Get your patient on Disulfiram - Disulfiram tablet (Disulfiram)
Disulfiram - Disulfiram tablet prescribing information
WARNING
Disulfiram should never be administered to a patient when he is in a state of alcohol intoxication, or without his full knowledge. The physician should instruct relatives accordingly.
INDICATIONS AND USAGE
Disulfiram is an aid in the management of selected chronic alcohol patients who want to remain in a state of enforced sobriety so that supportive and psychotherapeutic treatment may be applied to best advantage.
Disulfiram is not a cure for alcoholism. When used alone, without proper motivation and supportive therapy, it is unlikely that it will have any substantive effect on the drinking pattern of the chronic alcoholic.
DOSAGE AND ADMINISTRATION
Disulfiram should never be administered until the patient has abstained from alcohol for at least 12 hours.
Initial Dosage Schedule
In the first phase of treatment, a maximum of 500 mg daily is given in a single dose for one to two weeks. Although usually taken in the morning, disulfiram may be taken on retiring by patients who experience a sedative effect. Alternatively, to minimize, or eliminate, the sedative effect, dosage may be adjusted downward.
Maintenance Regimen
The average maintenance dose is 250 mg daily (range, 125 mg to 500 mg), it should not exceed 500 mg daily.
Note: Occasionally patients, while seemingly on adequate maintenance doses of disulfiram, report that they are able to drink alcoholic beverages with impunity and without any symptomatology. All appearances to the contrary, such patients must be presumed to be disposing of their tablets in some manner without actually taking them. Until such patients have been observed reliably taking their daily disulfiram tablets (preferably crushed and well mixed with liquid), it cannot be concluded that disulfiram is ineffective.
Duration of Therapy
The daily, uninterrupted administration of disulfiram must be continued until the patient is fully recovered socially and a basis for permanent self-control is established. Depending on the individual patient, maintenance therapy may be required for months or even years.
Trial with Alcohol
During early experience with disulfiram, it was thought advisable for each patient to have at least one supervised alcohol-drug reaction. More recently, the test reaction has been largely abandoned. Furthermore, such a test reaction should never be administered to a patient over 50 years of age. A clear, detailed and convincing description of the reaction is felt to be sufficient in most cases.
However, where a test reaction is deemed necessary, the suggested procedure is as follows: After the first one to two weeks' therapy with 500 mg daily, a drink of 15 mL (1/2 oz) of 100 proof whiskey, or equivalent, is taken slowly. This test dose of alcoholic beverage may be repeated once only, so that the total dose does not exceed 30 mL (1 oz) of whiskey. Once a reaction develops, no more alcohol should be consumed. Such tests should be carried out only when the patient is hospitalized, or comparable supervision and facilities, including oxygen, are available.
Management of Disulfiram-Alcohol Reaction
In severe reactions, whether caused by an excessive test dose or by the patient's unsupervised ingestion of alcohol, supportive measures to restore blood pressure and treat shock should be instituted. Other recommendations include: oxygen, carbogen (95% oxygen and 5% carbon dioxide), vitamin C intravenously in massive doses (1 g) and ephedrine sulfate. Antihistamines have also been used intravenously. Potassium levels should be monitored, particularly in patients on digitalis, since hypokalemia has been reported.
CONTRAINDICATIONS
Patients who are receiving or have recently received metronidazole, paraldehyde, alcohol, or alcohol-containing preparations, e.g., cough syrups, tonics and the like, should not be given disulfiram.
Disulfiram is contraindicated in the presence of severe myocardial disease or coronary occlusion, psychoses, and hypersensitivity to disulfiram or to other thiuram derivatives used in pesticides and rubber vulcanization.
ADVERSE REACTIONS
(See CONTRAINDICATIONS , WARNINGS , and PRECAUTIONS .)
OPTIC NEURITIS, PERIPHERAL NEURITIS, POLYNEURITIS, AND PERIPHERAL NEUROPATHY MAY OCCUR FOLLOWING ADMINISTRATION OF DISULFIRAM.
Multiple cases of hepatitis, including both cholestatic and fulminant hepatitis, as well as hepatic failure resulting in transplantation or death, have been reported with administration of disulfiram.
Occasional skin eruptions are, as a rule, readily controlled by concomitant administration of an antihistaminic drug.
In a small number of patients, a transient mild drowsiness, fatigability, impotence, headache, acneform eruptions, allergic dermatitis, or a metallic or garlic-like aftertaste may be experienced during the first two weeks of therapy. These complaints usually disappear spontaneously with the continuation of therapy, or with reduced dosage.
Psychotic reactions have been noted, attributable in most cases to high dosage, combined toxicity (with metronidazole or isoniazid), or to the unmasking of underlying psychoses in patients stressed by the withdrawal of alcohol.
To report SUSPECTED ADVERSE REACTIONS, contact Alvogen, Inc. at 1-866-770-3024 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Disulfiram appears to decrease the rate at which certain drugs are metabolized and therefore may increase the blood levels and the possibility of clinical toxicity of drugs given concomitantly.
DISULFIRAM SHOULD BE USED WITH CAUTION IN THOSE PATIENTS RECEIVING PHENYTOIN AND ITS CONGENERS, SINCE THE CONCOMITANT ADMINISTRATION OF THESE TWO DRUGS CAN LEAD TO PHENYTOIN INTOXICATION. PRIOR TO ADMINISTERING DISULFIRAM TO A PATIENT ON PHENYTOIN THERAPY, A BASELINE PHENYTOIN SERUM LEVEL SHOULD BE OBTAINED. SUBSEQUENT TO INITIATION OF DISULFIRAM THERAPY, SERUM LEVELS OF PHENYTOIN SHOULD BE DETERMINED ON DIFFERENT DAYS FOR EVIDENCE OF AN INCREASE OR FOR A CONTINUING RISE IN LEVELS. INCREASED PHENYTOIN LEVELS SHOULD BE TREATED WITH APPROPRIATE DOSAGE ADJUSTMENT.
It may be necessary to adjust the dosage of oral anticoagulants upon beginning or stopping disulfiram, since disulfiram may prolong prothrombin time.
Patients taking isoniazid when disulfiram is given should be observed for the appearance of unsteady gait or marked changes in mental status, the disulfiram should be discontinued if such signs appear.
In rats, simultaneous ingestion of disulfiram and nitrite in the diet for 78 weeks has been reported to cause tumors, and it has been suggested that disulfiram may react with nitrites in the rat stomach to form a nitrosamine, which is tumorigenic. Disulfiram alone in the rat's diet did not lead to such tumors. The relevance of this finding to humans is not known at this time.
DESCRIPTION
Disulfiram is an alcohol antagonist drug.
CHEMICAL NAME:
bis(diethylthiocarbamoyl) disulfide.
STRUCTURAL FORMULA:
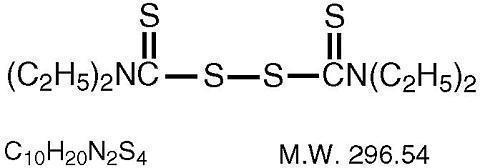
Disulfiram occurs as a white to off-white, odorless, and almost tasteless powder, soluble in water to the extent of about 20 mg in 100 mL, and in alcohol to the extent of about 3.8 g in 100 mL.
Each tablet for oral administration contains 250 mg disulfiram, USP. Tablets also contain colloidal silicon dioxide, anhydrous lactose, magnesium stearate, microcrystalline cellulose, sodium starch glycolate, and stearic acid.
CLINICAL PHARMACOLOGY
Disulfiram produces a sensitivity to alcohol which results in a highly unpleasant reaction when the patient under treatment ingests even small amounts of alcohol.
Disulfiram blocks the oxidation of alcohol at the acetaldehyde stage. During alcohol metabolism following disulfiram intake, the concentration of acetaldehyde occurring in the blood may be 5 to 10 times higher than that found during metabolism of the same amount of alcohol alone.
Accumulation of acetaldehyde in the blood produces a complex of highly unpleasant symptoms referred to hereinafter as the disulfiram-alcohol reaction. This reaction, which is proportional to the dosage of both disulfiram and alcohol, will persist as long as alcohol is being metabolized. Disulfiram does not appear to influence the rate of alcohol elimination from the body.
Disulfiram is absorbed slowly from the gastrointestinal tract and is eliminated slowly from the body. One (or even two) weeks after a patient has taken his last dose of disulfiram, ingestion of alcohol may produce unpleasant symptoms.
Prolonged administration of disulfiram does not produce tolerance; the longer a patient remains on therapy, the more exquisitely sensitive he becomes to alcohol.
HOW SUPPLIED
Disulfiram Tablets, USP are white, round, unscored tablets debossed 607.
Bottle of 30 NDC 47781-607-30
Dispense in a tight, light-resistant container as defined in the USP.
Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature].
Manufactured by: Norwich Pharmaceuticals, Inc. Norwich, NY 13815 USA
Distributed by: Alvogen, Inc. Morristown, NJ 07960 USA
PI607-03 Rev. 01/2020 PKG02698
Disulfiram - Disulfiram tablet PubMed™ news
- Journal Article • 2026 MayHarnessing Cuproptosis for Antitumor Immunity: A Review of Current Evidence and Therapeutic Strategies.
- Journal Article • 2026 MayIntegrated Bulk and Single-Cell Transcriptomic Analysis Reveals Xenobiotic Metabolism Genes Drive Progression From Liver Cirrhosis to Hepatocellular Carcinoma.
- Journal Article • 2026 MayProbable case of anaphylaxis due to hydrocortisone in a septic shock patient.
- Journal Article • 2026 MayStress-inducible circSCLT1 scaffolds HNRNPA1/CSDE1 to amplify IL1B/NF-κB signaling in TNBC via mRNA stabilization, suppressor degradation, and stress granule shielding.
- Journal Article • 2026 AprRevisiting disulfiram-like reactions with antibiotics: evidence, misattribution, and the need for updated clinical guidance.