Get your patient on Dihydroergotamine Mesylate - Dihydroergotamine Mesylate injection, Solution (Dihydroergotamine Mesylate)
Dihydroergotamine Mesylate - Dihydroergotamine Mesylate injection, Solution prescribing information
WARNING: PERIPHERAL ISCHEMIA FOLLOWING COADMINISTRATION WITH POTENT CYP3A4 INHIBITORS
Serious and/or life-threatening peripheral ischemia has been associated with the coadministration of dihydroergotamine with potent CYP3A4 inhibitors including protease inhibitors and macrolide antibiotics. Because CYP3A4 inhibition elevates the serum levels of dihydroergotamine, the risk for vasospasm leading to cerebral ischemia and/or ischemia of the extremities is increased. Hence, concomitant use of these medications is contraindicated (see also CONTRAINDICATIONS and WARNINGS ).
INDICATIONS AND USAGE
Dihydroergotamine Mesylate Injection, USP is indicated for the acute treatment of migraine headaches with or without aura and the acute treatment of cluster headache episodes.
DOSAGE AND ADMINISTRATION
Dihydroergotamine Mesylate Injection, USP should be administered in a dose of 1 mL intravenously, intramuscularly or subcutaneously. The dose can be repeated, as needed, at 1-hour intervals to a total dose of 3 mL for intramuscular or subcutaneous delivery or 2 mL for intravenous delivery in a 24-hour period. The total weekly dosage should not exceed 6 mL. Dihydroergotamine Mesylate Injection, USP should not be used for chronic daily administration.
CONTRAINDICATIONS
There have been a few reports of serious adverse events associated with the coadministration of dihydroergotamine and potent CYP3A4 inhibitors, such as protease inhibitors and macrolide antibiotics, resulting in vasospasm that led to cerebral ischemia and/or ischemia of the extremities. The use of potent CYP3A4 inhibitors (i.e., ritonavir, nelfinavir, indinavir, erythromycin, clarithromycin, troleandomycin, ketoconazole, itraconazole) with dihydroergotamine is, therefore, contraindicated (see WARNINGS, CYP3A4 Inhibitors ).
Dihydroergotamine Mesylate Injection, USP should not be given to patients with ischemic heart disease (e.g., angina pectoris, history of myocardial infarction, or documented silent ischemia) or to patients who have clinical symptoms or findings consistent with coronary artery vasospasm including Prinzmetal's variant angina (see WARNINGS ).
Because Dihydroergotamine Mesylate Injection, USP may increase blood pressure, it should not be given to patients with uncontrolled hypertension.
Dihydroergotamine Mesylate Injection, USP, 5-HT 1 agonists (e.g., sumatriptan), ergotamine-containing or ergot-type medications or methysergide should not be used within 24 hours of each other.
Dihydroergotamine Mesylate Injection, USP should not be administered to patients with hemiplegic or basilar migraine.
In addition to those conditions mentioned above, Dihydroergotamine Mesylate Injection, USP is also contraindicated in patients with known peripheral arterial disease, sepsis, following vascular surgery and severely impaired hepatic or renal function.
Dihydroergotamine Mesylate Injection, USP is contraindicated in patients who have previously shown hypersensitivity to ergot alkaloids.
Dihydroergotamine mesylate should not be used with peripheral and central vasoconstrictors because the combination may result in additive or synergistic elevation of blood pressure.
ADVERSE REACTIONS
Serious cardiac events, including some that have been fatal, have occurred following use of Dihydroergotamine Mesylate Injection, USP, but are extremely rare. Events reported have included coronary artery vasospasm, transient myocardial ischemia, myocardial infarction, ventricular tachycardia, and ventricular fibrillation (see CONTRAINDICATIONS , WARNINGS , and PRECAUTIONS ). Fibrotic complications have been reported in association with long term use of injectable dihydroergotamine mesylate (see WARNINGS, Fibrotic Complications ).
Post-introduction Reports
The following events derived from postmarketing experience have been occasionally reported in patients receiving Dihydroergotamine Mesylate Injection, USP: vasospasm, paraesthesia, hypertension, dizziness, anxiety, dyspnea, headache, flushing, diarrhea, rash, increased sweating, and pleural and retroperitoneal fibrosis after long-term use of dihydroergotamine. Extremely rare cases of myocardial infarction and stroke have been reported. A causal relationship has not been established.
Dihydroergotamine Mesylate Injection, USP is not recommended for prolonged daily use (see DOSAGE AND ADMINISTRATION ).
To report SUSPECTED ADVERSE REACTIONS, contact Provepharm Inc., at 1–833-727-6556 or safety-us@provepharm.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Vasoconstrictors
Dihydroergotamine Mesylate Injection, USP should not be used with peripheral vasoconstrictors because the combination may cause synergistic elevation of blood pressure.
Sumatriptan
Sumatriptan has been reported to cause coronary artery vasospasm, and its effect could be additive with Dihydroergotamine Mesylate Injection, USP. Sumatriptan and Dihydroergotamine Mesylate Injection, USP should not be taken within 24 hours of each other (see CONTRAINDICATIONS ).
Beta Blockers
Although the results of a clinical study did not indicate a safety problem associated with the administration of Dihydroergotamine Mesylate Injection, USP to subjects already receiving propranolol, there have been reports that propranolol may potentiate the vasoconstrictive action of ergotamine by blocking the vasodilating property of epinephrine.
Nicotine
Nicotine may provoke vasoconstriction in some patients, predisposing to a greater ischemic response to ergot therapy.
CYP3A4 Inhibitors (e.g., Macrolide Antibiotics and Protease Inhibitors) (see CONTRAINDICATIONS and WARNINGS )
SSRI's
Weakness, hyperreflexia, and incoordination have been reported rarely when 5-HT 1 agonists have been co-administered with SSRI's (e.g., fluoxetine, fluvoxamine, paroxetine, sertraline). There have been no reported cases from spontaneous reports of drug interaction between SSRI's and Dihydroergotamine Mesylate Injection, USP.
Oral Contraceptives
The effect of oral contraceptives on the pharmacokinetics of Dihydroergotamine Mesylate Injection, USP has not been studied.
DESCRIPTION
Dihydroergotamine Mesylate Injection, USP is ergotamine hydrogenated in the 9, 10 position as the mesylate salt. Dihydroergotamine Mesylate Injection, USP is known chemically as ergotaman-3´,6´,18-trione,9,10-dihydro-12´-hydroxy-2´-methyl-5´-(phenylmethyl)-,(5´α)-, monomethanesulfonate. Its molecular weight is 679.80 and its empirical formula is C 33 H 37 N 5 O 5 •CH 4 O 3 S.
The chemical structure is:
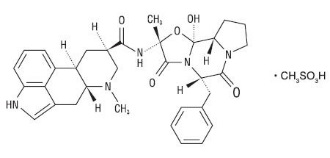
Dihydroergotamine mesylate C 33 H 37 N 5 O 5 •CH 4 O 3 S Mol. Wt. 679.80 |
Dihydroergotamine Mesylate Injection, USP is a clear, colorless solution supplied in sterile ampules for intravenous, intramuscular, or subcutaneous administration. Each mL contains 1 mg Dihydroergotamine Mesylate, USP; Alcohol, USP 6.1% by volume; Glycerin, USP 15% by weight; Water for Injection, USP; Methanesulfonic Acid and/or Sodium Hydroxide for pH adjustment (pH range is 3.4 to 4.9).
CLINICAL PHARMACOLOGY
Mechanism of Action
Dihydroergotamine binds with high affinity to 5-HT 1D α and 5-HT 1D β receptors. It also binds with high affinity to serotonin 5-HT 1A , 5-HT 2A , and 5-HT 2C receptors, noradrenaline α 2A , α 2B and α 1 receptors, and dopamine D 2L and D 3 receptors.
The therapeutic activity of dihydroergotamine in migraine is generally attributed to the agonist effect at 5-HT 1D receptors. Two current theories have been proposed to explain the efficacy of 5-HT 1D receptor agonists in migraine. One theory suggests that activation of 5-HT 1D receptors located on intracranial blood vessels, including those on arteriovenous anastomoses, leads to vasoconstriction, which correlates with the relief of migraine headache. The alternative hypothesis suggests that activation of 5-HT 1D receptors on sensory nerve endings of the trigeminal system results in the inhibition of pro-inflammatory neuropeptide release.
In addition, dihydroergotamine possesses oxytocic properties.
Pharmacokinetics
Absorption
Absolute bioavailability for the subcutaneous and intramuscular route have not been determined; however, no difference was observed in dihydroergotamine bioavailability from intramuscular and subcutaneous doses.
Dihydroergotamine mesylate is poorly bioavailable following oral administration.
Distribution
Dihydroergotamine mesylate is 93% plasma protein bound. The apparent steady-state volume of distribution is approximately 800 liters.
Metabolism
Four dihydroergotamine mesylate metabolites have been identified in human plasma following oral administration. The major metabolite, 8´-β-hydroxydihydroergotamine, exhibits affinity equivalent to its parent for adrenergic and 5-HT receptors and demonstrates equivalent potency in several venoconstrictor activity models, in vivo and in vitro. The other metabolites, (i.e., dihydrolysergic acid, dihydrolysergic amide) and a metabolite formed by oxidative opening of the proline ring are of minor importance. Following nasal administration, total metabolites represent only 20% to 30% of plasma AUC. Quantitative pharmacokinetic characterization of the four metabolites has not been performed.
Excretion
The major excretory route of dihydroergotamine is via the bile in the feces. The total body clearance is 1.5 L/min which reflects mainly hepatic clearance. Only 6% to 7% of unchanged dihydroergotamine is excreted in the urine after intramuscular injection. The renal clearance (0.1 L/min) is unaffected by the route of dihydroergotamine administration. The decline of plasma dihydroergotamine after intramuscular or intravenous administration is multi-exponential with a terminal half-life of about 9 hours.
Subpopulations
No studies have been conducted on the effect of renal or hepatic impairment, gender, race, or ethnicity on dihydroergotamine pharmacokinetics. Dihydroergotamine Mesylate Injection, USP is contraindicated in patients with severely impaired hepatic or renal function (see CONTRAINDICATIONS ).
Interactions
Pharmacokinetic interactions have been reported in patients treated orally with other ergot alkaloids (e.g., increased levels of ergotamine) and macrolide antibiotics, principally troleandomycin, presumably due to inhibition of cytochrome P4503A metabolism of the alkaloids by troleandomycin.
Dihydroergotamine has also been shown to be an inhibitor of cytochrome P4503A catalyzed reactions and rare reports of ergotism have been obtained from patients treated with dihydroergotamine and macrolide antibiotics (e.g., troleandomycin, clarithromycin, erythromycin), and in patients treated with dihydroergotamine and protease inhibitors (e.g., ritonavir), presumably due to inhibition of cytochrome P4503A metabolism of ergotamine (see CONTRAINDICATIONS ). No pharmacokinetic interactions involving other cytochrome P450 isoenzymes are known.
HOW SUPPLIED/STORAGE AND HANDLING
Dihydroergotamine Mesylate Injection, USP
Available as a clear, colorless, sterile solution in single 1 mL sterile ampules containing 1 mg of dihydroergotamine mesylate per mL, in packages of 5 (NDC 81284-411-05) and 10 (NDC 81284-411-10).
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Use carton to protect contents from light until used.
Do not refrigerate or freeze.
To assure constant potency, protect the ampules from light and heat. Administer only if clear and colorless.
•Trademark of PDR Network, LLC
Manufactured for: Provepharm SAS
Distributed by: Provepharm, Inc. Collegeville, PA 19426
Made in France
Revised: 11/2022
Mechanism of Action
Dihydroergotamine binds with high affinity to 5-HT 1D α and 5-HT 1D β receptors. It also binds with high affinity to serotonin 5-HT 1A , 5-HT 2A , and 5-HT 2C receptors, noradrenaline α 2A , α 2B and α 1 receptors, and dopamine D 2L and D 3 receptors.
The therapeutic activity of dihydroergotamine in migraine is generally attributed to the agonist effect at 5-HT 1D receptors. Two current theories have been proposed to explain the efficacy of 5-HT 1D receptor agonists in migraine. One theory suggests that activation of 5-HT 1D receptors located on intracranial blood vessels, including those on arteriovenous anastomoses, leads to vasoconstriction, which correlates with the relief of migraine headache. The alternative hypothesis suggests that activation of 5-HT 1D receptors on sensory nerve endings of the trigeminal system results in the inhibition of pro-inflammatory neuropeptide release.
In addition, dihydroergotamine possesses oxytocic properties.
Dihydroergotamine Mesylate - Dihydroergotamine Mesylate injection, Solution PubMed™ news
- Journal Article • 2026 MayTotal Synthesis of 8-Hydroxy-dihydroergotamine, the Major Human Metabolite of Dihydroergotamine.
- Journal Article • 2026 AprHealth Care Utilization in Refractory Migraine: A Cross-Sectional Analysis of a Cross-Institutional Electronic Health Care Records Database.
- Journal Article • 2026 AprGene-guided repurposing identifies dihydroergotamine as a candidate inhibitor of the BCL2-SIVA1 axis in advanced gastric cancer in vitro.
- Journal Article • 2026 MarDHE - past, present, and future: a narrative review.
- Journal Article • 2026 FebIntegrative analysis of transcriptome and single-cell sequencing combined with experimental validation identifies biomarkers associated with T cell and senescence in sepsis.