Diflunisal - Diflunisal tablet prescribing information
Cardiovascular Risk
• Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (see WARNINGS and PRECAUTIONS ).
• Diflunisal tablets are contraindicated in the setting of coronary artery bypass graft (CABG) surgery (see CONTRAINDICATIONS and WARNINGS ).
Gastrointestinal Risk
• NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events (see WARNINGS ).
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of diflunisal tablets and other treatment options before deciding to use diflunisal tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
Diflunisal tablets are indicated for acute or long-term use for symptomatic treatment of the following:
1. Mild to moderate pain
2. Osteoarthritis
3. Rheumatoid arthritis
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of diflunisal tablets and other treatment options before deciding to use diflunisal tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
After observing the response to initial therapy with diflunisal tablets, the dose and frequency should be adjusted to suit an individual patient's needs.
Concentration-dependent pharmacokinetics prevail when diflunisal is administered; a doubling of dosage produces a greater than doubling of drug accumulation. The effect becomes more apparent with repetitive doses.
For mild to moderate pain, an initial dose of 1000 mg followed by 500 mg every 12 hours is recommended for most patients. Following the initial dose, some patients may require 500 mg every 8 hours.
A lower dosage may be appropriate depending on such factors as pain severity, patient response, weight, or advanced age; for example, 500 mg initially, followed by 250 mg every 8 to 12 hours.
For osteoarthritis and rheumatoid arthritis, the suggested dosage range is 500 mg to 1000 mg daily in two divided doses. The dosage of diflunisal may be increased or decreased according to patient response.
Maintenance doses higher than 1500 mg a day are not recommended.
Tablets should be swallowed whole, not crushed or chewed.
CONTRAINDICATIONS
Diflunisal tablets are contraindicated in patients with known hypersensitivity to diflunisal or the excipients (see DESCRIPTION ).
Diflunisal tablets should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic/analphylactoid reactions to NSAIDs have been reported in such patients (see WARNINGS , Anaphylactic/Anaphylactoid Reactions and PRECAUTIONS , Preexisting Asthma ).
Diflunisal tablets are contraindicated in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS ).
ADVERSE REACTIONS
The adverse reactions observed in controlled clinical trials encompass observations in 2,427 patients.
Listed below are the adverse reactions reported in the 1,314 of these patients who received treatment in studies of two weeks or longer. Five hundred thirteen patients were treated for at least 24 weeks, 255 patients were treated for at least 48 weeks, and 46 patients were treated for 96 weeks. In general, the adverse reactions listed below were 2 to 14 times less frequent in the 1,113 patients who received short-term treatment for mild to moderate pain.
Incidence Greater Than 1%
Gastrointestinal
The most frequent types of adverse reactions occurring with diflunisal are gastrointestinal: these include nausea•, vomiting, dyspepsia•, gastrointestinal pain•, diarrhea•, constipation, and flatulence.
Psychiatric
Somnolence, insomnia.
Central Nervous System
Dizziness.
Special Senses
Tinnitus.
Dermatologic
Rash•.
Miscellaneous
Headache•, fatigue/tiredness.
• Incidence between 3% and 9%. Those reactions occurring in 1% to 3% are not marked with an asterisk.
Incidence Less Than 1 in 100
The following adverse reactions, occurring less frequently than 1 in 100, were reported in clinical trials or since the drug was marketed. The probability exists of a causal relationship between diflunisal and these adverse reactions.
Dermatologic
Erythema multiforme, exfoliative dermatitis, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), fixed drug reaction (FDE), urticaria, pruritus, sweating, dry mucous membranes, stomatitis, and photosensitivity.
Gastrointestinal
Peptic ulcer, gastrointestinal bleeding, anorexia, eructation, gastrointestinal perforation, gastritis. Liver function abnormalities; jaundice, sometimes with fever; cholestasis; hepatitis.
Hematologic
Thrombocytopenia; agranulocytosis; hemolytic anemia.
Genitourinary
Dysuria; renal impairment, including renal failure; interstitial nephritis; hematuria; proteinuria.
Psychiatric
Nervousness, depression, hallucinations, confusion, disorientation.
Central Nervous System
Vertigo; light-headedness; paresthesias.
Special Senses
Transient visual disturbances including blurred vision.
Hypersensitivity Reactions
Acute anaphylactic reaction with bronchospasm; angioedema; flushing. Hypersensitivity vasculitis. Hypersensitivity syndrome (see PRECAUTIONS ).
Miscellaneous
Asthenia, edema.
Causal Relationship Unknown
Other reactions have been reported in clinical trials or since the drug was marketed, but occurred under circumstances where a causal relationship could not be established. However, in these rarely reported events, that possibility cannot be excluded. Therefore, these observations are listed to serve as alerting information to physicians.
Respiratory
Dyspnea.
Cardiovascular
Palpitation, syncope.
Musculoskeletal
Muscle cramps.
Genitourinary
Nephrotic syndrome.
Special Senses
Hearing loss.
Miscellaneous
Chest pain.
A rare occurrence of fulminant necrotizing fasciitis, particularly in association with Group A β -hemolytic streptococcus, has been described in persons treated with non-steroidal anti-inflammatory agents, including diflunisal, sometimes with fatal outcome (see also PRECAUTIONS, General ).
Potential Adverse Effects
In addition, a variety of adverse effects not observed with diflunisal in clinical trials or in marketing experience, but reported with other non-steroidal analgesic/anti-inflammatory agents should be considered potential adverse effects of diflunisal.
Drug Interactions
ACE-inhibitors and Angiotensin II Anagonists
Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors and angiotensin II antagonists. These interactions should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors or angiotensin II antagonists. In some patients with compromised renal function, the coadministration of an NSAID and an ACE-inhibitor or an angiotensin II antagonist may result in further deterioration of renal function, including possible acute renal failure, which is usually reversible.
Acetaminophen
In normal volunteers, concomitant administration of diflunisal and acetaminophen resulted in an approximate 50% increase in plasma levels of acetaminophen. Acetaminophen had no effect on plasma levels of diflunisal. Since acetaminophen in high doses has been associated with hepatotoxicity, concomitant administration of diflunisal tablets and acetaminophen should be used cautiously, with careful monitoring of patients.
Concomitant administration of diflunisal and acetaminophen in dogs, but not in rats, at approximately 2 times the recommended maximum human therapeutic dose of each (40 to 52 mg/kg/day of diflunisal/acetaminophen), resulted in greater gastrointestinal toxicity than when either drug was administered alone. The clinical significance of these findings has not been established.
Antacids
Concomitant administration of antacids may reduce plasma levels of diflunisal. This effect is small with occasional doses of antacids, but may be clinically significant when antacids are used on a continuous schedule.
Aspirin
When diflunisal is administered with aspirin, its protein binding is reduced, although the clearance of free diflunisal is not altered. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of diflunisal tablets and aspirin is not generally recommended because of the potential of increased adverse effects.
In normal volunteers, a small decrease in diflunisal levels was observed when multiple doses of diflunisal and aspirin were administered concomitantly.
Cyclosporine
Administration of non-steroidal anti-inflammatory drugs concomitantly with cyclosporine has been associated with an increase in cyclosporine-induced toxicity, possibly due to decreased synthesis of renal prostacyclin. NSAIDs should be used with caution in patients taking cyclosporine, and renal function should be carefully monitored.
Diuretics
Clinical studies, as well as postmarketing observations, have shown that diflunisal can reduce the natriuretic effect of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis.
In normal volunteers, concomitant administration of diflunisal and hydrochlorothiazide resulted in significantly increased plasma levels of hydrochlorothiazide. Diflunisal decreased the hyperuricemic effect of hydrochlorothiazide. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see WARNINGS , Renal Effects ), as well as to assure diuretic efficacy.
Lithium
NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Methotrexate
NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
NSAIDs
The administration of diflunisal to normal volunteers receiving indomethacin decreased the renal clearance and significantly increased the plasma levels of indomethacin. In some patients the combined use of indomethacin and diflunisal has been associated with fatal gastrointestinal hemorrhage. Therefore, indomethacin and diflunisal tablets should not be used concomitantly.
The concomitant use of diflunisal tablets and other NSAIDs is not recommended due to the increased possibility of gastrointestinal toxicity, with little or no increase in efficacy. The following information was obtained from studies in normal volunteers.
Sulindac
The concomitant administration of diflunisal and sulindac in normal volunteers resulted in lowering of the plasma levels of the active sulindac sulfide metabolite by approximately one-third.
Naproxen
The concomitant administration of diflunisal and naproxen in normal volunteers had no effect on the plasma levels of naproxen, but significantly decreased the urinary excretion of naproxen and its glucuronide metabolite. Naproxen had no effect on plasma levels of diflunisal.
Oral Anticoagulants
In some normal volunteers, the concomitant administration of diflunisal and warfarin, acenocoumarol, or phenprocoumon resulted in prolongation of prothrombin time. This may occur because diflunisal competitively displaces coumarins from protein binding sites. Accordingly, when diflunisal tablets are administered with oral anticoagulants, the prothrombin time should be closely monitored during and for several days after concomitant drug administration. Adjustment of dosage of oral anticoagulants may be required. The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
Tolbutamide
In diabetic patients receiving diflunisal and tolbutamide, no significant effects were seen on tolbutamide plasma levels or fasting blood glucose.
DESCRIPTION
Diflunisal is 2',4'-difluoro-4-hydroxy-3-biphenylcarboxylic acid. Its structural formula is:
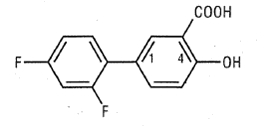
C 13 H 8 F 2 O 3 M.W. 250.20
Diflunisal is a stable, white to off-white, crystalline compound with a melting point of 211° to 213°C. It is practically insoluble in hexane and water. It is soluble in most organic solvents including acetone and ethyl acetate and it is slightly soluble in chloroform, carbon tetrachloride and methylene chloride.
Each diflunisal tablet intended for oral administration contains 500 mg of diflunisal. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hypromellose, microcrystalline cellulose, pregelatinised starch and sodium stearyl fumarate. Additionally, each diflunisal tablets contain opadry blue 03B505010 which contains FD&C blue #2, FD&C yellow #6, hypromellose, polyethylene glycol and titanium dioxide.
CLINICAL PHARMACOLOGY
Action
Diflunisal is a non-steroidal drug with analgesic, anti-inflammatory and antipyretic properties. It is a peripherally-acting non-narcotic analgesic drug. Habituation, tolerance, and addiction have not been reported.
Diflunisal is a difluorophenyl derivative of salicylic acid. Chemically, diflunisal differs from aspirin (acetylsalicylic acid) in two respects. The first of these two is the presence of a difluorophenyl substituent at carbon 1. The second difference is the removal of the O -acetyl group from the carbon 4 position. Diflunisal is not metabolized to salicylic acid, and the fluorine atoms are not displaced from the difluorophenyl ring structure.
The precise mechanism of the analgesic and anti-inflammatory actions of diflunisal is not known. Diflunisal is a prostaglandin synthetase inhibitor. In animals, prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain. Since prostaglandins are known to be among the mediators of pain and inflammation, the mode of action of diflunisal may be due to a decrease of prostaglandins in peripheral tissues.
Pharmacokinetics and Metabolism
Diflunisal is rapidly and completely absorbed following oral administration with peak plasma concentrations occurring between 2 to 3 hours. The drug is excreted in the urine as two soluble glucuronide conjugates accounting for about 90% of the administered dose. Little or no diflunisal is excreted in the feces. Diflunisal appears in human milk in concentrations of 2 to 7% of those in plasma. More than 99% of diflunisal in plasma is bound to proteins.
As is the case with salicylic acid, concentration-dependent pharmacokinetics prevail when diflunisal is administered; a doubling of dosage produces a greater than doubling of drug accumulation. The effect becomes more apparent with repetitive doses. Following single doses, peak plasma concentrations of 41 ± 11 mcg/mL (mean ± S.D.) were observed following 250 mg doses, 87 ± 17 mcg/mL were observed following 500 mg and 124 ± 11 mcg/mL following single 1000 mg doses. However, following administration of 250 mg b.i.d., a mean peak level of 56 ± 14 mcg/mL was observed on day 8, while the mean peak level after 500 mg b.i.d. for 11 days was 190 ± 33 mcg/mL. In contrast to salicylic acid which has a plasma half-life of 2 1/2 hours, the plasma half-life of diflunisal is 3 to 4 times longer (8 to 12 hours), because of a difluorophenyl substituent at carbon 1. Because of its long half-life and nonlinear pharmacokinetics, several days are required for diflunisal plasma levels to reach steady state following multiple doses. For this reason, an initial loading dose is necessary to shorten the time to reach steady-state levels, and 2 to 3 days of observation are necessary for evaluating changes in treatment regimens if a loading dose is not used.
Studies in baboons to determine passage across the blood-brain barrier have shown that only small quantities of diflunisal, under normal or acidotic conditions are transported into the cerebrospinal fluid (CSF). The ratio of blood/CSF concentrations after intravenous doses of 50 mg/kg or oral doses of 100 mg/kg of diflunisal was 100:1. In contrast, oral doses of 500 mg/kg of aspirin resulted in a blood/CSF ratio of 5:1.
Mild to Moderate Pain
Diflunisal is a peripherally-acting analgesic agent with a long duration of action. Diflunisal produces significant analgesia within 1 hour and maximum analgesia within 2 to 3 hours.
Consistent with its long half-life, clinical effects of diflunisal mirror its pharmacokinetic behavior, which is the basis for recommending a loading dose when instituting therapy. Patients treated with diflunisal, on the first dose, tend to have a slower onset of pain relief when compared with drugs achieving comparable peak effects. However, diflunisal produces longer lasting responses than the comparative agents.
Comparative single dose clinical studies have established the analgesic efficacy of diflunisal at various dose levels relative to other analgesics. Analgesic effect measurements were derived from hourly evaluations by patients during eight and twelve hour postdosing observation periods. The following information may serve as a guide for prescribing diflunisal.
Diflunisal 500 mg was comparable in analgesic efficacy to aspirin 650 mg, acetaminophen 600 mg or 650 mg, and acetaminophen 650 mg with propoxyphene napsylate 100 mg. Patients treated with diflunisal had longer lasting responses than the patients treated with the comparative analgesics.
Diflunisal 1000 mg was comparable in analgesic efficacy to acetaminophen 600 mg with codeine 60 mg. Patients treated with diflunisal had longer lasting responses than the patients who received acetaminophen with codeine.
A loading dose of 1000 mg provides faster onset of pain relief, shorter time to peak analgesic effect, and greater peak analgesic effect than an initial 500 mg dose.
In contrast to the comparative analgesics, a significantly greater proportion of patients treated with diflunisal did not remedicate and continued to have a good analgesic effect eight to twelve hours after dosing. Seventy-five percent (75%) of patients treated with diflunisal continued to have a good analgesic response at four hours. When patients having a good analgesic response at four hours were followed, 78% of these patients continued to have a good analgesic response at eight hours and 64% at twelve hours.
Chronic Anti-Inflammatory Therapy in Osteoarthritis and Rheumatoid Arthritis
In the controlled, doubleblind clinical trials in which diflunisal (500 mg to 1000 mg a day) was compared with anti-inflammatory doses of aspirin (2 to 4 grams a day), patients treated with diflunisal had a significantly lower incidence of tinnitus and of adverse effects involving the gastrointestinal system than patients treated with aspirin (see also Effect on Fecal Blood Loss ).
Osteoarthritis
The effectiveness of diflunisal for the treatment of osteoarthritis was studied in patients with osteoarthritis of the hip and/or knee. The activity of diflunisal was demonstrated by clinical improvement in the signs and symptoms of disease activity.
In a doubleblind multicenter study of 12 weeks' duration in which dosages were adjusted according to patient response, diflunisal 500 or 750 mg daily was shown to be comparable in effectiveness to aspirin 2000 or 3000 mg daily. In open-label extensions of this study to 24 or 48 weeks, diflunisal continued to show similar effectiveness and generally was well tolerated.
Rheumatoid Arthritis
In controlled clinical trials, the effectiveness of diflunisal was established for both acute exacerbations and long-term management of rheumatoid arthritis. The activity of diflunisal was demonstrated by clinical improvement in the signs and symptoms of disease activity.
In a doubleblind multicenter study of 12 weeks' duration in which dosages were adjusted according to patient response, diflunisal 500 or 750 mg daily was comparable in effectiveness to aspirin 2600 or 3900 mg daily. In open-label extensions of this study to 52 weeks, diflunisal continued to be effective and was generally well tolerated.
Diflunisal 500, 750, or 1000 mg daily was compared with aspirin 2000, 3000, or 4000 mg daily in a multicenter study of 8 weeks’ duration in which dosages were adjusted according to patient response. In this study, diflunisal was comparable in efficacy to aspirin.
In a doubleblind multicenter study of 12 weeks' duration in which dosages were adjusted according to patient needs, diflunisal 500 or 750 mg daily and ibuprofen 1600 or 2400 mg daily were comparable in effectiveness and tolerability.
In a doubleblind multicenter study of 12 weeks' duration, diflunisal 750 mg daily was comparable in efficacy to naproxen 750 mg daily. The incidence of gastrointestinal adverse effects and tinnitus was comparable for both drugs. This study was extended to 48 weeks on an open-label basis. Diflunisal continued to be effective and generally well tolerated.
In patients with rheumatoid arthritis, diflunisal and gold salts may be used in combination at their usual dosage levels. In clinical studies, diflunisal added to the regimen of gold salts usually resulted in additional symptomatic relief but did not alter the course of the underlying disease.
Antipyretic Activity
Diflunisal tablets are not recommended for use as an antipyretic agent. In single 250 mg, 500 mg, or 750 mg doses, diflunisal produced measurable but not clinically useful decreases in temperature in patients with fever; however, the possibility that it may mask fever in some patients, particularly with chronic or high doses, should be considered.
Uricosuric Effect
In normal volunteers, an increase in the renal clearance of uric acid and a decrease in serum uric acid was observed when diflunisal was administered at 500 mg or 750 mg daily in divided doses. Patients on long-term therapy taking diflunisal at 500 mg to 1000 mg daily in divided doses showed a prompt and consistent reduction across studies in mean serum uric acid levels, which were lowered as much as 1.4 mg%. It is not known whether diflunisal interferes with the activity of other uricosuric agents.
Effect on Platelet Function
As an inhibitor of prostaglandin synthetase, diflunisal has a dose-related effect on platelet function and bleeding time. In normal volunteers, 250 mg b.i.d. for 8 days had no effect on platelet function, and 500 mg b.i.d., the usual recommended dose, had a slight effect. At 1000 mg b.i.d., which exceeds the maximum recommended dosage, however, diflunisal inhibited platelet function. In contrast to aspirin, these effects of diflunisal were reversible, because of the absence of the chemically labile and biologically reactive O -acetyl group at the carbon 4 position. Bleeding time was not altered by a dose of 250 mg b.i.d., and was only slightly increased at 500 mg b.i.d. At 1000 mg b.i.d., a greater increase occurred, but was not statistically significantly different from the change in the placebo group.
Effect on Fecal Blood Loss
When diflunisal was given to normal volunteers at the usual recommended dose of 500 mg twice daily, fecal blood loss was not significantly different from placebo. Aspirin at 1000 mg four times daily produced the expected increase in fecal blood loss. Diflunisal at 1000 mg twice daily (NOTE: exceeds the recommended dosage) caused a statistically significant increase in fecal blood loss, but this increase was only one-half as large as that associated with aspirin 1300 mg twice daily.
Effect on Blood Glucose
Diflunisal did not affect fasting blood sugar in diabetic patients who were receiving tolbutamide or placebo.
HOW SUPPLIED
Diflunisal Tablets USP, 500 mg are light blue to blue color, capsule shaped, biconvex, coated tablets, debossed with "530" on one side and plain on the other side and are supplied as follows:
NDC 70771-1083-3 in bottle of 30 tablets
NDC 70771-1083-6 in bottle of 60 tablets
NDC 70771-1083-9 in bottle of 90 tablets
NDC 70771-1083-1 in bottle of 100 tablets
NDC 70771-1083-5 in bottle of 500 tablets
NDC 70771-1083-0 in bottle of 1000 tablets
NDC 70771-1083-8 in unit-dose blister cartons of 100 (10 X 10) unit-dose tablets
Storage
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight container (USP).
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Manufactured by:
Zydus Lifesciences Limited.,
Baddi-173205, India
Rev.: 07/24