Get your patient on Diphenoxylate Hydrochloride And Atropine Sulfate - Diphenoxylate Hydrochloride And Atropine Sulfate tablet (Diphenoxylate Hydrochloride And Atropine Sulfate)
Diphenoxylate Hydrochloride And Atropine Sulfate - Diphenoxylate Hydrochloride And Atropine Sulfate tablet prescribing information
INDICATIONS AND USAGE
Diphenoxylate hydrochloride and atropine sulfate tablets are indicated as adjunctive therapy in the management of diarrhea in patients 13 years of age and older.
DOSAGE AND ADMINISTRATION
Management of Diarrhea in Patients 13 Years of Age and Older
Diphenoxylate hydrochloride and atropine sulfate tablets are recommended as adjunctive therapy for the management of diarrhea in patients 13 years of age and older. Consider the nutritional status and degree of dehydration in patients prior to initiating therapy with diphenoxylate hydrochloride and atropine sulfate tablets. The use of diphenoxylate hydrochloride and atropine sulfate tablets should be accompanied by appropriate fluid and electrolyte therapy, when indicated. If severe dehydration or electrolyte imbalance is present, do not administer diphenoxylate hydrochloride and atropine sulfate tablets until appropriate corrective therapy has been indicated (see WARNINGS ).
Initial and Maximum Recommended Dosage in Patients 13 Years of Age and Older
The initial adult dosage is 2 diphenoxylate hydrochloride and atropine sulfate tablets four times daily (maximum total daily dose of 20 mg per day of diphenoxylate hydrochloride). Most patients will require this dosage until initial control of diarrhea has been achieved. Clinical improvement of acute diarrhea is usually observed within 48 hours.
Dosage after Initial Control of Diarrhea
After initial control has been achieved, the diphenoxylate hydrochloride and atropine sulfate tablets dosage may be reduced to meet individual requirements. Control may often be maintained with as little as two diphenoxylate hydrochloride and atropine sulfate tablets daily.
Duration of Treatment
If clinical improvement of chronic diarrhea after treatment with the maximum recommended daily dosage is not observed within 10 days, discontinue diphenoxylate hydrochloride and atropine sulfate tablets as symptoms are unlikely to be controlled by further administration.
CONTRAINDICATIONS
Diphenoxylate hydrochloride and atropine sulfate tablets are contraindicated in:
- Pediatric patients less than 6 years of age due to the risks of respiratory and central nervous system (CNS) depression (see WARNINGS ).
- Patients with diarrhea associated with pseudomembranous enterocolitis ( Clostridium difficile ) or other enterotoxin-producing bacteria due to the risk of gastrointestinal (GI) complications, including sepsis (see WARNINGS ).
- Patients with known hypersensitivity to diphenoxylate or atropine.
- Patients with obstructive jaundice.
ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in labeling:
- Respiratory and/or CNS depression (see WARNINGS )
- Anticholinergic and opioid-toxicities, including atropinism (see WARNINGS and PRECAUTIONS )
- Dehydration and electrolyte imbalance (see WARNINGS )
- GI Complications in patients with infectious diarrhea (see WARNINGS )
- Toxic megacolon in patients with acute ulcerative colitis (see WARNINGS )
At therapeutic doses of diphenoxylate hydrochloride and atropine sulfate tablets, the following other adverse reactions have been reported; they are listed in decreasing order of severity, but not of frequency:
Nervous system: numbness of extremities, euphoria, depression, malaise/lethargy, confusion, sedation/drowsiness, dizziness, restlessness, headache, hallucination
Allergic: anaphylaxis, angioneurotic edema, urticaria, swelling of the gums, pruritus
Gastrointestinal system: megacolon, paralytic ileus, pancreatitis, vomiting, nausea, anorexia, abdominal discomfort
The following adverse reactions related to atropine sulfate are listed in decreasing order of severity, but not of frequency: hyperthermia, tachycardia, urinary retention, flushing, dryness of the skin and mucous membranes.
To report SUSPECTED ADVERSE REACTIONS, contact Unichem Pharmaceuticals (USA) Inc. at 1-866-562-4616 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug interactions:
Alcohol
Alcohol may increase the CNS depressant effects of diphenoxylate hydrochloride and atropine sulfate tablets and may cause drowsiness (see WARNINGS ). Avoid concomitant use of diphenoxylate hydrochloride and atropine sulfate tablets with alcohol.
Other Drugs that Cause CNS Depression
The concurrent use of diphenoxylate hydrochloride and atropine sulfate tablets with other drugs that cause CNS depression (e.g., barbiturates, benzodiazepines, opioids, buspirone, antihistamines, muscle relaxants), may potentiate the effects of diphenoxylate hydrochloride and atropine sulfate tablets (see WARNINGS ). Either diphenoxylate hydrochloride and atropine sulfate tablets or the other interacting drug should be chosen, depending on the importance of the drug to the patient. If CNS-acting drugs cannot be avoided, monitor patients for CNS adverse reactions.
MAO Inhibitors
Diphenoxylate may interact with monoamine oxidase inhibitors (MAOIs) and precipitate a hypertensive crisis . Avoid use of diphenoxylate hydrochloride and atropine sulfate tablets in patients who take MAOIs and monitor for signs and symptoms of hypertensive crisis (headache, hyperthermia, hypertension).
DESCRIPTION
Each diphenoxylate hydrochloride and atropine sulfate tablet contains:
2.5 mg of diphenoxylate hydrochloride USP (equivalent to 2.3 mg of diphenoxylate) and 0.025 mg of atropine sulfate USP (equivalent to 0.01 mg of atropine)
Diphenoxylate hydrochloride, an antidiarrheal, is ethyl 1-(3-cyano-3,3-diphenylpropyl)-4-phenylisonipecotate monohydrochloride and has the following structural formula:
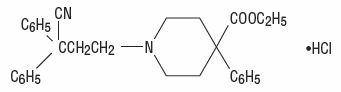
Atropine sulfate, an anticholinergic, is endo-(±)-α-(hydroxymethyl) benzeneacetic acid 8-methyl-8-azabicyclo[3.2.1] oct-3-yl ester sulfate (2:1) (salt) monohydrate and has the following structural formula:
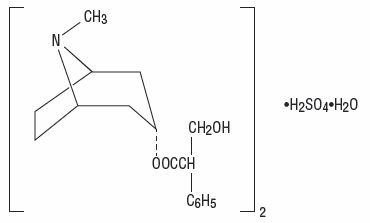
A subtherapeutic amount of atropine sulfate is present to discourage deliberate overdosage.
Inactive ingredients of diphenoxylate hydrochloride and atropine sulfate tablets include mannitol, pregelatinized starch, hypromellose, colloidal silicon dioxide, magnesium stearate
CLINICAL PHARMACOLOGY
Diphenoxylate is rapidly and extensively metabolized in man by ester hydrolysis to diphenoxylic acid (difenoxine), which is biologically active and the major metabolite in the blood. After a 5-mg oral dose of carbon-14 labeled diphenoxylate hydrochloride in ethanolic solution was given to three healthy volunteers, an average of 14% of the drug plus its metabolites was excreted in the urine and 49% in the feces over a four-day period. Urinary excretion of the unmetabolized drug constituted less than 1% of the dose, and diphenoxylic acid plus its glucuronide conjugate constituted about 6% of the dose. In a 16-subject crossover bioavailability study, a linear relationship in the dose range of 2.5 to 10 mg was found between the dose of diphenoxylate hydrochloride (given as diphenoxylate hydrochloride and atropine sulfate liquid) and the peak plasma concentration, the area under the plasma concentration-time curve, and the amount of diphenoxylic acid excreted in the urine. In the same study the bioavailability of the tablet compared with an equal dose of the liquid was approximately 90%. The average peak plasma concentration of diphenoxylic acid following ingestion of four 2.5-mg tablets was 163 ng/ml at about 2 hours, and the elimination half-life of diphenoxylic acid was approximately 12 to 14 hours.
In dogs, diphenoxylate hydrochloride has a direct effect on circular smooth muscle of the bowel that conceivably results in segmentation and prolongation of gastrointestinal transit time. The clinical antidiarrheal action of diphenoxylate hydrochloride may thus be a consequence of enhanced segmentation that allows increased contact of the intraluminal contents with the intestinal mucosa.
HOW SUPPLIED
Tablets — round, off-white to white tablet with BY3 on one side; and plain on the other side and containing 2.5 mg of diphenoxylate hydrochloride and 0.025 mg of atropine sulfate, supplied as:
| NDC Number | Size |
| 76385-107-01 | bottle of 100 |
| 76385-107-10 | bottle of 1000 |
Store below 25°C (77°F).
Rx only
MADE IN USA
Distributed by:
UNICHEM PHARMACEUTICALS (USA), INC.
East Brunswick, NJ 08816 USA.
400035
Rev. 10/2025