Get your patient on Dolishale - Levonorgestrel And Ethinyl Estradiol tablet (Levonorgestrel And Ethinyl Estradiol)
Dolishale - Levonorgestrel And Ethinyl Estradiol tablet prescribing information
Cigarette smoking increases the risk of serious cardiovascular side effects from oral contraceptive use. This risk increases with age and with the extent of smoking (in epidemiologic studies, 15 or more cigarettes per day was associated with a significantly increased risk) and is quite marked in women over 35 years of age. Women who use oral contraceptives should be strongly advised not to smoke.
INDICATIONS AND USAGE
DOLISHALE is indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Oral contraceptives are highly effective for pregnancy prevention. Table 2 lists the typical unintended pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization, the IUD, and implants, depend upon the reliability with which they are used. Correct and consistent use of methods can result in lower failure rates.
Table 2: Percentage of Women Experiencing an Unintended Pregnancy During The First Year of Typical Use and The First Year of Perfect Use of Contraception and The Percentage Continuing Use at The End of the First Year. United States.
| % of Women Experiencing an Unintended Pregnancy within the First Year of Use | % of Women Continuing Use at One Year 3 | ||
| Method (1) | Typical Use 1 (2) | Perfect Use 2 (3) | (4) |
| Chance 4 | 85 | 85 | |
| Spermicides 5 | 26 | 6 | 40 |
| Periodic abstinence | 25 | 63 | |
| Calendar | 9 | ||
| Ovulation Method | 3 | ||
| Sympto-Thermal 6 | 2 | ||
| Post-Ovulation | 1 | ||
| Cap 7 | |||
| Parous Women | 40 | 26 | 42 |
| Nulliparous Women | 20 | 9 | 56 |
| Sponge | |||
| Parous Women | 40 | 20 | 42 |
| Nulliparous Women | 20 | 9 | 56 |
| Diaphragm 7 | 20 | 6 | 56 |
| Withdrawal | 19 | 4 | |
| Condom 8 | |||
| Female (Reality™) | 21 | 5 | 56 |
| Male | 14 | 3 | 61 |
| Pill | 5 | 71 | |
| Progestin only | 0.5 | ||
| Combined | 0.1 | ||
| IUD | |||
| Progesterone T | 2.0 | 1.5 | 81 |
| Copper T380A | 0.8 | 0.6 | 78 |
| LNg 20 | 0.1 | 0.1 | 81 |
| Depo-Provera ® | 0.3 | 0.3 | 70 |
| Levonorgestrel Implants (Norplant ® ) | 0.05 | 0.05 | 88 |
| Female Sterilization | 0.5 | 0.5 | 100 |
| Male Sterilization | 0.15 | 0.10 | 100 |
Emergency Contraceptive Pills: The FDA has concluded that certain combined oral contraceptives containing ethinyl estradiol and norgestrel or levonorgestrel are safe and effective for use as postcoital emergency contraception. Treatment initiated within 72 hours after unprotected intercourse reduces the risk of pregnancy by at least 75%. 9
Lactation Amenorrhea Method: LAM is a highly effective, temporary method of contraception. 10
Source: Trussell J. Contraceptive efficacy. In: Hatcher RA, Trussell J, Stewart F, Cates W, Stewart GK, Kowel D, Guest F. Contraceptive Technology: Seventeenth Revised Edition. New York NY: Irvington Publishers; 1998.
1. Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
2. Among couples who initiate use of a method (not necessarily for the first time) and who use it perfectly (both consistently and correctly), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
3. Among couples attempting to avoid pregnancy, the percentage who continue to use a method for one year.
4. The percents becoming pregnant in columns (2) and (3) are based on data from populations where contraception is not used and from women who cease using contraception in order to become pregnant. Among such populations, about 89% become pregnant within one year. This estimate was lowered slightly (to 85%) to represent the percent who would become pregnant within one year among women now relying on reversible methods of contraception if they abandoned contraception altogether.
5. Foams, creams, gels, vaginal suppositories, and vaginal film.
6. Cervical mucus (ovulation) method supplemented by calendar in the pre-ovulatory and basal body temperature in the post-ovulatory phases.
7. With spermicidal cream or jelly.
8. Without spermicides.
9. The treatment schedule is one dose within 72 hours after unprotected intercourse, and a second dose 12 hours after the first dose. The FDA has declared the following dosage regimens of oral contraceptives to be safe and effective for emergency contraception: for tablets containing 50 mcg of ethinyl estradiol and 500 mcg of norgestrel 1 dose is 2 tablets; for tablets containing 20 mcg of ethinyl estradiol and 100 mcg of levonorgestrel 1 dose is 5 tablets; for tablets containing 30 mcg of ethinyl estradiol and 150 mcg of levonorgestrel 1 dose is 4 tablets.
10. However, to maintain effective protection against pregnancy, another method of contraception must be used as soon as menstruation resumes, the frequency or duration of breastfeeds is reduced, bottle feeds are introduced, or the baby reaches 6 months of age.
DOSAGE AND ADMINISTRATION
To achieve maximum contraceptive effectiveness, DOLISHALE (levonorgestrel and ethinyl estradiol tablets) must be taken exactly as directed and at intervals not exceeding 24 hours. The possibility of ovulation and conception prior to initiation of medication should be considered. Women who do not wish to become pregnant after discontinuation should be advised to immediately use another method of birth control. The dosage of DOLISHALE is one green tablet daily without any tablet-free interval.
It is recommended that DOLISHALE tablets be taken at the same time each day.
Initiation of Therapy
Instructions for beginning DOLISHALE are provided in Table 4 below.
Table 4
| Current contraceptive therapy | DOLISHALE start day | Nonhormonal back-up method of birth control needed when correctly starting DOLISHALE? |
| None | Day 1 of patient's menstrual cycle (during the first 24 hours of her period) | No |
| 21-day COC regimen OR 28-day COC regimen | Day 1 of patient's withdrawal bleed, at the latest 7 days after her last active tablet. | No |
| Progestin-only pill | Day after taking a progestin-only pill | Yes, for the first 7 days of DOLISHALE tablet taking |
| Implant | Day of implant removal | Yes, for the first 7 days of DOLISHALE tablet taking |
| Injection | Day the next injection is due | Yes, for the first 7 days of DOLISHALE tablet taking |
If spotting or unscheduled bleeding occurs, the patient is instructed to continue on the same regimen. This type of bleeding is usually transient and without significance; however, if the bleeding is persistent or prolonged, the patient is advised to consult her health care professional. The possibility of ovulation increases with each successive day that scheduled green tablets are missed. If the patient has not adhered to the prescribed schedule (missed one or more tablets or started taking them on a day later than she should have), the probability of pregnancy should be considered. Hormonal contraception must be discontinued if pregnancy is confirmed.
The risk of pregnancy increases with each tablet missed. For additional patient instructions regarding missed tablets, see the WHAT TO DO IF YOU MISS PILLS section in the DETAILED PATIENT LABELING below.
DOLISHALE may be initiated no earlier than day 28 postpartum in the nonlactating mother or after a second-trimester abortion due to the increased risk for thromboembolism (see CONTRAINDICATIONS , WARNINGS , and PRECAUTIONS concerning thromboembolic disease). The patient should be advised to use a nonhormonal back-up method for the first 7 days of tablet-taking. However, if intercourse has already occurred, pregnancy should be excluded before the start of combined oral contraceptive use or the patient must wait for her first menstrual period.
In the case of first-trimester abortion, if the patient starts DOLISHALE immediately, additional contraceptive measures are not needed.
CONTRAINDICATIONS
Combination oral contraceptives should not be used in women with any of the following conditions:
• Thrombophlebitis or thromboembolic disorders
• History of deep-vein thrombophlebitis or thromboembolic disorders
• Cerebrovascular or coronary artery disease (current or past history)
• Valvular heart disease with thrombogenic complications
• Thrombogenic rhythm disorders
• Hereditary or acquired thrombophilias
• Major surgery with prolonged immobilization
• Diabetes with vascular involvement
• Headaches with focal neurological symptoms such as aura
• Uncontrolled hypertension
• Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive
• Carcinoma of the endometrium or other known or suspected estrogen-dependent neoplasia Undiagnosed abnormal genital bleeding
• Cholestatic jaundice of pregnancy or jaundice with prior pill use
• Hepatic adenomas or carcinomas, or active liver disease
• Known or suspected pregnancy
• Hypersensitivity to any of the components of DOLISHALE
• Are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations (see Warnings, RISK OF LIVER ENZYME ELEVATIONS WITH CONCOMITANT HEPATITIS C TREATMENT ).
ADVERSE REACTIONS
Post Marketing Experience Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 6).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 6). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
Figure 6. Risk of Breast Cancer with Combined Oral Contraceptive Use
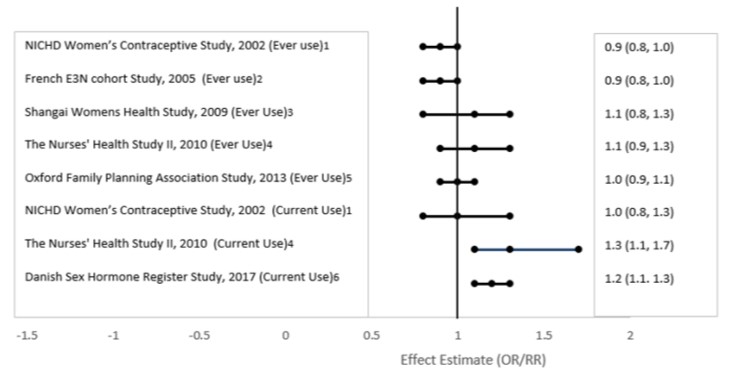 RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs.
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs.
An increased risk of the following serious adverse reactions (see WARNINGS section for additional information) has been associated with the use of oral contraceptives:
Thromboembolic and thrombotic disorders and other vascular problems (including thrombophlebitis and venous thrombosis with or without pulmonary embolism, mesenteric thrombosis, arterial thromboembolism, myocardial infarction, cerebral hemorrhage, cerebral thrombosis, transient ischemic attack), carcinoma of the reproductive organs and breasts, hepatic neoplasia/liver disease (including hepatic adenomas or benign liver tumors), ocular lesions (including retinal vascular thrombosis), gallbladder disease, carbohydrate and lipid effects, elevated blood pressure, and headache including migraine.
The following adverse reactions have been reported in patients receiving oral contraceptives and are believed to be drug related (alphabetically listed):
• Acne
• Amenorrhea
• Anaphylactic/anaphylactoid reactions, including urticaria, angioedema, and severe reactions with respiratory and circulatory symptoms
• Breast changes: tenderness, pain, enlargement, secretion
• Budd-Chiari syndrome
• Cervical erosion and secretion, change in
• Cholestatic jaundice
• Chorea, exacerbation of
• Colitis
• Contact lenses, intolerance to
• Corneal curvature (steepening), change in
• Dizziness
• Edema/fluid retention
• Erythema multiforme
• Erythema nodosum
• Focal nodular hyperplasia
• Gastrointestinal symptoms (such as abdominal pain, cramps, and bloating)
• Hirsutism
• Infertility after discontinuation of treatment, temporary
• Lactation, diminution in, when given immediately postpartum
• Libido, change in
• Melasma/chloasma which may persist
• Menstrual flow, change in
• Mood changes, including depression
• Nausea
• Nervousness
• Pancreatitis
• Porphyria, exacerbation of
• Rash (allergic)
• Scalp hair, loss of
• Serum folate levels, decrease in
• Spotting
• Systemic lupus erythematosus, exacerbation of
• Unscheduled bleeding
• Vaginitis, including candidiasis
• Varicose veins, aggravation of
• Vomiting
• Weight or appetite (increase or decrease), change in
The following adverse reactions have been reported in users of oral contraceptives:
• Cataracts
• Cystitis-like syndrome
• Dysmenorrhea
• Hemolytic uremic syndrome
• Hemorrhagic eruption
• Optic neuritis, which may lead to partial or complete loss of vision
• Premenstrual syndrome
• Renal function, impaired
To report SUSPECTED ADVERSE REACTIONS, contact Ingenus Pharmaceuticals, LLC Toll-Free at 1-877-748-1970 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Twenty-eight (28) green tablets each containing 90 mcg of levonorgestrel (17α)-(–)13-ethyl-17-hydroxy-18, 19-dinorpregn-4-en-20-yn-3-one, a totally synthetic progestogen, and 20 mcg of ethinyl estradiol, (17α)-19-norpregna-1,3,5(10)-trien-20-yne-3,17-diol. The inactive ingredients present are titanium dioxide, macrogol/PEG 3000 NF, talc, polyvinyl alcohol, lecithin (soya), FD&C Blue #2 Aluminum Lake, FD&C Yellow #5 Aluminum Lake, FD&C Red #40 Aluminum Lake, lactose monohydrate, magnesium stearate and pregelatinized starch.
CLINICAL PHARMACOLOGY
Mode of Action
Combination oral contraceptives act by suppression of gonadotropins. Although the primary mechanism of this action is inhibition of ovulation, other alterations include changes in the cervical mucus (which increase the difficulty of sperm entry into the uterus) and the endometrium (which reduce the likelihood of implantation).
Pharmacokinetics
Absorption
No specific investigation of the absolute bioavailability of levonorgestrel and ethinyl estradiol tablets in humans has been conducted. However, literature indicates that levonorgestrel is rapidly and completely absorbed after oral administration (bioavailability about 100%) and is not subject to first-pass metabolism. Ethinyl estradiol is rapidly and almost completely absorbed from the gastrointestinal tract but, due to first-pass metabolism in gut mucosa and liver, the bioavailability of ethinyl estradiol is between 38% and 48%.
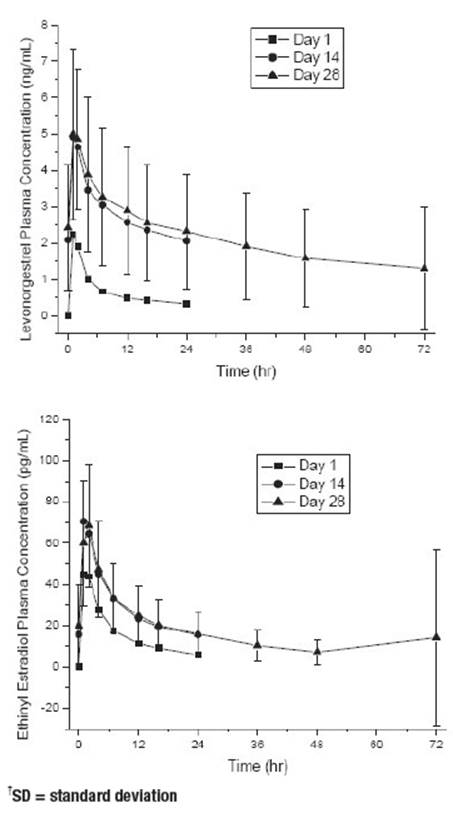
A summary of the single dose and multiple dose levonorgestrel and ethinyl estradiol pharmacokinetic parameters for 18 women under fasting conditions is provided in Table 1. The plasma concentrations of levonorgestrel and ethinyl estradiol reached steady-state by approximately day 14. Levonorgestrel and ethinyl estradiol concentrations did not increase from days 14 to 28, but did increase from days 1 to 28.
Table 1: Mean (SD) Pharmacokinetic Parameters of Levonorgestrel and Ethinyl Estradiol Tablets Over a 28-Day Dosing Period
| LNG | ||||
Day | C max (ng/mL) | T max (h) | t 1/2 (h) | AUC 0-24 (ng•h/mL) |
| 1 | 2.4 (0.9) | 1.2 (0.4) | - | 16 (8) |
| 14 | 5.4 (2.1) | 1.7 (1.4) | - | 68 (36) |
| 28 | 5.7 (2.1) | 1.3 (0.8) | 36 (19) | 74 (41) |
| EE | ||||
| Day | (pg/mL) | (h) | (h) | (pg h/mL) |
| 1 | 47.7 (20.1) | 1.3 (0.5) | - | 378 (140) |
| 14 | 72.7 (37.2) | 1.4 (0.5) | - | 695 (361) |
| 28 | 74.4 (29.7) | 1.4 (0.5) | 21 (7) | 717 (351) |
The mean plasma concentrations of levonorgestrel and ethinyl estradiol following single (day 1) and multiple (days 14 and 28) oral administrations of levonorgestrel 90 mcg in combination with ethinyl estradiol 20 mcg to 18 healthy women is provided in Figure 1.
Figure 1: Mean Plasma ± SD † Concentrations of Levonorgestrel and Ethinyl Estradiol Following Single (Day 1) and Multiple (Days 14 and 28) Oral Administrations of Levonorgestrel 90 mcg in Combination with Ethinyl Estradiol 20 mcg to Healthy Women
The effect of food on the rate and the extent of levonorgestrel and ethinyl estradiol absorption following oral administration of DOLISHALE has not been evaluated.
Distribution
Levonorgestrel in serum is primarily bound to sex hormone-binding globulin (SHBG). Ethinyl estradiol is about 97% bound to serum albumin. Ethinyl estradiol does not bind to SHBG, but induces SHBG synthesis.
Metabolism
Levonorgestrel: The most important metabolic pathways are reduction of the Δ4-3-oxo group and hydroxylation at positions 2α, 1β, and 16β, followed by conjugation. Most of the circulating metabolites are sulfates of 3α, 5β-tetrahydro-levonorgestrel, while excretion occurs predominantly in the form of glucuronides. Some of the parent levonorgestrel also circulates as 17β-sulfate. Metabolic clearance rates may differ among individuals by several-fold, and this may account in part for the wide variation observed in levonorgestrel concentrations among users.
Ethinyl estradiol: Cytochrome P450 enzymes (CYP3A4) in the liver are responsible for the 2-hydroxylation that is the major oxidative reaction. The 2-hydroxy metabolite is further transformed by methylation, sulfation, and glucuronidation prior to urinary and fecal excretion. Levels of CYP3A4 vary widely among individuals and can explain the variation in rates of ethinyl estradiol 2-hydroxylation.
Excretion
The terminal elimination half-life for levonorgestrel in levonorgestrel and ethinyl estradiol tablets is about 36 hours. Levonorgestrel and its metabolites are excreted in the urine (40% to 68%) and in feces (16% to 48%). The terminal elimination half-life of ethinyl estradiol in levonorgestrel and ethinyl estradiol tablets is about 21 hours.
Ethinyl estradiol is excreted in the urine and feces as glucuronide and sulfate conjugates and undergoes enterohepatic recirculation.
Special Populations
Race
No formal studies on the effect of race on the pharmacokinetic parameters of levonorgestrel and ethinyl estradiol tablets were conducted.
Hepatic Insufficiency
No formal studies have evaluated the effect of hepatic disease on the disposition of levonorgestrel and ethinyl estradiol tablets. However, steroid hormones may be poorly metabolized in patients with impaired liver function.
Renal Insufficiency
No formal studies have evaluated the effect of renal disease on the disposition of levonorgestrel and ethinyl estradiol tablets.
Drug-Drug Interactions
See PRECAUTIONS section - Drug Interactions .
Clinical Studies
The efficacy and safety of levonorgestrel and ethinyl estradiol tablets were studied in 2 one-year clinical trials of subjects age 18 to 49. There were no exclusions for body mass index (BMI), weight, or bleeding history.
The primary efficacy and safety study (313-NA) was a one-year open-label clinical trial that treated 2,134 subjects in North America. Of these subjects 1,213 (56.8%) discontinued prematurely, including 102 (4.8%) discontinued by the Sponsor for early study closure. The mean weight of subjects in this study was 70.38 kg. The efficacy of levonorgestrel and ethinyl estradiol tablets was assessed by the number of pregnancies that occurred after the onset of treatment and within 14 days of the last dose. Among subjects 35 years or less, there were 23 pregnancies (4 of these occurred during the interval 1 to 14 days after the last day of pill use) during 12,572 28-day pill packs of use. The resulting total Pearl Index was 2.38 (95% CI: 1.51, 3.57) and the one-year life table pregnancy rate was 2.39 (95% CI: 1.57, 3.62). Pill pack cycles during which subjects used back-up contraception or were not sexually active were not included in these calculations. Among women 35 years or less who took the pills completely as directed, there were 15 pregnancies (method failures) resulting in a Pearl Index of 1.55 (95% CI: 0.87, 2.56) and the one-year life table pregnancy rate was 1.59 (95% CI: 0.95 to 2.67).
In a second supportive study conducted in Europe (315-EU), 641 subjects were randomized to levonorgestrel and ethinyl estradiol tablets (n=323) or the cyclic comparator of 100 mcg levonorgestrel and 20 mcg ethinyl estradiol (n=318). The mean weight of subjects in this study was 63.86 kg. The efficacy analysis among women 35 years or less included 2,756 levonorgestrel and ethinyl estradiol tablet pill packs and 2,886 cyclic comparator pill packs. There was one pregnancy in the levonorgestrel and ethinyl estradiol group that occurred within 14 days following the last dose. There were three pregnancies in the cyclic comparator group.
HOW SUPPLIED
DOLISHALE(90 mcg levonorgestrel and 20 mcg ethinyl estradiol) tablets are available in a blister pack, arranged in 4 rows of 7 active tablets as follows:
28 round, green biconvex tablets debossed with "H1" on one side.
DOLISHALE Tablets are available in the following packaging configurations:
Carton of 1 Blister Card of 28 Tablets NDC 50742-659-28
Carton of 3 Blister Cards of 28 Tablets Each NDC 50742-659-84
Carton of 6 Blister Cards of 28 Tablets Each NDC 50742-659-68
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature].
References available upon request.
Rx Only
Iss: 12/2024
I0154
Rev B
Manufactured for:
Ingenus Pharmaceuticals, LLC
Orlando, FL 32839-6408
Product of China

Dolishale - Levonorgestrel And Ethinyl Estradiol tablet PubMed™ news
- Journal Article • 2026 MayCerebral venous sinus thrombosis risk across various oral contraceptives: a disproportionality analysis of the FDA adverse event reporting system.
- Journal Article • 2026 MayLevonorgestrel intrauterine system: do the contraceptive benefits outweigh the risks in light of new FSRH guidance?
- Journal Article • 2026 MayBarriers and reasons for initiating the use of long-acting reversible contraceptives (LARC) in a cohort of Brazilian women.
- Journal Article • 2026 MayManagement and outcomes of fertility preservation in rare Cowden syndrome-associated endometrial carcinoma: a case series.
- Journal Article • 2026 AprWho Is Still Paying Out-of-Pocket for Their IUDs Despite the ACA Contraceptive Coverage Requirement? Analysis of a Large National Cohort Using Claims Data.