Get your patient on Eovist - Gadoxetate Disodium injection, Solution (Gadoxetate Disodium)
Eovist - Gadoxetate Disodium injection, Solution prescribing information
WARNING: RISK ASSOCIATED WITH INTRATHECAL USE and NEPHROGENIC SYSTEMIC FIBROSIS
Risk Associated with Intrathecal Use
Intrathecal administration of gadolinium-based contrast agents (GBCAs) can cause serious adverse reactions including death, coma, encephalopathy, and seizures. EOVIST is not approved for intrathecal use [see Warnings and Precautions (5.1) ].
Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of EOVIST in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs.
The risk for NSF appears highest among patients with:
- Chronic, severe kidney disease (GFR < 30 mL/min/1.73m 2 ), or
- Acute kidney injury.
Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing.
For patients at highest risk for NSF, do not exceed the recommended EOVIST dose and allow a sufficient period of time for elimination of the drug from the body prior to any re-administration [see Warnings and Precautions (5.2) ].
INDICATIONS AND USAGE
EOVIST is indicated for use in magnetic resonance imaging (MRI) of the liver to detect and characterize lesions in adult and pediatric patients, including term neonates, with known or suspected focal liver disease.
DOSAGE AND ADMINISTRATION
Recommended Dose
The recommended dose of EOVIST for adult and pediatric patients, including term neonates, is 0.025 mmol/kg actual body weight (equivalent to 0.1 mL/kg) administered intravenously at a recommended rate of 1 mL/sec to 2 mL/sec.
Administration and Drug Handling
- EOVIST is for intravenous use only and must not be administered intrathecally [see Warnings and Precautions (5.1) ].
- Use aseptic technique when preparing and administering EOVIST.
- Visually inspect EOVIST for particulate matter and discoloration prior to administration. Do not use the solution if it is discolored or if particulate matter is present.
- Use EOVIST immediately after obtaining appropriate dose from vial.
- Pierce the rubber stopper only once. Discard any unused portion of an EOVIST vial.
- Do not mix EOVIST with other medications and do not administer EOVIST in the same intravenous line simultaneously with other medications.
- Flush the intravenous cannula with 0.9% Sodium Chloride Injection after EOVIST injection.
Imaging
- Liver lesions are detected and characterized with pre-contrast MRI and EOVIST MRI obtained during dynamic and hepatocyte imaging phases. Perform a pre-contrast MRI, inject EOVIST, and begin dynamic imaging approximately 15 seconds to 25 seconds after completion of the injection. Dynamic imaging consists of the arterial, the porto-venous (approximately 60 seconds post-injection), and the blood equilibrium (approximately 120 seconds) phases.
- Begin the hepatocyte imaging phase approximately 20 minutes post-injection. Hepatocyte phase imaging may be performed up to 120 minutes post-injection.
- Elevated intrinsic levels of bilirubin (>3 mg/dL) or ferritin can reduce the hepatic contrast effect of EOVIST. Perform MR imaging no later than 60 minutes following EOVIST administration to patients with these laboratory abnormalities, including patients who have elevated ferritin levels due to hemodialysis [see Warnings and Precautions (5.8) and Use in Specific Populations (8.6 , 8.7) ].
- Lesions with no or minimal hepatocyte function (cysts, metastases, and the majority of hepatocellular carcinomas) generally will not accumulate EOVIST. Well-differentiated hepatocellular carcinoma may contain functioning hepatocytes and can show some enhancement in the hepatocyte imaging phase. Additional clinical information is therefore needed to support a diagnosis of hepatocellular carcinoma.
DOSAGE FORMS AND STRENGTHS
Injection: 0.25 mmol/mL of gadoxetate disodium as a clear and colorless to pale yellow solution available in the following strengths:
- 2.5 mmol/10 mL (0.25 mmol/mL) in single-dose vial
- 3.75 mmol/15 mL (0.25 mmol/mL) in single-dose vial
USE IN SPECIFIC POPULATIONS
Pregnancy: Use only if imaging is essential during pregnancy and cannot be delayed. (8.1 )
Pregnancy
Risk Summary
GBCAs cross the placenta and result in fetal exposure. In human placental imaging studies, contrast was visualized in the placenta and fetal tissues after maternal GBCA administration. Based on animal studies, use of GBCAs during pregnancy may result in fetal gadolinium retention.
Published epidemiological studies on the association between GBCAs and adverse fetal outcomes have reported inconsistent findings and have important methodological limitations (see Data ).
In animal reproduction studies, no teratogenicity was observed with repeated daily intravenous administration of gadoxetate disodium to rats during organogenesis at doses up to 32 times the recommended single human dose; however, an increase in preimplantation loss was noted at doses 3.2 times the single human dose. Post implantation loss was observed with repeated daily intravenous administration of gadoxetate disodium to rabbits on gestation days 6 through 18 at doses 26 times the recommended single human dose (see Data ) . Because of the potential risks of gadolinium to the fetus, use EOVIST only if imaging is essential during pregnancy and cannot be delayed.
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
Available data regarding exposure to GBCAs during pregnancy from published epidemiological studies are not sufficient to assess the risk of adverse fetal and neonatal effects that may be associated with GBCAs. A retrospective cohort study of over 1.4 million pregnancies in Ontario, Canada, comparing pregnant women who had a GBCA MRI to pregnant women who did not have an MRI, reported a higher occurrence of stillbirths and neonatal deaths in the group receiving GBCA MRI. Limitations of this study include a lack of comparison with non-contrast MRI and lack of information about the maternal indication for MRI. Another retrospective cohort study of over 11 million pregnancies in the Medicaid database found no increased risk of fetal or neonatal death or Neonatal Intensive Care Unit admission when comparing pregnancies exposed to GBCA MRI versus non-contrast MRI. These two retrospective studies assessed a limited number of potential pregnancy outcomes and did not evaluate the full spectrum of potential fetal risk.
Animal Data
Gadolinium Retention
GBCAs administered to pregnant non-human primates (0.1 mmol/kg on gestational days 85 and 135) result in measurable gadolinium concentration in the offspring in bone, brain, skin, liver, kidney, and spleen for at least 7 months. GBCAs administered to pregnant mice (2 mmol/kg daily on gestational days 16 through 19) result in measurable gadolinium concentrations in the pups in bone, brain, kidney, liver, blood, muscle, and spleen at one month postnatal age.
Reproductive Toxicology
Animal reproductive and developmental toxicity studies were done in rats and rabbits. Gadoxetate disodium was not teratogenic when given intravenously during organogenesis to pregnant rats at doses up to 32 times the recommended single human dose (mmol/m 2 basis). However, an increase in preimplantation loss was noted at 3.2 times the human dose (mmol/m 2 basis). Compared to untreated controls, rates of post implantation loss and absorption increased and litter size decreased when pregnant rabbits received gadoxetate disodium at doses 26 times the recommended human single dose (mmol/m 2 basis). This occurred without evidence of maternal toxicity. Because pregnant animals received repeated daily doses of gadoxetate disodium, their overall exposure was significantly higher than that achieved with the standard single dose administered to humans.
Lactation
Risk Summary
There is no information regarding the presence of gadoxetate disodium in human milk, the effects of the drug in a breastfed infant, or the effects of the drug on milk production. However, published lactation data on other GBCAs report that 0.01 to 0.04% of the maternal gadolinium dose is present in breast milk and there is limited GBCA gastrointestinal absorption in the breastfed infant. In rat lactation studies with [ 153 Gd] gadoxetate disodium, less than 0.5% of the total administered radioactivity was transferred to the nursing pup (see Data ).
Clinical Considerations
A lactating woman may consider interrupting breastfeeding and pumping and discarding breast milk for up to 10 hours after EOVIST administration in order to minimize exposure to a breastfed infant.
Data
In lactating rats given 0.1 mmol/kg [ 153 Gd] gadoxetate disodium, less than 0.5% of the total administered radioactivity was transferred to the neonates via maternal milk, mostly within 2 hours.
Pediatric Use
The safety and effectiveness of EOVIST for magnetic resonance imaging (MRI) of the liver to detect and characterize lesions have been established in pediatric patients, including term neonates. Use of EOVIST in this age group is supported by evidence from adequate and well-controlled studies in adults and an observational study in 52 pediatric patients between 2 months of age and 18 years of age who were referred for evaluation of suspected or known focal liver lesions. In this observational study, EOVIST improved border delineation and increased contrast of the primary lesion in the majority of patients when compared to non-contrast images [see Adverse Reactions (6.1) and Clinical Studies (14) ] .
The safety and effectiveness of EOVIST have not been established in preterm neonates.
Geriatric Use
In clinical studies of EOVIST, 674 (34%) patients were 65 years of age and over, while 20 (1%) were 80 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences between the elderly and younger patients.
In general, use of EOVIST in an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal or cardiac function and of concomitant disease or other drug therapy.
Renal Impairment
In patients with renal impairment, the exposure of gadoxetate is increased compared to patients with normal renal function. This may increase the risk of adverse reactions such as nephrogenic systemic fibrosis (NSF). EOVIST can be removed by hemodialysis [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3) ] .
In patients with end-stage renal failure, hepatic contrast was reduced, which was attributed to significantly elevated serum ferritin levels [see Dosage and Administration (2.3) and Warnings and Precautions (5.8) ].
In patients with moderate renal impairment, hepatic contrast did not differ among the groups.
Hepatic Impairment
In patients with severe hepatic impairment, the exposure of gadoxetate is increased and EOVIST imaging performance may be impaired [see Dosage and Administration (2.3) , Warnings and Precautions (5.8) , and Clinical Pharmacology (12.3) ] .
In patients with mild or moderate hepatic impairment, the exposure of gadoxetate was moderately increased compared to healthy subjects with normal liver function, but hepatic contrast signal did not differ among the groups.
A dose adjustment is not necessary for patients with hepatic impairment.
In clinical studies, 489 patients had a diagnosis of liver cirrhosis (Child-Pugh category A, n = 270; category B, n = 98; category C, n = 24; unknown category, n = 97). No difference in diagnostic performance and safety was observed among these patients.
CONTRAINDICATIONS
EOVIST is contraindicated in patients with history of severe hypersensitivity reactions to EOVIST [see Warnings and Precautions (5.3) ] .
WARNINGS AND PRECAUTIONS
- Hypersensitivity Reactions: Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory and cutaneous manifestations, ranging from mild to severe reactions including shock can occur. Monitor patients closely for need of emergency cardiorespiratory support (5.3 )
- Gadolinium Retention: Gadolinium is retained for months or years in brain, bone, and other organs. (5.4 )
Risk Associated with Intrathecal Use
Intrathecal administration of GBCAs can cause serious adverse reactions including death, coma, encephalopathy, and seizures. The safety and effectiveness of EOVIST have not been established with intrathecal use. EOVIST is not approved for intrathecal use [see Dosage and Administration (2.2) ].
Nephrogenic Systemic Fibrosis
GBCAs increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of EOVIST among these patients unless the diagnostic information is essential and not available with non-contrast enhanced MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR < 30 mL/min/1.73m 2 ) as well as patients with acute kidney injury. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30 to 59 mL/min/1.73m 2 ) and little, if any, for patients with chronic, mild kidney disease (GFR 60 to 89 mL/min/1.73m 2 ). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. Report any diagnosis of NSF following EOVIST administration to Bayer HealthCare (1-888-842-2937) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days) and usually reversible decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (for example, age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing.
Among the factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA and degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administrated to a patient. For patients at highest risk for NSF, do not exceed the recommended EOVIST dose and allow a sufficient period of time for elimination of the drug prior to any re-administration. For patients receiving hemodialysis, consider the prompt initiation of hemodialysis following the administration of a GBCA in order to enhance the contrast agent's elimination [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3) ]. The usefulness of hemodialysis in the prevention of NSF is unknown.
Hypersensitivity Reactions
Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory and cutaneous manifestations, ranging from mild to severe, including shock have occurred following EOVIST administration. Most hypersensitivity reactions to EOVIST have occurred within half an hour after administration. Delayed reactions can occur up to several days after EOVIST administration [see Adverse Reactions (6.2) ] .
- Before EOVIST administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to EOVIST.
- EOVIST is contraindicated in patients with history of hypersensitivity reactions to EOVIST [see Contraindications (4) ].
- Administer EOVIST only in situations where trained personnel and therapies are promptly available for the treatment of hypersensitivity reactions, including personnel trained in resuscitation.
- Observe patients for signs and symptoms of hypersensitivity reactions during and following EOVIST administration.
Gadolinium Retention
Gadolinium is retained for months or years in several organs. The highest concentrations (nanomoles per gram of tissue) have been identified in the bone, followed by other organs (for example, brain, skin, kidney, liver, and spleen). The duration of retention also varies by tissue and is longest in bone. Linear GBCAs cause more retention than macrocyclic GBCAs. At equivalent doses, gadolinium retention varies among the linear agents with gadodiamide causing greater retention than other linear agents such as gadoxetate disodium and gadobenate dimeglumine. Retention is lowest and similar among the macrocyclic GBCAs such as gadoterate meglumine, gadobutrol, gadoteridol, and gadopiclenol.
Consequences of gadolinium retention in the brain have not been established. Pathologic and clinical consequences of GBCA administration and retention in skin and other organs have been established in patients with impaired renal function [see Warnings and Precautions (5.2) ] . There are rare reports of pathologic skin changes in patients with normal renal function. Adverse events involving multiple organ systems have been reported in patients with normal renal function without an established causal link to gadolinium retention [see Adverse Reactions (6.2) ] .
While clinical consequences of gadolinium retention have not been established in patients with normal renal function, certain patients might be at higher risk. These include patients requiring multiple lifetime doses, pregnant and pediatric patients, and patients with inflammatory conditions. Consider the retention characteristics of the agent when choosing a GBCA for these patients. Minimize repetitive GBCA imaging studies particularly closely spaced studies, when possible.
Acute Kidney Injury
In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of some GBCAs. The risk of acute kidney injury might be lower with EOVIST due to its dual excretory pathways. Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses.
Extravasation and Injection Site Reactions
Injection site reactions such as pain have been reported in clinical studies with EOVIST [see Adverse Reactions (6.1) ]. Extravasation into tissues during EOVIST administration may result in local tissue reactions such as myocyte necrosis and inflammation [see Nonclinical Toxicology (13.2) ] . Ensure catheter and venous patency before the injection of EOVIST.
Interference with Laboratory Tests
EOVIST can interfere with serum iron determination using complexometric methods (for example, ferrocene complexation method) [see Drug Interactions (7) ].
Interference with Visualization of Liver Lesions
Severe renal or hepatic failure may impair EOVIST imaging performance. In patients with end-stage renal failure, hepatic contrast was markedly reduced and was attributed to elevated serum ferritin levels. In patients with abnormally high (>3 mg/dL) serum bilirubin, reduced hepatic contrast was observed. If EOVIST is used in these patients, complete MRI no later than 60 minutes after EOVIST administration and use a paired non-contrast and contrast MRI set for diagnosis [see Dosage and Administration (2.3) ] .
ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed elsewhere in the labeling:
- Nephrogenic systemic fibrosis [see Warnings and Precautions (5.2) ]
- Hypersensitivity reactions [see Contraindications (4) and Warnings and Precautions (5.3) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Adverse Reactions in Adults
The safety of EOVIST was evaluated in 1,989 adult subjects who received EOVIST in clinical trials. Overall, 59% of the subjects were male, and the racial and ethnic distribution was 64% White, 22% Asian, 3% Hispanic or Latino, 2% Black or African American, and 0.5% other ethnic groups. The average age was 57 years (age range from 19 to 84 years).
Table 1 lists adverse reactions that occurred in ≥ 0.1% of subjects who received the recommended dose of EOVIST in clinical trials.
| Adverse Reaction | EOVIST n = 1,581 Rate (%) |
|---|---|
| Nausea | 1.1 |
| Headache | 1.1 |
| Feeling hot | 0.8 |
| Dizziness | 0.6 |
| Back pain | 0.6 |
| Vomiting | 0.4 |
| Blood pressure increased | 0.4 |
| Injection site reactions (pain, burning, coldness, extravasation, irritation) | 0.4 |
| Dysgeusia | 0.4 |
| Paresthesia | 0.3 |
| Flushing | 0.3 |
| Parosmia | 0.3 |
| Pruritus (generalized, eye) | 0.3 |
| Rash | 0.3 |
| Respiratory disorders (dyspnea, respiratory distress) | 0.2 |
| Fatigue | 0.2 |
| Chest pain | 0.1 |
| Vertigo | 0.1 |
| Dry mouth | 0.1 |
| Chills | 0.1 |
| Feeling abnormal | 0.1 |
Adverse reactions that occurred with a frequency of < 0.1% in subjects who received EOVIST include: tremor, akathisia, bundle branch block, palpitation, oral discomfort, salivary hypersecretion, maculopapular rash, hyperhidrosis, discomfort, and malaise.
Elevation of serum iron values and serum bilirubin laboratory values were reported in < 1% of subjects after administration of EOVIST. The values did not exceed more than 3 times the baseline values and returned to baseline within 1 to 4 days.
Adverse Reactions in Pediatric Patients
In a study of EOVIST in 52 pediatric patients between 2 months of age and 18 years of age, no new safety signals were observed [see Use in Specific Populations (8.4) ].
Postmarketing Experience
The following additional adverse reactions have been identified during the postmarketing use of EOVIST or other GBCAs. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: Tachycardia
Gastrointestinal Disorders: Acute pancreatitis with onset within 48 hours after GBCA administration
General Disorders and Administration Site Conditions: Fatigue, asthenia, pain syndromes, and heterogeneous clusters of symptoms in the neurological, cutaneous, and musculoskeletal systems with variable onset and duration after GBCA administration [see Warnings and Precautions (5.4) ]
Immune System Disorders: Hypersensitivity reactions (anaphylactic shock, hypotension, pharyngolaryngeal edema, urticaria, face edema, rhinitis, conjunctivitis, abdominal pain, hypoesthesia, sneezing, cough and pallor)
Psychiatric Disorders: Restlessness
Renal Disorders: Nephrogenic systemic fibrosis
Respiratory, Thoracic, and Mediastinal Disorders: Acute respiratory distress syndrome, pulmonary edema
Skin Disorders: Gadolinium associated plaques
DRUG INTERACTIONS
Serum Iron Test
EOVIST contains caloxetate trisodium that can interfere with serum iron determination using complexometric methods (for example, ferrocene complexation method) and may result in falsely high or low values for up to 24 hours after the administration of EOVIST. Conduct serum iron tests either before or at least 24 hours following administration of EOVIST.
DESCRIPTION
EOVIST (gadoxetate disodium) injection is a paramagnetic gadolinium-based contrast agent for intravenous use.
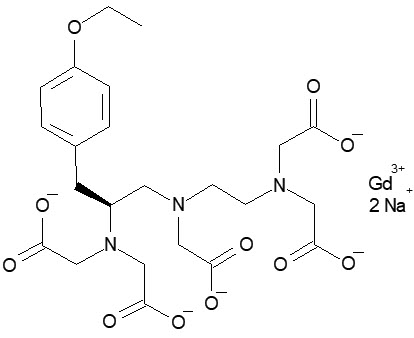
Gadoxetate disodium (Gd-EOB-DTPA) is a highly water-soluble, hydrophilic compound with a lipophilic moiety, the ethoxybenzyl group (EOB). EOB-DTPA forms a stable complex with the paramagnetic gadolinium ion with a thermodynamic stability of log KGdL=-23.46. The chemical name for gadoxetate disodium is (4S)-4-(4-Ethoxybenzyl)-3,6,9-tris(carboxylatomethyl)-3,6,9-triazaundecanedioic acid, gadolinium complex, disodium salt. Gadoxetate disodium has a molecular weight of 725.72 and an empirical formula of GdC 23 H 28 N 3 O 11 Na 2 . The structural formula of gadoxetate disodium is:
EOVIST is a sterile, clear, colorless to pale yellow solution. Each mL contains 181.43 mg (0.25 mmol) of gadoxetate disodium (containing 0.25 mmol of gadolinium) and the following inactive ingredients: 1 mg of caloxetate trisodium, 1.21 mg of trometamol, hydrochloric acid and/or sodium hydroxide (for pH adjustment), and water for injection. EOVIST contains no antimicrobial preservative.
Pertinent physiochemical properties of EOVIST are provided in Table 2.
| Parameter | Value |
|---|---|
| Osmolality at 37°C (Osm/kg H 2 O) | 0.688 |
| Viscosity at 37°C (cP) | 1.19 |
| Density at 37°C (g/mL) | 1.088 |
| pH | 6.8-8 |
CLINICAL PHARMACOLOGY
Mechanism of Action
Gadoxetate is a paramagnetic molecule that develops a magnetic moment when placed in a magnetic field. The magnetic moment alters the relaxation rates of water protons in its vicinity in the body, leading to an increase in signal intensity (brightness) of tissues. Gadoxetate is selectively taken up by hepatocytes, resulting in increased signal intensity in liver tissue.
Pharmacodynamics
In MRI, visualization of normal and pathological tissue depends in part on variations in the radiofrequency signal intensity that occurs with:
- Differences in proton density
- Differences of the spin-lattice or longitudinal relaxation times (T 1 )
- Differences in the spin-spin or transverse relaxation time (T 2 )
When placed in a magnetic field, gadoxetate shortens the T 1 and T 2 relaxation times in targeted tissues. The extent to which a contrast agent can affect the relaxation rate of tissue water (1/T 1 or 1/T 2 ) is termed relaxivity (r 1 or r 2 ). The relaxivity of gadoxetate in human plasma is about 6.9 L/mmol/sec at pH 7, 37°C and 1.5 T.
Pharmacokinetics
Distribution
Gadoxetate exhibits a biphasic mode of action: first, distribution in the extracellular space after injection and subsequently, selective uptake by hepatocytes (and biliary excretion) due to the lipophilic (EOB) moiety. After intravenous administration, the plasma concentration time profile of gadoxetate is characterized by a bi-exponential decline. The total distribution volume of gadoxetate at steady state is about 0.21 L/kg (extracellular space); plasma protein binding is less than 10%. Following GBCA administration, gadolinium is present for months or years in brain, bone, skin, and other organs [see Warnings and Precautions (5.4) ].
Elimination
The mean terminal elimination half-life of gadoxetate (0.01 to 0.1 mmol/kg) has been observed in healthy subjects of 22 to 39 years of age to be 0.91 to 0.95 hour. The pharmacokinetics are dose-linear up to a dose of 0.1 mmol/kg (4 times the recommended dose) .
A total serum clearance (Cl tot ) was 250 mL/min, whereas the renal clearance (Cl r ) corresponds to about 120 mL/min, a value similar to the glomerular filtration rate in healthy subjects.
Metabolism
Gadoxetate is not metabolized.
Excretion
Gadoxetate is equally eliminated via the renal and hepatobiliary routes.
Specific Populations
Geriatric Patients
In a clinical pharmacology study, the AUC and terminal half-life of gadoxetate were slightly increased (1.2-fold and 1.3-fold, respectively) and total clearance was decreased by 0.8-fold in geriatric patients compared to non-geriatric patients.
Patients with Renal Impairment
In a study of patients with end-stage renal failure, the AUC was increased about 6-fold and the terminal half-life was prolonged about 12-fold. Approximately 30% of the injected dose was removed by dialysis in a single 3-hour dialysis session, which started 1 hour after an EOVIST dose. Gadoxetate was almost completely eliminated via hemodialysis and biliary excretion within the observation period of 6 days, predominantly within the first 3 days [see Use in Specific Populations (8.6) ] .
In patients with moderate renal impairment, a moderate increase in AUC and terminal half-life (1.5-fold and 1.2-fold, respectively) was observed in comparison to healthy subjects with normal renal function.
Patients with Hepatic Impairment
In patients with severe hepatic impairment, especially in patients with abnormally high (> 3 mg/dL) serum bilirubin levels, the AUC of gadoxetate was increased up to 60% and the elimination half-life was increased up to 49%. The hepatobiliary excretion substantially decreased to about 5% of the administered dose [see Use in Specific Populations (8.7) ] .
In patients with mild or moderate hepatic impairment, a slight to moderate increase in plasma AUC (1.6-fold), half-life (1.1-fold), and urinary excretion (1.3-fold), as well as decrease in hepatobiliary excretion (0.7-fold) was observed in comparison to healthy subjects with normal liver function.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No carcinogenicity studies of EOVIST have been conducted.
Mutagenesis
Gadoxetate disodium was not mutagenic in in vitro reverse mutation tests in bacteria, or in chromosome aberration tests in human peripheral blood lymphocytes, and was negative in an in vivo micronucleus test in mice after intravenous injection of doses up to 4 mmol/kg.
Impairment of Fertility
Gadoxetate disodium had no effect on fertility and general reproductive performance of male and female rats when given in doses 6.5 times the human dose (based on body surface area).
Animal Toxicology and/or Pharmacology
A dose-related increase in QTc which was resolved by 30 minutes post dosing was observed in dogs when given a single dose of EOVIST. The increase was noted when given in doses equal to or greater than 0.1 mmol/kg (2.2 times the human dose). Maximum increase in QTcF was equal to or less than 20 ms at doses up to 0.5 mmol/kg (11 times the human dose).
A gait disturbance was observed in 1 of 3 mice when given EOVIST at a dose of approximately 1.1 mmol/kg (3.6 times the human dose); the disturbance occurred at 30 minutes post dosing and resolved at 4 hours post dosing.
Local intolerance reactions, including moderate interstitial hemorrhage, edema, and focal muscle fiber necrosis, were observed after intramuscular administration of EOVIST [see Warning and Precautions (5.5) ].
CLINICAL STUDIES
The effectiveness of EOVIST was evaluated in patients with suspected or known focal liver lesions in four non-randomized, intrapatient-controlled studies (i.e., Studies 1, 2, 3, and 4). Studies 1 and 2 ("detection" studies) evaluated predominantly the detection of liver lesions and enrolled patients who were scheduled for liver surgery. MRI results were compared to a reference standard that consisted of surgical histopathology and the results from intra-operative ultrasound of the liver. The studies assessed the sensitivity of pre-contrast MRI and EOVIST-contrasted MRI for the detection of liver lesions, when each set of images was compared to the reference.
Studies 3 and 4 ("characterization" studies) evaluated morphological characterization of liver lesions and enrolled patients with known or suspected focal liver lesions, including patients who were not scheduled for liver surgery. MRI results were compared to a reference standard that consisted of surgical histopathology and other prospectively defined criteria. The studies assessed the correctness of liver lesion characterization by pre-contrast MRI and EOVIST-contrasted MRI, when each set of images was compared to the reference. Lesions were characterized as one of the following choices: hepatocellular carcinoma, cholangiocarcinoma, metastasis, focal lymphoma, adenoma, focal nodular hyperplasia, hemangioma, abscess, focal liver fibrosis, regenerative nodule, focal fat, hydatid cyst, liver cyst, "not assessable", normal, no lesion or "other."
In all four studies, patients underwent a baseline, pre-contrast MRI followed by the administration of EOVIST at a dose of 0.025 mmol/kg, with MRI performed immediately (the "dynamic" phase) and at 10 minutes to 20 minutes following EOVIST administration (the "hepatocyte" phase). Patients also underwent computerized tomography with contrast examinations of the liver. Pre-contrast MRI and EOVIST-contrasted MR images were evaluated in a systematic, randomized, paired and unpaired fashion by three radiologists who were blinded to clinical information. CT images were also evaluated by the radiologists in a separate reading session.
Diagnostic efficacy was determined in 621 patients. The average age was 57 years (range 19 to 84 years) and 54% were male. The racial and ethnic representations were 90% White, 4% Black or African American, 3% Hispanic or Latino, 2% Asian, and 1% of other ethnic groups.
The combination of non-contrasted and EOVIST-contrasted MR images had improved sensitivity for the detection and characterization of liver lesions, compared to pre-contrasted MR images (Tables 3 and 4). The improved sensitivity in detection of lesions was predominantly related to the detection of additional lesions among patients with multiple lesions on the pre-contrast MR images. The false positive rates for detection of lesions were similar for non-contrasted MR images and EOVIST-contrasted MR images (32% versus 34%, respectively). Liver lesion detection and characterization results were similar between CT and the combination of pre-contrasted and EOVIST-contrasted MR images.
| Diagnostic Procedure | Reader | Study 1 Sensitivity (%) n=129 | Study 2 Sensitivity (%) n=126 |
|---|---|---|---|
| Pre-contrast MRI | Reader 1 | 76 | 77 |
| Reader 2 | 76 | 73 | |
| Reader 3 | 71 | 72 | |
| Combined pre- and EOVIST-contrast MRI | Reader 1 | 81 | 82 |
| Reader 2 | 78 | 76 | |
| Reader 3 | 74 | 78 | |
| Difference: combined pre + EOVIST-contrast MRI minus pre MRI (95% confidence interval) | Reader 1 | 5 (1, 9) Statistically significant improvement | 5 (1, 9) |
| Reader 2 | 2 (-1, 5) | 3 (-1, 7) | |
| Reader 3 | 3 (0, 6) | 6 (0, 10) |
| Diagnostic Procedure | Reader | Study 3 | Study 4 | ||
|---|---|---|---|---|---|
| n | Proportion correct (%) Proportion of correctly characterized lesions with respect to the reference | n | Proportion correct (%) | ||
| Pre-contrast MRI | Reader 1 | 182 | 51 | 177 | 60 |
| Reader 2 | 182 | 59 | 177 | 64 | |
| Reader 3 | 182 | 53 | 177 | 48 | |
| Combined pre- and EOVIST-contrast MRI | Reader 1 | 182 | 67 | 177 | 61 |
| Reader 2 | 182 | 76 | 177 | 76 | |
| Reader 3 | 182 | 58 | 177 | 67 | |
| Difference: combined pre- and EOVIST-contrast MRI minus pre-contrast MRI (95% confidence interval) | Reader 1 | 16 (7, 25) Statistically significant improvement | 1 (-7, 10) | ||
| Reader 2 | 17 (9, 25) | 11 (5, 18) | |||
| Reader 3 | 5 (-2, 12) | 19 (11, 27) | |||
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
EOVIST (gadoxetate disodium) injection is supplied at a concentration of 0.25 mmol/mL of gadoxetate disodium as a clear and colorless to pale yellow solution available in the following strengths:
- 2.5 mmol/10 mL (2.5 mmol/mL) in single-dose vials, boxes of 5 (NDC 50419-320-05)
- 3.75 mmol/15 mL (2.5 mmol/mL) in single-dose vials, boxes of 5 (NDC 50419-320-15)
Storage and Handling
Store at 20°C to 25° C (68°F to 77° F); excursions permitted to 15°C to 30° C (59°F to 86°F) [see USP Controlled Room Temperature].
Mechanism of Action
Gadoxetate is a paramagnetic molecule that develops a magnetic moment when placed in a magnetic field. The magnetic moment alters the relaxation rates of water protons in its vicinity in the body, leading to an increase in signal intensity (brightness) of tissues. Gadoxetate is selectively taken up by hepatocytes, resulting in increased signal intensity in liver tissue.
Eovist - Gadoxetate Disodium injection, Solution PubMed™ news
- Journal Article • 2026 AprClinical Safety of Gadoxetate Disodium: Insights From 20 Years of Use and More Than 12 Million Administrations.
- Journal Article • 2026 MarComparison of Gd-EOB-DTPA-enhanced MRI versus multiphasic enhanced CT for detecting occult recurrence after curative ablation of hepatocellular carcinoma.
- Journal Article • 2026 FebIntegration of quantitative MRI and liver function indices to predict early tumor response after stereotactic body radiotherapy for hepatocellular carcinoma: A prospective study.
- Journal Article • 2026 FebSafety of a Tailored Gadolinium-Based Contrast Agent Protocol Considering Excretion Pathways in Patients with Renal Impairment.
- Journal Article • 2026 FebThe Use of MRI With Double-Dose Eovist for the Detection of Bile Leaks.