Get your patient on Ensacove - Ensartinib capsule (Ensartinib)
Ensacove - Ensartinib capsule prescribing information
INDICATIONS AND USAGE
ENSACOVE is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive locally advanced or metastatic non-small cell lung cancer (NSCLC)as detected by an FDA-approved test [see Dosage and Administration (2.1 )] who have not previously received an ALK-inhibitor.
DOSAGE AND ADMINISTRATION
2.1 Patient Selection
Select patients for the treatment of locally advanced or metastatic NSCLC with ENSACOVE based on the presence of ALK rearrangement(s) in tumor specimens [see Clinical Studies (14.1 )] .
Information on FDA-approved tests for the detection of ALK rearrangements in NSCLC is available at http://www.fda.gov/CompanionDiagnostics .
2.2 Recommended Testing and Advice Prior to Initiating ENSACOVE
2.3 Recommended Dosage
The recommended dosage of ENSACOVE is 225 mg orally once daily, with or without food [see Clinical Pharmacology (12.3 )] , until disease progression or unacceptable toxicity.
Swallow capsules whole, do not crush or chew. Do not open or dissolve the contents of the capsule. Take ENSACOVE at the same time each day.
Missed dose
If a dose is missed, then take the missed dose as soon as possible unless the next dose is due within 12 hours. Do not take 2 doses on the same day.
Vomiting
If vomiting occurs after taking a dose, do not take an additional dose and take the next dose at its scheduled time.
2.4 Dosage Modification for Adverse Reactions
The recommended dose reductions for adverse reactions are provided in Table 1.
| Dose Reduction | Recommended Dose and Schedule |
| First | 200 mg orally once daily |
| Second | 150 mg orally once daily |
| Permanently discontinue ENSACOVE if patients are unable to tolerate 150 mg orally once daily. | |
Once the dose has been reduced for adverse reactions, do not subsequently increase the dose of ENSACOVE.
The recommended dosage modifications for the management of adverse reactions are provided in Table 2.
| Adverse Reaction | Severity• | ENSACOVE Dose Modification and Management for Adverse Reactions |
| Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.1 )] | Any Grade | Permanently discontinue ENSACOVE. |
| Hepatotoxicity [see Warnings and Precautions (5.2 )] | Grade 3 or 4 elevation (greater than 5 times ULN) of either ALT or AST with concurrent total bilirubin less than or equal to 2 times ULN |
|
| Grade 2 to 4 elevation (greater than 3 times ULN) of either ALT or AST with concurrent total bilirubin elevation greater than 2 times ULN in the absence of cholestasis or hemolysis | Permanently discontinue ENSACOVE. | |
| Dermatologic Adverse Reactions [see Warnings and Precautions (5.3 )] | Grade 1 | Consider topical corticosteroids. |
| Grade 2 |
| |
| Grade 3 |
| |
| Grade 4 |
| |
| Bradycardia (HR less than 60 bpm) [see Warnings and Precautions (5.4 )] | Symptomatic bradycardia |
|
| Bradycardia with life-threatening consequences, urgent intervention indicated |
| |
| Hyperglycemia [see Warnings and Precautions (5.5 )] | Grade 3 (greater than 250 mg/dL) despite optimal anti- hyperglycemic therapy OR Grade 4 |
|
| Visual Disturbance [see Warnings and Precautions (5.6 )] | Grade 2 or 3 | Withhold ENSACOVE until recovery to Grade 1 or baseline, then consider resuming at reduced dose as per Table 1. |
| Grade 4 | Permanently discontinue ENSACOVE. | |
| Increased Creatine Phosphokinase [see Warnings and Precautions (5.7 )] | CPK elevation greater than 5 times ULN |
|
| CPK elevation greater than 10 times ULN or second occurrence of CPK elevation of greater than 5 times ULN |
| |
| Hyperuricemia [see Warnings and Precautions (5.8 )] | Symptomatic or Grade 4 |
|
| Other Adverse Reactions [see Adverse Reactions (6.1 )] | Grade 3 or 4 |
|
| Recurrent Grade 4 | Permanently discontinue ENSACOVE. | |
| ALT = alanine aminotransferase; AST = aspartate aminotransferase; bpm = beats per minute; HR = heart rate; ULN = upper limit of normal •Graded per National Cancer Institute Common Terminology Criteria for Adverse Events. Version 4.03. | ||
DOSAGE FORMS AND STRENGTHS
ENSACOVE capsules are available as:
- 25 mg: size 2 capsule, white opaque cap and body, with “X-396” on the cap and “25 mg” on the body printed in blue ink. Each capsule contains the equivalent of 25 mg of ensartinib.
- 100 mg: size 0 capsule, blue opaque cap and yellow opaque body, with “X-396” on the cap and “100 mg” on the body printed in white ink. Each capsule contains the equivalent of 100 mg of ensartinib.
USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action [see Clinical Pharmacology (12.1 )] , ENSACOVE can cause fetal harm when administered to a pregnant woman. There are no available data on the use of ENSACOVE in pregnant women to inform a drug-associated risk. Oral administration of ensartinib to pregnant rats during the period of organogenesis caused adverse developmental outcomes, including embryo-fetal mortality, alterations to growth, and structural abnormalities. Adverse embryo-fetal findings were seen at maternal exposures approximately equivalent to the human exposure at the recommended dose of 225 mg/day based on AUC ( see Data ). Advise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In an embryo-fetal development study, pregnant rats received 20, 40, or 80 mg/kg/day of ensartinib during the period of organogenesis (gestation day 6 to 17). Ensartinib doses ≥40 mg/kg/day (approximately equivalent to the human exposure at the recommended dose of 225 mg/day based on AUC) resulted in an increase in the incidence of fetal malformations (aortic dislocation, ventricular septal defect, separated vertebrae) and dose-dependent delayed skeletal development, including decreased or delayed ossification of the vertebrae. Ensartinib at 80 mg/kg (approximately 6.4 times the human exposure at the recommended dose of 225 mg/day based on AUC) resulted in maternal toxicity (reduced body weight and food consumption), decreased fetal body weight, increased pre- and post-implantation loss, decreases in the number of live fetuses, and visceral variations (missing renal papillae, pyelectasis).
8.2 Lactation
Risk Summary
There are no data on the presence of ensartinib or its metabolites in human milk, the effects on the breastfed child, or the effects on milk production. Because of the potential for serious adverse reactions in breastfed children, advise women not to breastfeed during treatment with ENSACOVE and for 1 week after the last dose.
8.3 Females and Males of Reproductive Potential
ENSACOVE can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1 ) ].
Pregnancy Testing
Verify the pregnancy status of females of reproductive potential prior to initiating ENSACOVE.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with ENSACOVE and for at 1 week after the last dose.
Males
Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENSACOVE and for 1 week after the last dose.
8.4 Pediatric Use
The safety and effectiveness of ENSACOVE in pediatric patients have not been established.
8.5 Geriatric Use
Of the 458 patients enrolled in clinical studies and received ENSACOVE 225 mg once daily, 16% of the participants were aged 65 years or older. Clinical studies of ENSACOVE did not include sufficient numbers of patients ages 65 and over to determine whether they respond differently from younger patients. Exploratory analysis suggests a higher incidence of serious adverse events (43% vs 27%), more frequent adverse events leading to treatment discontinuations (18% vs 10%) and dose modifications (34% vs 16%) in patients 65 years or older as compared to those younger than 65 years.
8.6 Hepatic Impairment
Ensartinib is primarily metabolized by the liver and patients with hepatic impairment may have increased exposures [see Clinical Pharmacology (12.3 )]. Avoid use of ENSACOVE for patients with severe hepatic impairment (total bilirubin >3 times ULN and any AST) since it has not been studied in this population. Monitor patients with moderate hepatic impairment (total bilirubin >1.5 to ≤ 3 ULN and any AST) for increased adverse reactions and adjust ENSACOVE dosage as clinically indicated [see Dosage and Administration (2.4 ), Warnings and Precautions (5.2 )]. No dosage modification is recommended for patients with mild hepatic impairment (total bilirubin ≤ upper limit of normal (ULN) and AST > ULN or total bilirubin 1 to 1.5 x ULN and any AST).
CONTRAINDICATIONS
ENSACOVE is contraindicated in patients who have experienced a severe hypersensitivity reaction to ENSACOVE, FD&C Yellow No. 5 (tartrazine), or to any of its components [see Warnings and Precautions (5.10 )] .
WARNINGS AND PRECAUTIONS
5.1 Interstitial Lung Disease/Pneumonitis
ENSACOVE can cause severe interstitial lung disease (ILD)/pneumonitis.
In the pooled safety population [see Adverse Reactions (6.1 )], ILD/pneumonitis occurred in 5% of patients treated with ENSACOVE, including Grade 3 in 1.3% and Grade 4 in 0.4%. ILD/pneumonitis leading to dose interruption occurred in 0.4% and permanent discontinuation of ENSACOVE in 1.5% of patients.
Monitor patients for new or worsening symptoms indicative of ILD/pneumonitis (e.g., dyspnea, cough, and fever) during treatment with ENSACOVE. Immediately withhold ENSACOVE in patients with suspected ILD/pneumonitis. Permanently discontinue ENSACOVE if ILD/pneumonitis is confirmed [see Dosage and Administration (2.4 )].
5.2 Hepatotoxicity
ENSACOVE can cause hepatotoxicity including drug-induced liver injury.
In the pooled safety population [see Adverse Reactions (6.1 )], 59% of patients treated with ENSACOVE had increased alanine aminotransferase (ALT), including 5% Grade 3. Increased aspartate aminotransferase (AST) occurred in 58% of patients treated with ENSACOVE, including 1.8% Grade 3. Increased bilirubin occurred in 12% of patients treated with ENSACOVE, including 2.3% Grade 3 and 0.2% Grade 4. There was one case of drug-induced liver injury in ENSACOVE-treated patients.
The median time to first onset of increased ALT or AST was 5.3 weeks (range: 0.4 to 152 weeks). The dose of ENSACOVE was interrupted in 4.6% of patients for increased ALT or AST. Increased ALT or AST leading to dose reduction occurred in 2.6% and permanent discontinuation of ENSACOVE in 1.1% of patients. The dose of ENSACOVE was interrupted in 1.3% of patients for increased bilirubin. Increased bilirubin leading to dose reduction occurred in 0.7% and permanent discontinuation of ENSACOVE in 1.3% of patients.
Monitor liver function tests including ALT, AST, and total bilirubin at baseline and every 2 weeks during the first cycle of treatment with ENSACOVE, and then monthly and as clinically indicated. Withhold, reduce the dose, or permanently discontinue ENSACOVE based on the severity of the adverse reaction [see Dosage and Administration (2.4 )] .
5.3 Dermatologic Adverse Reactions
ENSACOVE can cause dermatologic adverse reactions, including drug reaction with eosinophilia and systemic symptoms (DRESS), rash, pruritus, and photosensitivity.
In the pooled safety population [see Adverse Reactions (6.1 )], dermatologic adverse reactions occurred in 80% of patients receiving ENSACOVE, including Grade 3 in 14% of patients. Rash occurred in 72% of patients receiving ENSACOVE, including Grade 3 in 12% of patients. The median time to onset of rash was 9 days (range: 1 day to 17.3 months). Pruritus occurred in 32% of patients receiving ENSACOVE, with Grade 3 in 2.4%. There was one Grade 3 case (0.2%) of drug reaction with eosinophilia and systemic symptoms (DRESS).
The dose of ENSACOVE was interrupted in 12% of patients for dermatologic adverse reactions. Dermatologic adverse reactions leading to dose reduction occurred in 11% and permanent discontinuation of ENSACOVE in 1.5% of patients.
In the pooled safety population [see Adverse Reactions (6.1 ) ], photosensitivity occurred in 0.9% of patients receiving ENSACOVE; all were Grade 1.
Monitor patients for dermatologic adverse reactions during treatment with ENSACOVE. If dermatologic adverse reactions occur, treat with antihistamine, topical or systemic steroids based on the severity. Advise patients to limit direct sun exposure while taking ENSACOVE and for at least 1 week after discontinuation. Withhold, reduce the dose, or permanently discontinue ENSACOVE based on the severity of the adverse reaction [see Dosage and Administration (2.4 )].
5.4 Bradycardia
ENSACOVE can cause symptomatic bradycardia.
In the pooled safety population [see Adverse Reactions (6.1 )], bradycardia (heart rate less than 60 beats per minute) occurred in 6% of patients treated with ENSACOVE. All bradycardia events were Grade 1 or 2. Bradycardia requiring dose reduction occurred in 0.2% and led to dose interruption in 0.4% of ENSACOVE-treated patients.
Monitor heart rate regularly during treatment with ENSACOVE. Withhold, reduce the dose, or permanently discontinue ENSACOVE based on severity [see Dosage and Administration (2.4 )] .
5.5 Hyperglycemia
ENSACOVE can cause hyperglycemia.
In the pooled safety population [see Adverse Reactions (6.1 )], based on laboratory data, 44% of patients receiving ENSACOVE experienced increased blood glucose, including Grade 3 in 2.5%. The median time to onset of increased blood glucose was 5.9 weeks (0.4 weeks to 3.4 years).
Assess fasting serum glucose at baseline and monitor serum glucose periodically during treatment with ENSACOVE. Withhold, reduce the dose, or permanently discontinue ENSACOVE based on severity [see Dosage and Administration (2.4 )].
5.6 Visual Disturbances
ENSACOVE can cause visual disturbances including blurred vision, diplopia, photopsia, vitreous floaters, visual impairment, visual field defect, and reduced visual acuity.
In the pooled safety population [see Adverse Reactions (6.1 )], 8% of patients receiving ENSACOVE experienced visual disturbance, including 0.2% Grade 3. Visual disturbances led to dose interruption in 0.4% of patients.
Obtain an ophthalmologic evaluation in patients with new or worsening visual symptoms during treatment with ENSACOVE. Withhold, reduce the dose, or permanently discontinue ENSACOVE based on severity [see Dosage and Administration (2.4 )].
5.7 Increased Creatine Phosphokinase
In the pooled safety population [see Adverse Reactions (6.1 )], of the 203 patients with creatine phosphokinase (CPK) laboratory data available, increased CPK occurred in 43% of patients who received ENSACOVE. The incidence of Grade 3 increased CPK was 1.5% and 0.5% were Grade 4. The median time to onset of increased CPK was 123 days (range: 13 days to 22 months). Increased CPK leading to dose interruption occurred in 0.2% and dose reduction in 0.4%.
Advise patients to report any unexplained muscle pain, tenderness, or weakness. Monitor CPK levels during treatment with ENSACOVE. Withhold, reduce the dose, or permanently discontinue ENSACOVE based on severity [see Dosage and Administration (2.4 ), Adverse Reactions (6.1 )].
5.8 Hyperuricemia
ENSACOVE can cause hyperuricemia.
In the pooled safety population [see Adverse Reactions (6.1 )], based on adverse reactions, 6% of patients experienced hyperuricemia, with 0.4% Grade 3 and 0.7% Grade 4. Nine patients (1.9%) required hydration and two patients (0.4%) required urate-lowering medication.
Monitor serum uric acid levels prior to initiating ENSACOVE and periodically during treatment. Initiate treatment with urate-lowering medications as clinically indicated. Withhold, reduce the dose, or permanently discontinue ENSACOVE based on severity [see Dosage and Administration (2.4 )].
5.9 Embryo-Fetal Toxicity
Based on findings from animal studies and its mechanism of action, ENSACOVE can cause fetal harm when administered to a pregnant woman. In embryo-fetal developmental studies, oral administration of ensartinib to pregnant rats during the period of organogenesis caused adverse developmental outcomes, including embryo-fetal mortality, alterations to growth, and structural abnormalities. Adverse embryo-fetal findings were seen at maternal exposures approximately equivalent to the human exposure at the recommended dose of 225 mg/day based on area under the curve (AUC). Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ENSACOVE and for 1 week after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with ENSACOVE and for 1 week after the last dose [see Use in Specific Populations (8.1 , 8.3 ) and Clinical Pharmacology (12.1 )].
5.10 FD&C Yellow No. 5 (Tartrazine)
This product contains FD&C Yellow No. 5 (tartrazine) which may cause allergic-type reactions (including bronchial asthma) in certain susceptible persons. Although the overall incidence of FD&C Yellow No. 5 (tartrazine) sensitivity in the general population is low, it is frequently seen in patients who also have aspirin hypersensitivity.
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1 )]
- Hepatoxicity [see Warnings and Precautions (5.2 )]
- Dermatologic Adverse Reactions [see Warnings and Precautions (5.3 )]
- Bradycardia [see Warnings and Precautions (5.4 )]
- Hyperglycemia [see Warnings and Precautions (5.5 )]
- Visual Disturbances [see Warnings and Precautions (5.6 )]
- Increased Creatine Phosphokinase [see Warnings and Precautions (5.7 )]
- Hyperuricemia [see Warnings and Precautions (5.8 )]
- FD&C Yellow No. 5 (Tartrazine) [see Warnings and Precautions (5.10 ) ]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflects exposure to ENSACOVE as a single agent in 458 patients with locally advanced or metastatic ALK-positive NSCLC in the following trials: eXALT3 Study (N=143) [see Clinical Studies (14.1 ) ], Study 101 (NCT01625234, N=98), Study BTP-28311 (NCT02959619, N=35), and Study BTP-42322 (NCT03215693, N=182). Patients received ENSACOVE 225 mg orally once daily, with or without food, until disease progression or unacceptable toxicity. Among 458 patients who received ENSACOVE, 63% were exposed for 6 months or longer and 47% were exposed for greater than one year. In this pooled safety population, the most common adverse reactions (≥20%) were rash, musculoskeletal pain, constipation, pruritus, cough, nausea, edema, vomiting, fatigue, and pyrexia. The most frequent Grade 3 or 4 laboratory abnormalities (≥2%) were increased uric acid, decreased lymphocytes, increased alanine aminotransferase, decreased phosphate, increased gamma glutamyl transferase, increased magnesium, increased amylase, decreased sodium, increased glucose, decreased hemoglobin, increased bilirubin, decreased potassium, and increased creatine phosphokinase.
TKI-naive ALK-Positive Locally Advanced or Metastatic NSCLC
The safety of ENSACOVE was evaluated in the eXALT3 study [see Clinical Studies (14.1 ) ]. Patients received ENSACOVE 225 mg orally once daily, with or without food, until disease progression or unacceptable toxicity . Among patients who received ENSACOVE, 78% were exposed for 6 months or longer and 66% were exposed for greater than one year.
The median age of patients who received ENSACOVE was 54 years (range: 25-86); 50% male; 54% Asian, 43% White; 0.7% Black or African American; and 11% Hispanic or Latino.
Serious adverse reactions occurred in 23% of patients treated with ENSACOVE. Serious adverse reactions that occurred in ≥1% were pneumonia (4.9%), hemorrhage (2.1%), rash (2.1%) and cellulitis (1.4%). One fatal adverse reaction (0.7%) occurred due to bronchopneumonia.
Permanent discontinuation of ENSACOVE due to an adverse reaction occurred in 12% of patients. Adverse reactions which resulted in permanent discontinuation of ENSACOVE (≥1%) included increased blood bilirubin (1.4%), increased conjugated bilirubin (1.4%), increased ALT (2.1%), increased AST (2.1%), and pneumonitis/ILD (2.1%).
Dose interruptions of ENSACOVE due to an adverse reaction occurred in 41% of patients. Adverse reactions which required dose interruptions (≥2%) included rash (13%), increased ALT (6%), edema (2.8%), pruritus (2.8%), pyrexia (2.8%), pneumonia (3.5%), increased AST (2.1%), hemorrhage (2.1%), and decreased appetite (2.1%).
Dose reductions of ENSACOVE due to an adverse reaction occurred in 24% of patients. Adverse reactions which required dose reductions (≥2%) included rash (11%), increased ALT (4.2%), pruritus (2.8%), and edema (2.1%).
Tables 3 and 4 summarize the most frequent adverse reactions and laboratory abnormalities, respectively.
| ENSACOVE N = 143 | Crizotinib N = 146 | ||||
| Adverse Reaction | All Grades % | Grade 3 or 4 % | All Grades % | Grade 3 or 4 % | |
| Skin and Subcutaneous Tissue Disorders | |||||
| Rash a | 66 | 12 | 10 | 0 | |
| Pruritus b | 30 | 2.1 | 4.1 | 0 | |
| Alopecia | 11 | 0 | 4.8 | 0 | |
| Dry Skin | 10 | 0.7 | 0.7 | 0 | |
| Musculoskeletal and Connective Tissue Disorders | |||||
| Musculoskeletal Pain c | 36 | 1.4 | 20 | 0 | |
| Respiratory, Thoracic and Mediastinal Disorders | |||||
| Cough d | 31 | 0.7 | 16 | 0 | |
| Gastrointestinal Disorders | |||||
| Constipation | 31 | 0 | 26 | 0 | |
| Nausea | 28 | 1.4 | 30 | 2.1 | |
| Vomiting e | 16 | 0.7 | 32 | 0 | |
| General Disorders and Administration Site Conditions | |||||
| Edema f | 27 | 2.1 | 28 | 2.1 | |
| Pyrexia g | 22 | 0.7 | 10 | 0.7 | |
| Fatigue h | 21 | 0.7 | 14 | 1.4 | |
| Metabolism and Nutrition Disorders | |||||
| Decreased appetite | 15 | 0 | 12 | 1.4 | |
| Infection and Infestation | |||||
| Respiratory Tract Infection | 13 | 0.7 | 10 | 0 | |
| Nervous System Disorders | |||||
| Dizziness i | 12 | 0.7 | 14 | 0.7 | |
| Dysgeusia | 10 | 0 | 11 | 0 | |
| Vascular Disorders | |||||
| Hemorrhage j | 10 | 1.4 | 4.8 | 0 | |
| Adverse reactions were graded using NCI CTCAE version 4.03. a Includes dermatitis, dermatitis acneiform, dermatitis bullous, drug eruption, eczema, exfoliative rash, palmar-plantar erythrodysaesthesia, rash, rash erythematous, rash generalized, rash macular, rash maculo-papular, rash morbilliform, rash papular, rash pruritic, rash pustular, skin exfoliation, and vulvovaginal rash b Includes ear pruritus, eye pruritus, eyelids pruritus, lip pruritus, pruritus, and pruritus generalized c Includes arthritis, spinal pain, myalgia, musculoskeletal pain, back pain, pain in extremity, neck pain, arthralgia, non-cardiac chest pain, bone pain, musculoskeletal chest pain, musculoskeletal discomfort d Includes cough, productive cough, upper-airway cough syndrome e Includes vomiting and retching f Includes eyelid edema, face edema, generalized edema, localized edema, edema, edema peripheral, gravitational edema, skin edema, eye edema, and periorbital edema g Includes pyrexia and hyperthermia h Includes fatigue and asthenia i Includes dizziness, vertigo, postural dizziness j Includes hemoptysis, intracranial hemorrhage, gastrointestinal hemorrhage, hematuria, upper gastrointestinal hemorrhage, vaginal hemorrhage, gingival bleeding, vitreous hemorrhage, epistaxis, rectal hemorrhage, anal hemorrhage | |||||
| ENSACOVE N = 143 | Crizotinib N = 146 | |||
| Lab Abnormality | All Grades % | Grade 3 or 4 % | All Grades % | Grade 3 or 4 % |
| Chemistry | ||||
| Alanine aminotransferase increased | 73 | 5 | 74 | 8 |
| Alkaline phosphatase increased | 64 | 2.2 | 50 | 0.7 |
| Aspartate aminotransferase increased | 64 | 1.4 | 62 | 3.5 |
| Glucose increased | 49 | 5 | 35 | 0.7 |
| Albumin decreased | 46 | 0.7 | 56 | 1.4 |
| Phosphate decreased | 39 | 7 | 42 | 4.9 |
| Urate increased | 39 | 39 | 27 | 27 |
| Creatinine increased | 37 | 0 | 27 | 0 |
| Calcium decreased | 36 | 1.4 | 64 | 4.9 |
| Sodium decreased | 27 | 4.3 | 27 | 4.2 |
| Hematology | ||||
| Lymphocytes decreased | 57 | 7 | 47 | 5 |
| Hemoglobin decreased | 43 | 0.7 | 31 | 1.4 |
Adverse reactions were graded using NCI CTCAE version 4.03. ALT = Alanine aminotransferase; AST = Aspartate aminotransferase | ||||
Clinically relevant adverse reactions in <10% of patients who received ENSACOVE included interstitial lung disease, photosensitivity, increased creatinine phosphokinase, bradycardia, and visual disturbances.
DRUG INTERACTIONS
7.1 Effect of Other Drugs on ENSACOVE
Table 5 describes drug interactions where concomitant use of another drug affects ENSACOVE.
| Strong or Moderate CYP3A Inhibitors | |
| Prevention or Management | Avoid concomitant use of strong or moderate CYP3A inhibitors with ENSACOVE. |
| Mechanism and Clinical Effect(s) | This recommendation is based upon a mechanistic understanding of ensartinib pharmacokinetics and it being a CYP3A4 substrate in vitro [see Clinical Pharmacology (12.3 )] . Concomitant use with strong or moderate CYP3A inhibitors may increase ensartinib exposure; however, this has not been studied clinically. |
| Strong or Moderate CYP3A Inducers | |
| Prevention or Management | Avoid concomitant use of strong or moderate CYP3A inducers with ENSACOVE. |
| Mechanism and Clinical Effect(s) | This recommendation is based upon a mechanistic understanding of ensartinib pharmacokinetics and it being a CYP3A4 substrate in vitro [see Clinical Pharmacology (12.3 )] . Concomitant use with strong or moderate CYP3A inducers may decrease ensartinib exposure; however, this has not been studied clinically. |
| P-gp Inhibitors | |
| Prevention or Management | Avoid concomitant use of P-gp inhibitors with ENSACOVE. |
| Mechanism and Clinical Effect(s) | This recommendation is based upon a mechanistic understanding of ensartinib pharmacokinetics and it being a P-gp substrate in vitro [see Clinical Pharmacology (12.3 )] . Concomitant use with P-gp inhibitors may increase ensartinib exposure; however, this has not been studied clinically. |
DESCRIPTION
ENSACOVE capsules contain ensartinib, a kinase inhibitor, present as ensartinib hydrochloride with the chemical name 6-amino-5-[(1 R )-1-(2,6-dichloro-3-fluorophenyl)ethoxy]-N-{4-[(3 R ,5 S )- 3,5-dimethylpiperazine-1-carbonyl]phenyl}pyridazine-3-carboxamide, dihydrochloride. The molecular formula is C26H27Cl2FN6O3·2HCl and its molecular weight is 634.4 g/mol with the following structure:
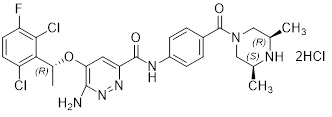
ENSACOVE capsules are intended for oral administration and are available in two dosage strengths: 25 mg ensartinib (equivalent to 28.25 mg ensartinib hydrochloride) and 100 mg ensartinib (equivalent to 113.02 mg ensartinib hydrochloride).
The inactive ingredients of ENSACOVE capsules are microcrystalline cellulose and stearic acid. The inactive ingredients of the 25 mg empty capsule shells are hypromellose and titanium dioxide. The inactive ingredients of the 100 mg empty capsules shells are black iron oxide, FD&C Blue No. 1, FD&C Yellow No. 5, hypromellose, red iron oxide, and titanium dioxide.
The imprinting ink for the 25 mg capsules contains butyl alcohol, dehydrated alcohol, FD&C Blue No. 2, isopropyl alcohol, propylene glycol, shellac, and strong ammonia solution. The imprinting ink for the 100 mg capsules contains butyl alcohol, dehydrated alcohol, isopropyl alcohol, povidone, propylene glycol, shellac, sodium hydroxide, and titanium dioxide.
CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ensartinib is a kinase inhibitor of anaplastic lymphoma kinase (ALK) and inhibits other kinases including MET and ROS1. In vitro, ensartinib inhibited phosphorylation of ALK and its downstream signaling proteins AKT, ERK, and S6, thereby blocking ALK-mediated signaling pathways and inhibiting proliferation in cell lines harboring ALK fusions and mutations. In vivo, ensartinib showed anti-tumor activity in a mouse xenograft model of human NSCLC harboring an ALK fusion.
12.2 Pharmacodynamics
Exposure-response relationship
Ensartinib exposure-response relationships and the time course of the pharmacodynamic response have not been fully characterized.
Cardiac Electrophysiology
At the approved recommended dosage, a mean increase in the QTc interval > 20 ms was not observed.
12.3 Pharmacokinetics
Ensartinib mean (coefficient of variation [CV%]) maximum concentration (Cmax) is 292 ng/mL (60%), and the area under the concentration-time curve (AUC0–24h) is 4,920 ng·h /ml (62%) at the approved recommended dosage. Ensartinib steady state is reached within 15 days with a mean accumulation ratio of 2.7.
Absorption
Ensartinib median (minimum, maximum) time to reach Cmax (Tmax) at steady state is 3 hours (2, 8 hours).
Effect of Food
No clinically significant differences in ensartinib pharmacokinetics were observed following administration of ENSACOVE with a high-fat meal (total 800-1000 calories, > 50% fat) compared to fasted conditions.
Distribution
Ensartinib mean (CV%) apparent volume of distribution is 1,720 L (42%). Ensartinib is 91.6% bound to human plasma protein.
Elimination
Ensartinib mean (standard deviation [SD]) steady-state half-life (t1/2) is 30 (20) hours.
Metabolism
Ensartinib is predominantly metabolized by CYP3A.
Excretion
Following a single oral 200 mg dose of radiolabeled ensartinib, 91% of the radioactivity was recovered in feces (38% as unchanged) and 10% in urine (4.4% as unchanged).
Specific Populations
No clinically significant differences in the pharmacokinetics of ensartinib were observed based on age (20 to 86 years), sex, race (Asian vs White), body weight (38 to 148 kg), mild to moderate renal impairment (eGFR 30 to 89 mL/min) and mild hepatic impairment (total bilirubin ≤ upper limit of normal (ULN) and AST > ULN or total bilirubin 1 to 1.5 x ULN and any AST).
The effect of severe renal impairment (eGFR 15 to 29 mL/min), end-stage renal disease (eGFR <15 mL/min) with or without hemodialysis, and moderate (total bilirubin >1.5 to ≤ 3 ULN and any AST) or severe (total bilirubin >3 times ULN and any AST) hepatic impairment on ensartinib pharmacokinetics is unknown.
Drug Interaction Studies
In Vitro Studies
Cytochrome P450 (CYP) Enzymes: Ensartinib does not inhibit CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6 and does not induce CYP1A2, CYP2B6, or CYP3A.
Transporter Systems: Ensartinib is a P-gp substrate but is not a substrate of BCRP, OATP1B1, OATP1B3, OAT1, OAT3, OCT1 or OCT2.
Ensartinib does not inhibit BCRP, P-gp, OATP1B1, OATP1B3, OAT1, OAT3, OCT1, OCT2 or OCT3.
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with ensartinib.
Ensartinib was not mutagenic in a bacterial reverse mutation (Ames) assay and was not clastogenic in an in vitro human lymphocyte chromosome aberration assay or an in vivo rat bone marrow micronucleus assay.
Dedicated fertility studies were not conducted with ensartinib. No adverse effects on male or female reproductive organs were observed in up to 3-month repeat-dose toxicology studies conducted in rats and dogs.
CLINICAL STUDIES
14.1 TKI-naive ALK-Positive Locally Advanced or Metastatic NSCLC (eXALT3 Study)
The efficacy of ENSACOVE was evaluated in eXALT3 (NCT02767804), an open-label, randomized, active-controlled, multicenter study in adult patients with locally advanced (stage IIIB following prior chemotherapy or chemoradiation or not amenable to curative intent therapy) or metastatic ALK-positive NSCLC. Patients were required to have ALK-positive NSCLC in tumor specimens as determined in local laboratories using immunohistochemistry (IHC), next-generation sequencing (NGS), polymerase chain reaction (PCR) or fluorescence in situ hybridization (FISH) tests. Of these patients, 85% (247/290) had centrally confirmed ALK rearrangements with the Vysis Break Apart FISH Probe Kit. Patients could have received one prior regimen of chemotherapy but could not have previously received an ALK-targeted therapy; patients with an ECOG performance status of 0, 1, or 2 were eligible. Patients with asymptomatic, untreated brain metastases who were not on corticosteroids and patients with asymptomatic, treated brain metastases who were on stable or decreasing dose of corticosteroids were eligible. Patients were required to have completed radiation therapy at least 2 weeks, or chemotherapy at least 4 weeks, prior to enrollment. Patients with leptomeningeal disease were ineligible.
Patients were randomized 1:1 to receive ENSACOVE 225 mg orally once daily or crizotinib 250 mg orally twice daily in 28-day cycles until disease progression or unacceptable toxicity. Randomization was stratified by prior chemotherapy (0 vs. 1), ECOG performance status (0 or 1 vs. 2), presence of central nervous system (CNS) metastases (yes or no), and geographic region (Asia vs. the rest of the world). Tumor assessments were performed every 8 weeks.
The main efficacy outcome measure was progression-free survival (PFS) as evaluated by Blinded Independent Central Review (BICR) according to RECIST version 1.1. The key secondary efficacy outcome measure was overall survival (OS); other secondary outcome measures included CNS response rate, time to CNS progression, and overall response rate (ORR).
A total of 290 patients were randomized to ENSACOVE (n=143) or crizotinib (n=147). The baseline demographic characteristics of the overall study population were median age 54 years (range: 25-90); 16% age > 65 years; 51% male; 56% Asian; 41% White and 1% Black; 7% Hispanic or Latino; ECOG PS 0 or 1 (95%); and 62% never smokers. Patients had Stage IIIB (8%) or Stage IV NSCLC (92%); 32% had received prior chemotherapy and 17% had received prior radiation. Baseline CNS metastases were present in 36% of the patients.
The eXALT3 study demonstrated a statistically significant improvement in PFS for patients randomized to ENSACOVE compared to patients randomized to crizotinib. The efficacy results as assessed by BICR are summarized in Table 6 and Figure 1.
| Efficacy Parameter | ENSACOVE N=143 | Crizotinib N=147 |
| Progression-free survival | ||
| Number of events, n (%) | 59 (41%) | 80 (54%) |
| Progressive disease, n (%) | 51 (36%) | 77 (52%) |
| Death, n (%) | 8 (6%) | 3 (2%) |
| Median, months (95% CI) | 25.8 (21.8, NE) | 12.7 (9.2, 16.6) |
| Hazard ratio (95% CI) | 0.56 (0.40, 0.79) | |
| p-value a | 0.0008 | |
| Overall response rate | ||
| Overall response rate % (95% CI) | 74% (66, 81) | 67% (58, 74) |
| Complete response % | 12% | 5% |
| Partial response % | 62% | 61% |
| Duration of response | ||
| Number of responders, n | 106 | 98 |
| Median, months (95% CI) | NE (22.0, NE) | 27.3 (12.9, NE) |
| CI = Confidence Interval, NE=not estimable, BICR = Blinded Independent Central Review a p-value based on unstratified log-rank test | ||
Figure 1 Kaplan-Meier Plot of Progression-Free Survival by IRR from Study 301 (eXALT3)
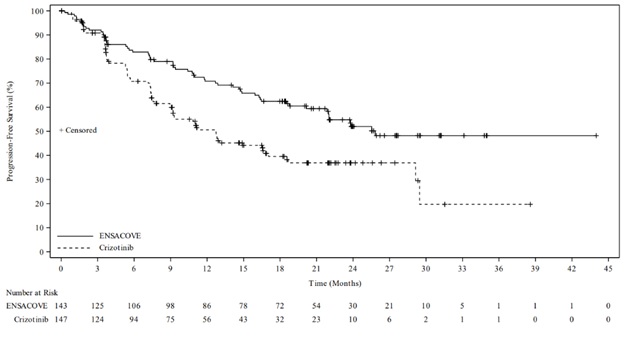
At the time of the primary PFS analysis, OS results were immature. At the time of final analysis of OS, there was no statistically significant difference (p-value = 0.4570) between ENSACOVE and crizotinib. Median OS was 63.2 months in the ENSACOVE arm and 55.7 months in the crizotinib arm, with the hazard ratio of 0.88 (95% CI: 0.63, 1.23).
The results of the pre-specified analyses of CNS response rate by BICR in patients with baseline measurable CNS disease are summarized in Table 7.
| Efficacy Parameter | ENSACOVE N=17 | crizotinib N=24 |
| CNS overall response rate % (95% CI) | 59% (33, 82) | 21% (7, 42) |
| Complete response % | 24% | 8% |
| Partial response % | 35% | 13% |
| Duration of Response | ||
| Number of responders, n | 10 | 5 |
| Patients with DOR ≥ 12 months | 30% | 40% |
| BICR = Blinded Independent Central Review; CI = Confidence Interval | ||
HOW SUPPLIED
ENSACOVE (ensartinib) capsules are supplied as follows:
| Capsule Strength | Description | Package Configuration | NDC Code |
| 25 mg | Size 2 capsule, white opaque cap and body, with “X-396” on the cap and “25 mg” on the body printed in blue ink. | Bottles of 30 | 83076-1025-3 |
| 100 mg | Size 0 capsule, blue opaque cap and yellow opaque body, with “X- 396” on the cap and “100 mg” on the body printed in white ink. | Bottles of 60 | 83076-1100-6 |
Store at controlled room temperature 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15ºC to 30ºC (59ºF to 86ºF) [see USP Controlled Room Temperature] . Store and dispense in the original bottle with desiccant to protect from moisture. Do not remove desiccant from bottle. Keep out of reach of children
12.1 Mechanism of Action
Ensartinib is a kinase inhibitor of anaplastic lymphoma kinase (ALK) and inhibits other kinases including MET and ROS1. In vitro, ensartinib inhibited phosphorylation of ALK and its downstream signaling proteins AKT, ERK, and S6, thereby blocking ALK-mediated signaling pathways and inhibiting proliferation in cell lines harboring ALK fusions and mutations. In vivo, ensartinib showed anti-tumor activity in a mouse xenograft model of human NSCLC harboring an ALK fusion.