (1 mL) given. Do not use small veins, such as those on the dorsum of the hand or wrist. Extreme care should be taken to avoid intra-arterial administration or extravasation. Do not mix or dilute diazepam with other solutions or drugs in syringe or infusion container. If it is not feasible to administer diazepam directly intravenous, it may be injected slowly through the infusion tubing as close as possible to the vein insertion.
USUAL ADULT DOSAGE | DOSAGE RANGE IN CHILDREN (Intravenous administration should be made slowly) | ||
Moderate Anxiety Disorders and Symptoms of Anxiety | 2 mg to 5 mg, intramuscular or intravenous . Repeat in 3 to 4 hours, if necessary. | ||
Severe Anxiety Disorders and Symptoms of Anxiety | 5 mg to 10 mg, intramuscular or intravenous . Repeat in 3 to 4 hours, if necessary. | ||
Acute Alcohol Withdrawal: | |||
As an aid in symptomatic relief of acute agitation, tremor, impending or acute delirium tremens, and hallucinosis. | 10 mg, intramuscular or intravenous initially, then 5 mg to 10 mg in 3 to 4 hours, if necessary. | ||
Endoscopic Procedures: | |||
Adjunctively, if apprehension, anxiety or acute stress reaction are present prior to endoscopic procedures. Dosage of narcotics should be reduced by at least a third and in some cases may be omitted. See PRECAUTIONS for peroral procedures. | Titrate intravenous dosage to desired sedative response, such as slurring of speech, with slow administration immediately prior to the procedure. Generally 10 mg or less is adequate, but up to 20 mg intravenous may be given, particularly when concomitant narcotics are omitted. If intravenous cannot be used, 5 mg to10 mg intramuscular approximately 30 minutes prior to the procedure. | ||
Muscle Spasm | |||
Associated with local pathology, cerebral palsy, athetosis, stiff-man syndrome, or tetanus. | 5 mg to 10 mg, intramuscular or intravenous initially, then 5 mg to 10 mg in 3 to 4 hours, if necessary. For tetanus, larger doses may be required. | For tetanus in infants over 30 days of age, 1 mg to 2 mg intramuscular or intravenous, slowly, repeated every 3 to 4 hours as necessary. In children 5 years or older, 5 mg to 10 mg repeated every 3 to 4 hours may be required to control tetanus spasms. Respiratory assistance should be available. | |
Status Epilepticus and Severe Recurrent Convulsive Seizures: | |||
In the convulsing patient, the intravenous route is by far preferred. This injection should be administered slowly. However, if intravenous administration is impossible, the intramuscular route may be used. | 5 mg to 10 mg initially (intravenous preferred). This injection may be repeated if necessary at 10 to 15 minute intervals up to a maximum dose of 30 mg. If necessary, therapy with diazepam may be repeated in 2 to 4 hours; however, residual active metabolites may persist, and readministration should be made with this consideration. Extreme caution must be exercised with individuals with chronic lung disease or unstable cardiovascular status. | Children 3 months up to 17 years of age with status epilepticus: First dose: 0.2 mg/kg (maximum 8 mg) by slow intravenous push (one minute in duration). Second dose (if necessary; 5 minutes after the first dose): 0.1 mg/kg (maximum 4 mg) by slow intravenous push (one minute in duration). EEG monitoring of the seizure may be helpful. | |
Preoperative Medication | |||
To relieve anxiety and tension. (If atropine, scopolamine or other premedications are desired, they must be administered in separate syringes.) | 10 mg, intramuscular (preferred route), before surgery. | ||
Cardioversion | |||
To relieve anxiety and tension and to reduce recall of procedure. | 5 mg to 15 mg, intravenous, within 5 to 10 minutes prior to the procedure. | ||
Once the acute symptomatology has been properly controlled with diazepam injection, the patient may be placed on oral therapy with diazepam if further treatment is required. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
Diazepam is contraindicated in patients with a known hypersensitivity to this drug; acute narrow angle glaucoma; and open angle glaucoma unless patients are receiving appropriate therapy.
ADVERSE REACTIONS
Side effects most commonly reported were drowsiness, fatigue, and ataxia; venous thrombosis and phlebitis at the site of injection. Other adverse reactions less frequently reported include:
CNS: Confusion, depression [including respiratory depressing (see WARNINGS; Pediatric Use )], dysarthria, headache, hypoactivity, slurred speech, syncope, tremor, vertigo. G.I.: Constipation, nausea. G.U.: Incontinence, changes in libido, urinary retention. Cardiovascular: Bradycardia, cardiovascular collapse, hypotension. EENT: Blurred vision, diplopia, nystagmus. Skin: Urticaria, skin rash. Other: Hiccups, changes in salivation, neutropenia, jaundice. Paradoxical reactions such as acute hyperexcited states, anxiety, hallucinations, increased muscle spasticity, insomnia, rage, sleep disturbances and stimulation have been reported; should these occur, use of the drug should be discontinued. Minor changes in EEG patterns, usually low-voltage fast activity, have been observed in patients during and after diazepam therapy and are of no known significance. In peroral endoscopic procedures, coughing, depressed respiration, dyspnea, hyperventilation, laryngospasm, and pain in throat or chest have been reported. Because of isolated reports of neutropenia and jaundice, periodic blood counts and liver function tests are advisable during long-term therapy.
Drug Interactions
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABA A sites and opioids interact primarily at mµ receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and monitor patients closely for respiratory depression and sedation. If diazepam is to be combined with other psychotropic agents or anticonvulsant drugs, careful consideration should be given to the pharmacology of the agents to be employed, particularly with known compounds which may potentiate the action of diazepam, such as phenothiazines, narcotics, barbiturates, MAO inhibitors, and other antidepressants. In highly anxious patients with evidence of accompanying depression, particularly those who may have suicidal tendencies, protective measures may be necessary. Diazepam injection has produced hypotension or muscular weakness in some patients particularly when used with narcotics, barbiturates, or alcohol. The clearance of diazepam and certain other benzodiazepines can be delayed in association with cimetidine administration. The clinical significance of this is unclear.
DESCRIPTION
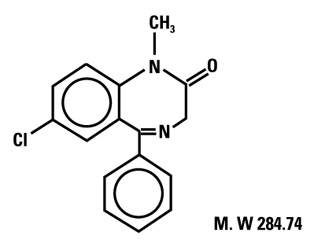
Diazepam is a benzodiazepine derivative. Chemically, diazepam is 7-chloro-1,3-dihydro-1-methyl-5- phenyl-2H-1,4-benzodiazepin-2-one. It is a colorless crystalline compound, insoluble in water, with a molecular weight of 284.74 and with the following molecular structure:
Diazepam Injection, USP is a sterile solution and each mL contains 5 mg diazepam compounded with 40% propylene glycol, 10% alcohol, 5% sodium benzoate and benzoic acid as buffers, and 1.5% benzyl alcohol as preservative.
CLINICAL PHARMACOLOGY
In animals, diazepam appears to act on parts of the limbic system, the thalamus and hypothalamus, and induces calming effects. Diazepam, unlike chlorpromazine and reserpine, has no demonstrable peripheral autonomic blocking action, nor does it produce extrapyramidal side effects; however, animals treated with diazepam do have a transient ataxia at higher doses. Diazepam was found to have transient cardiovascular depressor effects in dogs. Long-term experiments in rats revealed no disturbances of endocrine function. Injections into animals have produced localized irritation of tissue surrounding injection sites and some thickening of veins after intravenous use. Population PK analysis in 87 pediatric patients 0.4 – 17.8 years of age with status epilepticus showed that, after initial dosing, the median plasma half-life was 0.5 hours and the median terminal elimination plasma half-life was 18 to 25 hours.
HOW SUPPLIED
Diazepam Injection, USP 50 mg/10 mL is a clear, colorless to slightly yellow solution supplied in 10 mL multiple-dose vial.
Unit of Sale | Concentration | Each |
NDC 69339-137-05 Carton of 5 | 50 mg/10 mL (5 mg/mL) | NDC 69339-137-01 |
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]
Protect from light.
Diazepam - Diazepam injection, Solution PubMed™ news
- Journal Article • 2026 MayDevelopment and Assessment of Phytoconstituents-based Nanoemulsion as a Potent Combined Therapy for Neuroprotection in Scopolamine-induced Alzheimer's Disease Rat Model.
- Journal Article • 2026 MayIncreased interval between seizure clusters across time in pediatric patients treated with the immediate-use seizure medication diazepam nasal spray.
- Journal Article • 2026 MayCorrection: Efficacy, safety, route of administration of midazolam and diazepam for pediatric status epilepticus: systematic review, meta-analysis, and trial sequential analysis.
- Journal Article • 2026 MayThe autophagy-inhibitory tissue hormone DBI/ACBP is essential for glucocorticoid-induced immunosuppression.
- Journal Article • 2026 AprOn-demand seizures facilitate rapid screening of therapeutics for epilepsy.