Epinephrine - Epinephrine injection prescribing information
INDICATIONS AND USAGE
Epinephrine Injection is indicated for the emergency treatment of type I allergic reactions, including anaphylaxis in adult and pediatric patients who weigh 15 kg or greater.
DOSAGE AND ADMINISTRATION
Recommended Dosage
Recommended dosage for patients who weigh 15 kg or greater is based on weight and the dosage is provided in Table 1. Administer Epinephrine Injection intramuscularly or subcutaneously into the anterolateral aspect of the thigh.
Table 1 Recommended Dosage of Epinephrine Injection Based on Patient’s Weight
Patient’s Weight | Dosage |
30 kg or greater | 0.3 mg |
15 kg to less than 30 kg | 0.15 mg |
- Since the doses of epinephrine delivered from Epinephrine Injection are fixed, use other forms of injectable epinephrine if doses lower than 0.15 mg are deemed necessary.
- In the absence of clinical improvement or if symptoms worsen after the initial treatment, a second dose of Epinephrine Injection may be administered with a second auto-injector starting 5 minutes after the first dose.
- Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required.
- It is recommended that patients are prescribed and have immediate access to two Epinephrine Injection devices at all times.
Administration Instructions
Injection:
- 0.3 mg (0.3 mg/0.3 mL) of clear and colorless solution in single-dose pre-filled auto-injector
- 0.15 mg (0.15 mg/0.15 mL) of clear and colorless solution in single-dose pre-filled auto-injector
DOSAGE FORMS AND STRENGTHS
- Injection: 0.3 mg (0.3 mg/0.3 mL) of clear and colorless solution in single-dose pre-filled auto-injector
- Injection: 0.15 mg (0.15 mg/0.15 mL) of clear and colorless solution in single-dose pre-filled auto-injector
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Prolonged experience with epinephrine use in pregnant women over several decades, based on published literature, have not identified a drug associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. There are risks to the mother and fetus associated with anaphylaxis, and treatment with epinephrine should not be delayed (see Clinical Considerations) . In animal reproduction studies, epinephrine administered by the subcutaneous route to pregnant rabbits, mice, and hamsters, during the period of organogenesis was teratogenic at doses 7 times and higher than the maximum recommended human intramuscular and subcutaneous dose on a mg/m 2 basis (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and Embryo/Fetal Risk:
During pregnancy, anaphylaxis can be catastrophic and can lead to hypoxic-ischemic encephalopathy and permanent central nervous system damage or death in the mother and, more commonly, in the fetus or neonate. Treatment of anaphylaxis during pregnancy should not be delayed.
Data
Animal Data
In an embryofetal development study with rabbits dosed during the period of organogenesis, epinephrine was shown to be teratogenic (including gastroschisis and embryonic lethality) at doses approximately 40 times the maximum recommended intramuscular or subcutaneous dose (on a mg/m 2 basis at a maternal subcutaneous dose of 1.2 mg/kg/day for two to three days).
In an embryofetal development study with mice dosed during the period of organogenesis, epinephrine was shown to be teratogenic (including embryonic lethality) at doses approximately 8 times the maximum recommended intramuscular or subcutaneous dose (on a mg/m 2 basis at maternal subcutaneous dose of 1 mg/kg/day for 10 days). These effects were not seen in mice at approximately 4 times the maximum recommended daily intramuscular or subcutaneous dose (on a mg/m 2 basis at a subcutaneous maternal dose of 0.5 mg/kg/day for 10 days).
In an embryofetal development study with hamsters dosed during the period of organogenesis from gestation days 7 to 10, epinephrine was shown to be teratogenic at doses approximately 7 times the maximum recommended intramuscular or subcutaneous dose (on a mg/m 2 basis at a maternal subcutaneous dose of 0.5 mg/kg/day).
Lactation
Risk Summary
There is no information on the presence of epinephrine in human milk, the effects on the breastfed infants, or the effects on milk production. However, due to its poor oral bioavailability and short half-life, transfer of epinephrine into breastmilk is expected to be low. Treatment of anaphylaxis in breastfeeding patients should not be delayed.
Pediatric Use
The safety and effectiveness of Epinephrine Injection for the emergency treatment of type I allergic reactions, including anaphylaxis have been established in pediatric patients who weigh 15 kg or greater. The use of Epinephrine Injection for this indication is supported by clinical experience. Clinical experience with the use of epinephrine suggests that the adverse reactions seen in pediatric patients are similar in nature and extent to those both expected and reported in adults. Since the doses of epinephrine delivered from Epinephrine Injection are fixed, use other forms of injectable epinephrine if doses lower than 0.15 mg are deemed necessary.
The safety and effectiveness of Epinephrine Injection have not been established in pediatric patients who weigh less than 15 kg.
Geriatric Use
Clinical studies of Epinephrine Injection for emergency treatment of type I allergic reactions, including anaphylaxis, were not conducted in geriatric patients aged 65 and over to determine whether they respond differently from younger adult patients. However, other reported clinical experience with use of epinephrine for the treatment of anaphylaxis has identified that geriatric patients may be particularly sensitive to the effects of epinephrine. Therefore, these patients may be at greater risk for developing adverse reactions after epinephrine administration.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
Injection-Related Complications
Epinephrine Injection should only be injected into the anterolateral aspect of the thigh [see Dosage and Administration (2.2) ] .
Do not inject intravenously
Large doses or accidental intravenous injection of epinephrine may result in cerebral hemorrhage due to a sharp rise in blood pressure. Rapidly acting vasodilators can counteract the marked pressor effects of epinephrine for this inadvertent administration.
Do not inject into buttock
Injection into the buttock may not provide effective treatment of anaphylaxis. If Epinephrine Injection is injected into the buttock, advise the patient to administer a second dose of Epinephrine Injection into the anterolateral aspect of the thigh if symptoms worsen or persist, and then go immediately to the nearest emergency room for further treatment of anaphylaxis. Additionally, injection into the buttock has been associated with the development of Clostridial infections (gas gangrene). Cleansing with alcohol does not kill bacterial spores, and therefore, does not lower the risk.
Do not inject into digits, hands or feet
Since epinephrine is a strong vasoconstrictor, accidental injection into the digits, hands or feet may result in loss of blood flow to the affected area and may not provide effective treatment of anaphylaxis. Advise the patient to administer a second dose of Epinephrine Injection into the anterolateral aspect of the thigh if experiencing anaphylaxis, and then go immediately to the nearest emergency room and inform the healthcare provider in the emergency room of the location of the accidental injection. Treatment of such inadvertent administration should consist of vasodilation, in addition to further appropriate treatment of anaphylaxis [see Adverse Reactions (6)] .
Hold leg firmly during injection
To minimize the risk of injection related injury when administering Epinephrine Injection to young children or infants, instruct caregivers to hold the child’s leg firmly in place and limit movement prior to and during injection.
Serious Infections at the Injection Site
Rare cases of serious skin and soft tissue infections, including necrotizing fasciitis and myonecrosis caused by Clostridia (gas gangrene), have been reported at the injection site following epinephrine injection for anaphylaxis. Clostridium spores can be present on the skin and introduced into the deep tissue with subcutaneous or intramuscular injection. While cleansing with alcohol may reduce presence of bacteria on the skin, alcohol cleansing does not kill Clostridium spores. To decrease the risk of Clostridium infection, do not inject Epinephrine Injection into the buttock [see Warnings and Precautions (5.1) ] . Advise patients to seek medical care if they develop signs or symptoms of infection, such as persistent redness, warmth, swelling, or tenderness, at the epinephrine injection site.
Risks Associated with Use of Epinephrine in Certain Coexisting Conditions
Some patients may be at greater risk for developing adverse reactions after epinephrine administration. Despite these concerns, it should be recognized that the presence of these conditions is not a contraindication to epinephrine administration in an acute, life-threatening situation. Therefore, instruct patients with these conditions, and/or caregivers to the circumstances under which epinephrine should be used.
Administer epinephrine with caution to patients who have heart disease, including patients with cardiac arrhythmias, coronary artery or organic heart disease, or hypertension. In such patients, or in patients who are on drugs that may sensitize the heart to arrhythmias, epinephrine may precipitate or aggravate angina pectoris as well as produce ventricular arrhythmias [see Adverse Reactions (6) and Drug Interactions (7) ] .
Epinephrine can temporarily exacerbate the underlying condition or increase symptoms in
patients with the following: hyperthyroidism, Parkinson’s disease, diabetes, renal impairment.
Administer epinephrine with caution in patients with these conditions, including elderly patients and pregnant women.
Allergic Reactions Associated with Sulfite
Epinephrine is the preferred treatment for serious allergic reactions or other emergency situations even though Epinephrine Injection contains sodium bisulfite, a sulfite that may, in other products, cause allergic-type reactions including anaphylactic symptoms or life-threatening or less severe asthmatic episodes in certain susceptible persons. The alternatives to using epinephrine in a life-threatening situation may not be satisfactory. The presence of a sulfite in Epinephrine Injection should not deter administration of the drug for treatment of serious allergic or other emergency situations.
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Injection-Related Complications [see Warnings and Precautions (5.1) ]
- Serious Infections at the Injections [see Warnings and Precautions (5.2) ]
- Risks Associated with Use of Epinephrine in Certain Coexisting Conditions [see Warnings and Precautions (5.3) ]
- Allergic Reactions Associated with Sulfite [see Warnings and Precautions (5.4) ]
The following adverse reactions associated with the use of epinephrine were identified in observational trials, case reports, studies, or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular : angina, arrhythmias (including fatal ventricular fibrillation), cerebral hemorrhage, hypertension, pallor, palpitations, tachyarrhythmia, tachycardia, vasoconstriction, ventricular ectopy, and stress cardiomyopathy
Gastrointestinal Disorders : nausea and vomiting
Infections : Clostridial infections (gas gangrene)
Metabolism and Nutrition Disorders : transient hyperglycemia, sweating
Neurological: disorientation, impaired memory, panic, psychomotor agitation, sleepiness,
tingling, weakness, tremor, hypoesthesia, dizziness, headache
Psychiatric : anxiety, apprehensiveness, restlessness
Respiratory : respiratory difficulties
Skin and Subcutaneous Tissue Disorders: bruising, bleeding, discoloration, erythema, necrotizing fasciitis, myonecrosis
DRUG INTERACTIONS
Drugs Increasing Risk of Cardiac Arrhythmias
Patients who receive epinephrine while concomitantly taking cardiac glycosides, diuretics, or anti-arrhythmics should be observed carefully for the development of cardiac arrhythmias [see Warnings and Precautions (5.3) and Adverse Reactions (6) ] .
Drugs Potentiating Effects of Epinephrine
The effects of epinephrine may be potentiated by tricyclic antidepressants, monoamine oxidase inhibitors, levothyroxine sodium, and certain antihistamines, notably chlorpheniramine, tripelennamine, and diphenhydramine, and catechol-O-methyl transferase (COMT) inhibitors such as entacapone.
Drugs Antagonizing Effects of Epinephrine
The cardiostimulating and bronchodilating effects of epinephrine are antagonized by beta- adrenergic blocking drugs, such as propranolol.
The vasoconstricting and hypertensive effects of epinephrine are antagonized by alpha-adrenergic blocking drugs, such as phentolamine.
Ergot alkaloids may also reverse the pressor effects of epinephrine.
DESCRIPTION
Epinephrine Injection, USP 0.3 mg and 0.15 mg, are single-dose auto-injectors and combination products containing drug and device components.
Each Epinephrine Injection, USP 0.3 mg, delivers a single dose of 0.3 mg epinephrine from Epinephrine Injection, USP (0.3 mL) in a sterile solution.
Each Epinephrine Injection, USP 0.15 mg, delivers a single dose of 0.15 mg epinephrine from Epinephrine Injection, USP (0.15 mL) in a sterile solution.
Epinephrine Injection, USP 0.3 mg and Epinephrine Injection, USP 0.15 mg each contain 1.1 mL of epinephrine solution. 0.3 mL and 0.15 mL epinephrine solution are dispensed for Epinephrine Injection, USP 0.3 mg and Epinephrine Injection, USP 0.15 mg, respectively, when activated. The solution remaining after activation is not available for future use and should be discarded.
Each 0.3 mL in Epinephrine Injection, USP 0.3 mg, contains 0.3 mg epinephrine, 2.6 mg sodium chloride, not more than 1.5 mg chlorobutanol, 0.45 mg sodium bisulfite, hydrochloric acid and sodium hydroxide to adjust pH, and water for injection. The pH range is 2.2-5.0.
Each 0.15 mL in Epinephrine Injection, USP 0.15 mg, contains 0.15 mg epinephrine, 1.3 mg sodium chloride, not more than 0.75 mg chlorobutanol, 0.225 mg sodium bisulfite, hydrochloric acid and sodium hydroxide to adjust pH, and water for injection. The pH range is 2.2-5.0.
Epinephrine is a sympathomimetic catecholamine. Chemically, epinephrine is (-)-3,4-Dihydroxy-α-[(methylamino)methyl]benzyl alcohol with the following structure:
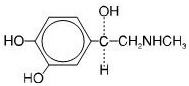
Epinephrine solution deteriorates rapidly on exposure to air or light, turning pink from oxidation to adrenochrome and brown from the formation of melanin. Replace Epinephrine Injection, USP if the epinephrine solution appears discolored (pinkish or brown color), cloudy, or contains particles.
CLINICAL PHARMACOLOGY
Mechanism of Action
Epinephrine acts on both alpha- and beta-adrenergic receptors.
Through its action on alpha-adrenergic receptors, epinephrine lessens the vasodilation and increased vascular permeability that occurs during anaphylaxis, which can lead to loss of intravascular fluid volume and hypotension.
Through its action on beta-adrenergic receptors, epinephrine causes bronchial smooth muscle relaxation and helps alleviate bronchospasm, wheezing, and dyspnea that may occur during anaphylaxis.
Epinephrine also alleviates pruritus, urticaria, and angioedema, and may relieve gastrointestinal and genitourinary symptoms associated with anaphylaxis because of its relaxer effects on the smooth muscle of the stomach, intestine, uterus and urinary bladder.
Pharmacodynamics
When given subcutaneously or intramuscularly, epinephrine has a rapid onset and short duration of action.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies to evaluate the carcinogenic potential of epinephrine have not been conducted.
Epinephrine and other catecholamines have been shown to have mutagenic potential in vitro . Epinephrine was positive in the Salmonella bacterial reverse mutation assay, positive in the mouse lymphoma assay, and negative in the in vivo micronucleus assay. Epinephrine is an oxidative mutagen based on the E. coli WP2 Mutoxitest bacterial reverse mutation assay. This should not prevent the use of epinephrine where indicated [see Indications and Usage (1) ] .
The potential for epinephrine to impair reproductive performance has not been evaluated, but epinephrine has been shown to decrease implantation in female rabbits dosed subcutaneously with 1.2 mg/kg/day (40-fold the highest human intramuscular or subcutaneous daily dose) during gestation days 3 to 9.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Epinephrine Injection is a clear and colorless solution for intramuscular or subcutaneous use. Epinephrine Injection is available as an auto-injector as described in Table 2.
Table 2 Epinephrine Injection Auto-injector Package Configurations and Strengths
Strength | Package Configuration | NDC |
0.3 mg/0.3 mL | Each carton contains 2 single-dose prefilled auto-injectors in individual carrier tubes | NDC 0115-1694-49 |
Auto-injector in carrier tube | NDC 0115-1694-30 | |
0.15 mg/0.15 mL | Each carton contains 2 single-dose prefilled auto-injectors in individual carrier tubes | NDC 0115-1695-49 |
Auto-injector in carrier tube | NDC 0115-1695-30 |
Storage and Handling
- Protect from light. Epinephrine Injection is light sensitive and should be stored in the individual carrier tube provided.
- Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
- Do not refrigerate or freeze.
- Before using, check to make sure the solution in the auto-injector is clear and colorless.
- Replace the auto-injector if the solution is discolored (pinkish or brown color), cloudy, or contains particles.
- Properly dispose of all used, unwanted, or expired Epinephrine Injection, USP.
Instructions for Use
EPINEPHRINE injection (ep″ i nef′ rin),
for intramuscular or subcutaneous use
Epinephrine Injection USP, 0.3 mg
Each Epinephrine Injection USP, 0.3 mg delivers a single dose of 0.3 mg epinephrine USP in 0.3 mL (0.3 mg/0.3 mL)
Epinephrine Injection USP, 0.15 mg
Each Epinephrine Injection USP, 0.15 mg delivers a single dose of 0.15 mg epinephrine USP in 0.15 mL (0.15 mg/0.15 mL)
For allergic emergencies (anaphylaxis)
This Instructions for Use contains information on how to properly administer Epinephrine Injection.
Your Epinephrine Injection
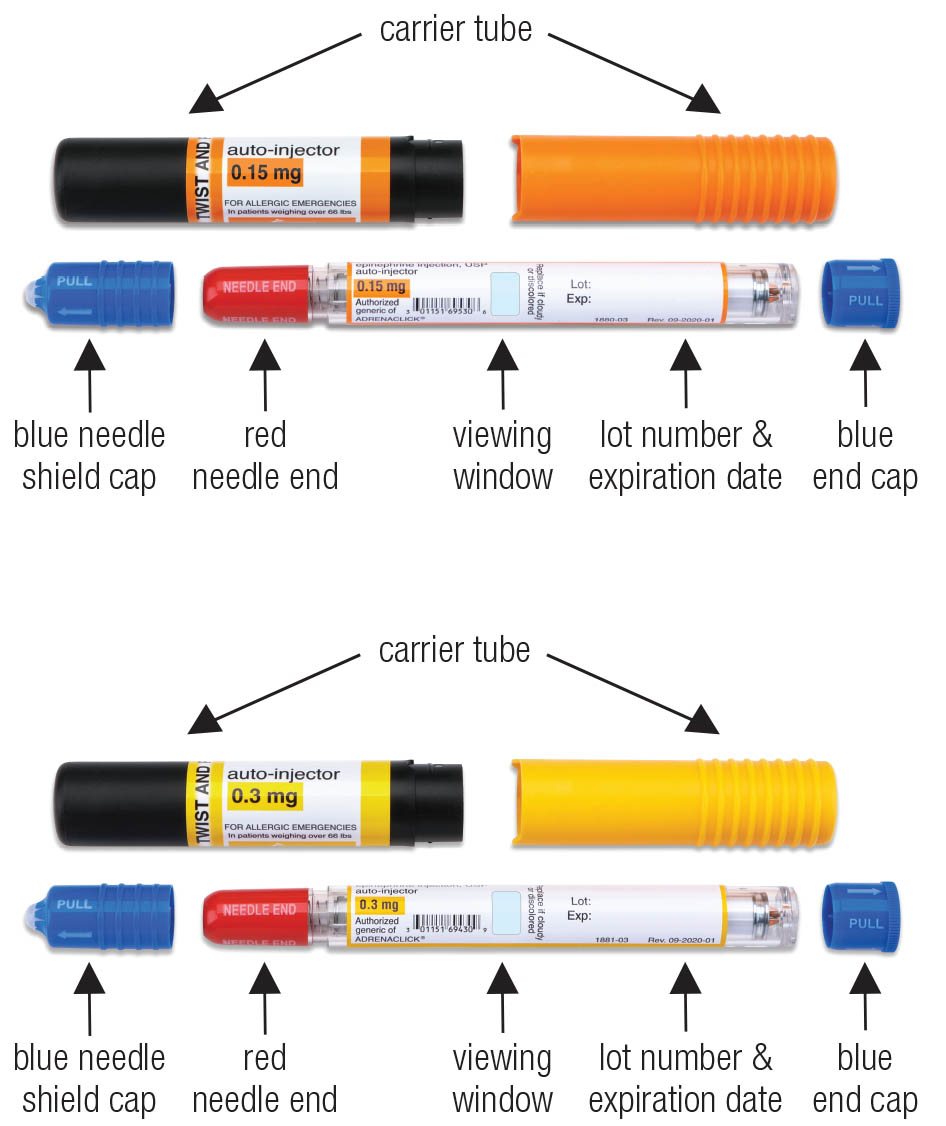
Figure A. Epinephrine Injection, USP 0.3 mg (yellow label) and
Epinephrine Injection, USP 0.15 mg (orange label), showing the auto-injector and carrier tube components
Important Information You Need to Know Before Administering Epinephrine Injection
- Epinephrine Injection is a single-dose auto-injector used for allergic emergencies (anaphylaxis) and should be used right away.
- Before you need to use your Epinephrine Injection, make sure your healthcare provider shows you the right way to use it. Anyone who may be able to administer Epinephrine Injection should also understand how to use it.
- Check the expiration date on your Epinephrine Injection, as shown in Figure A . Replace it before the expiration date.
- Read this Instructions for Use carefully in a non-emergency situation and make sure you understand them before using your Epinephrine Injection. If you have any questions, ask your healthcare provider.
- It is very important to hold Epinephrine Injection firmly in place for a full 10 seconds. If you do not hold it in place long enough, Epinephrine Injection may not have time to deliver the full dose of medicine.
- Always carry 2 Epinephrine Injection devices. One dose may not be enough to treat an allergic emergency. You may need to use a second Epinephrine Injection if symptoms continue or come back after the first dose. A second dose may be administered starting 5 minutes after the first dose while you wait for emergency medical help, or if the first Epinephrine Injection is activated before the dose can be given.
- Inject Epinephrine Injection into the muscle (intramuscular) or under the skin (subcutaneous) in the middle of the outer thigh (upper leg) (see Figure B). Do not inject Epinephrine Injection into any other part of the body.
- If you accidentally inject Epinephrine Injection into the fingers, toes, hands or feet , administer a second dose of Epinephrine Injection into the middle of the outer thigh (upper leg).
- Epinephrine Injection can be administered through clothing if needed.
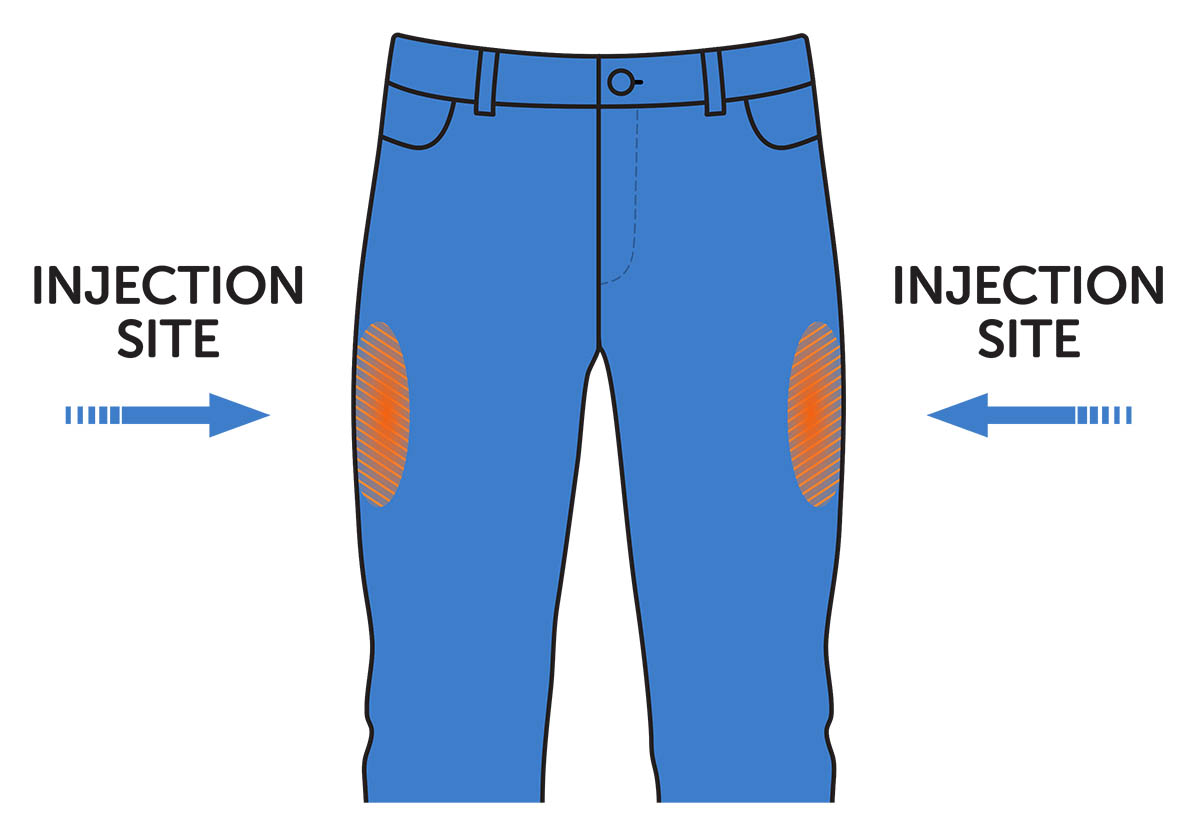
Figure B. Inject Epinephrine Injection into the middle of the outer thigh (upper leg)
Each Epinephrine Injection is for single use only.
The dose has been delivered if you see the needle sticking out of the red needle end after use. It is normal for most of the medicine to remain in the auto-injector after the dose is injected.
- Do not take the Epinephrine Injection apart.
- Do not remove the blue needle shield cap and blue end cap until you are ready to use Epinephrine Injection.
- Always point the red needle end downward (see Figure C ). Never put your thumb, fingers, or hand over the red needle end. The needle comes out of the red needle end.
- Accidental injection into the fingers, hands, or feet may cause loss of blood flow to these areas. If an accidental injection happens, get emergency medical help right away.
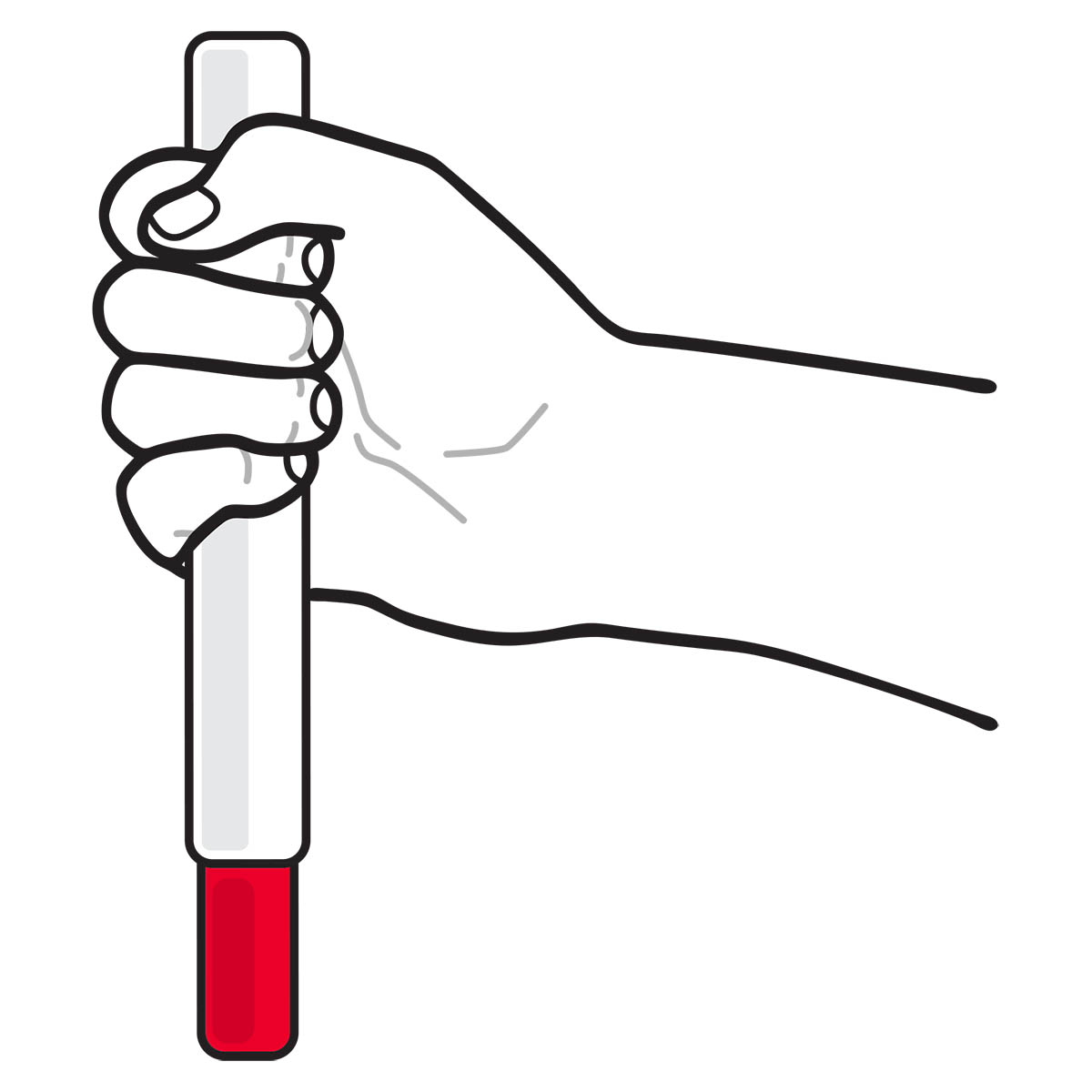
Figure C. Correct way to hold the Epinephrine Injection with red needle end pointing down
Preparing to Inject Epinephrine Injection
Read the following information while preparing to inject Epinephrine Injection:
- Epinephrine Injection can be administered through clothing. Remove any objects or clothing that may block access to the injection site.
- Check Epinephrine Injection before use. If the auto-injector appears damaged or broken, do not use it. Throw away (dispose of it) and use a new Epinephrine Injection.
- A separate Epinephrine Injection Trainer is available. The Trainer is beige in color and contains no medicine and no needle. Practice using the Trainer, but always carry your real Epinephrine Injection in case of an allergic emergency.
- You may request a separate Trainer at www.epinephrineautoinject.com or call 1-855-374-6374. Instructions for use of the Trainer device are provided on the Trainer label. Additional video instructions are available at www.epinephrineautoinject.com .
- Before injection, make sure you know where to inject (see Figure B ) and how to hold the Epinephrine Injection (see Figure C ).
Make sure Epinephrine Injection has not been used.
Epinephrine Injection has already been used if:
- the needle is sticking out of the red needle end, and
- the viewing window is blocked
Preparing to Inject a Child
- If you are administering Epinephrine Injection to a young child, hold their leg firmly in place before and during the 10 second injection to reduce the risk of injury.
- Ask your healthcare provider to show you how to:
- Remove the blue needle shield cap first and then the blue end cap (see Figure D)
- Hold the young child firmly in place (restrain movement).
- With 1 hand, grip the auto-injector with the RED needle end pointing down.
- Inject in the middle of the outer thigh (upper leg) (see Figure B ).
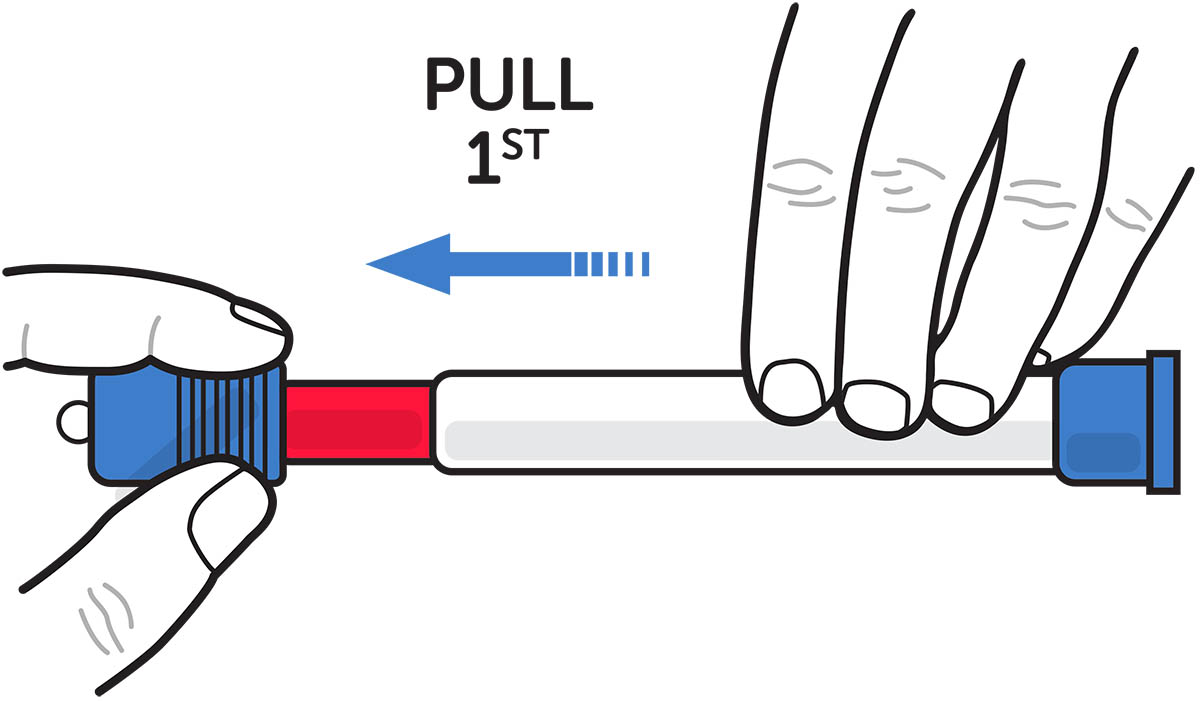
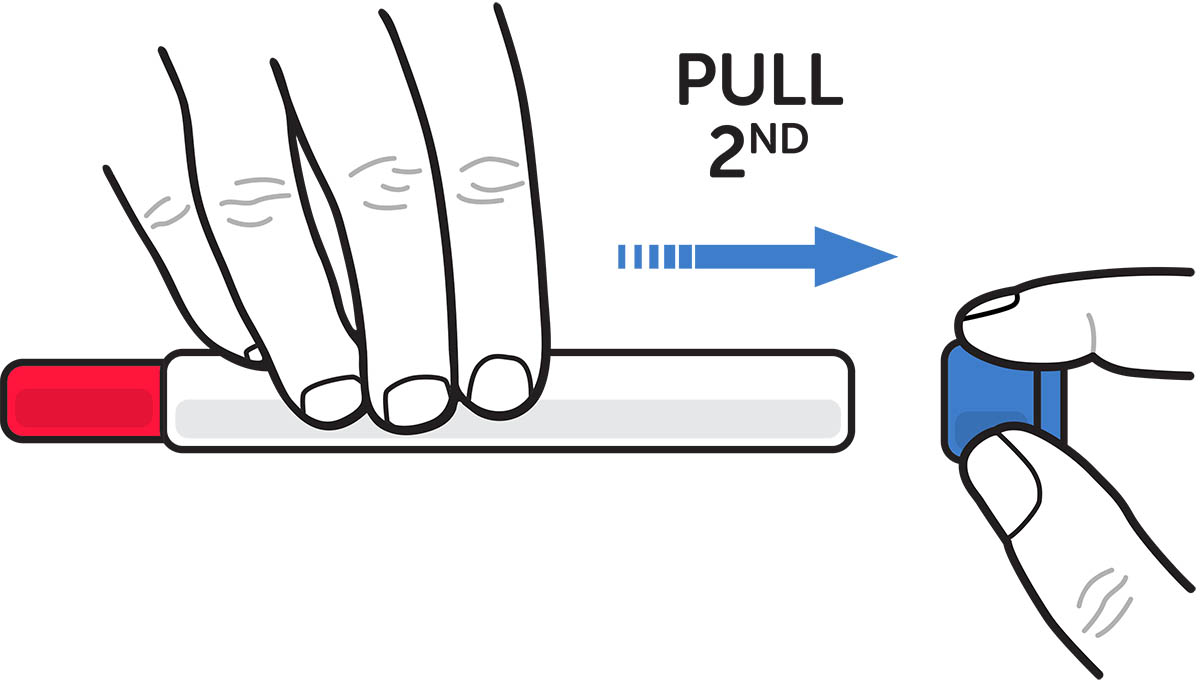
Figure D. Pull off the blue needle shield cap first and then the blue end cap
- Keep the Epinephrine Injection away from young children. The Epinephrine Injection is not a toy. Use by young children should be supervised by an adult.
- After the Epinephrine Injection is used, throw away both the blue needle shield cap and blue end cap immediately. These parts may pose a choking hazard for young children.
Checking the Solution Color
Examine the solution in the viewing window of your Epinephrine Injection regularly. See the information below:

Figure E. Solution Viewing Window
How to check the Solution Color:
- Remove Epinephrine Injection from the carrier tube. See Figure F.
- The solution can be seen through the viewing window located near the middle of the Epinephrine Injection. See Figure E.
- To check the solution color, hold the auto-injector in a well-lit area and look into the viewing window.
Solution Appearance
✓ Use Epinephrine Injection if the solution is clear and colorless.
X Do not use the Epinephrine Injection if the solution is discolored (pinkish or brown color), cloudy, or contains solid particles. Throw away (dispose of it) and use a new Epinephrine Injection (see “Disposing of an Expired, Unused or Used Epinephrine Injection” on the Patient Information).
Injecting Epinephrine Injection
Step 1. Remove Epinephrine Injection from the carrier tube. See Figure F.
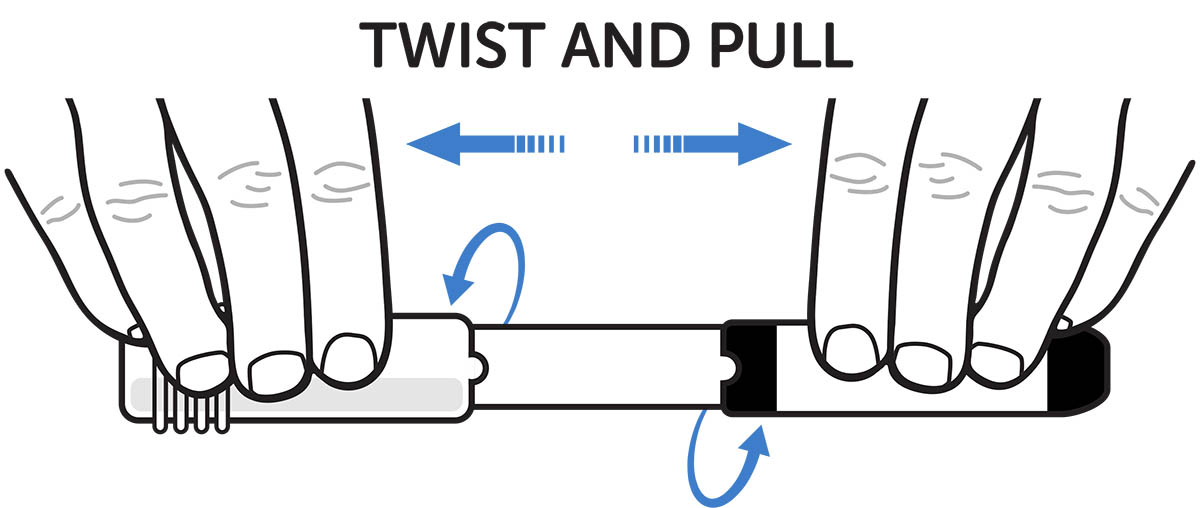
Figure F
Step 2. Pull off the blue needle shield cap first by grasping at the ribbed area and pulling. You will now see a red needle end. Then pull off the blue end cap. See Figure G.
Important:
- The needle comes out of the red needle end.
- To avoid an accidental injection, never put your thumb, fingers or hand over the red needle end.
- If an accidental injection occurs, administer a second dose of Epinephrine Injection into the middle of the outer thigh (upper leg) and get emergency medical help right away.
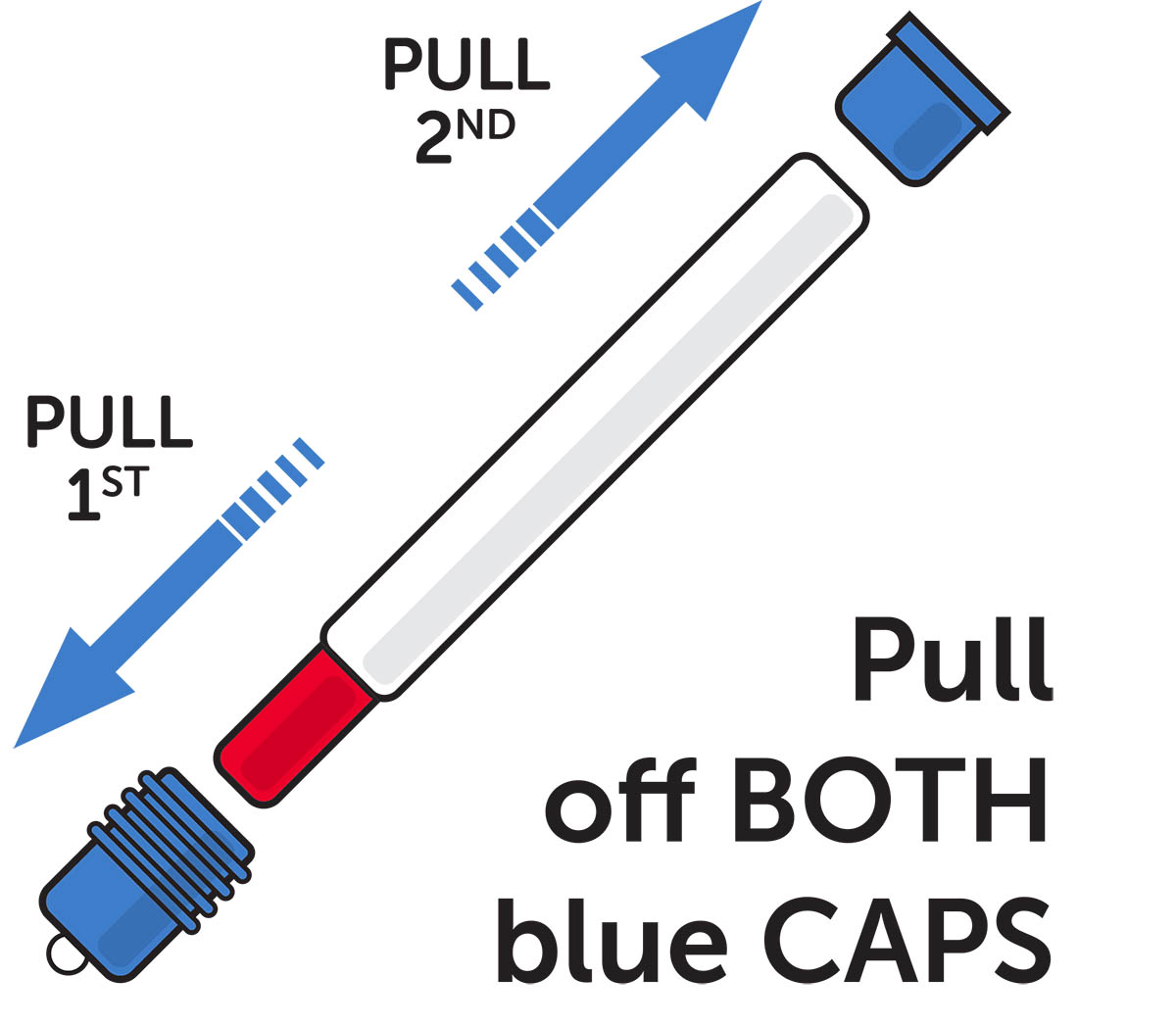
Figure G
Step 3. Inject the Epinephrine Injection
Self Administration (see Figure H) or Caregiver Administration (see Figure I)
- Grasp the Epinephrine Injection firmly in your fist with the red needle end pointing downward. See Figure C.
- Place the red needle end against the middle of the outer thigh (upper leg) at a 90° angle (perpendicular) to the thigh. The injection may be given through clothing if necessary.
- Press Down Hard and hold Epinephrine Injection firmly against the thigh for a full 10 seconds to deliver the medicine. See Figure H and Figure I.
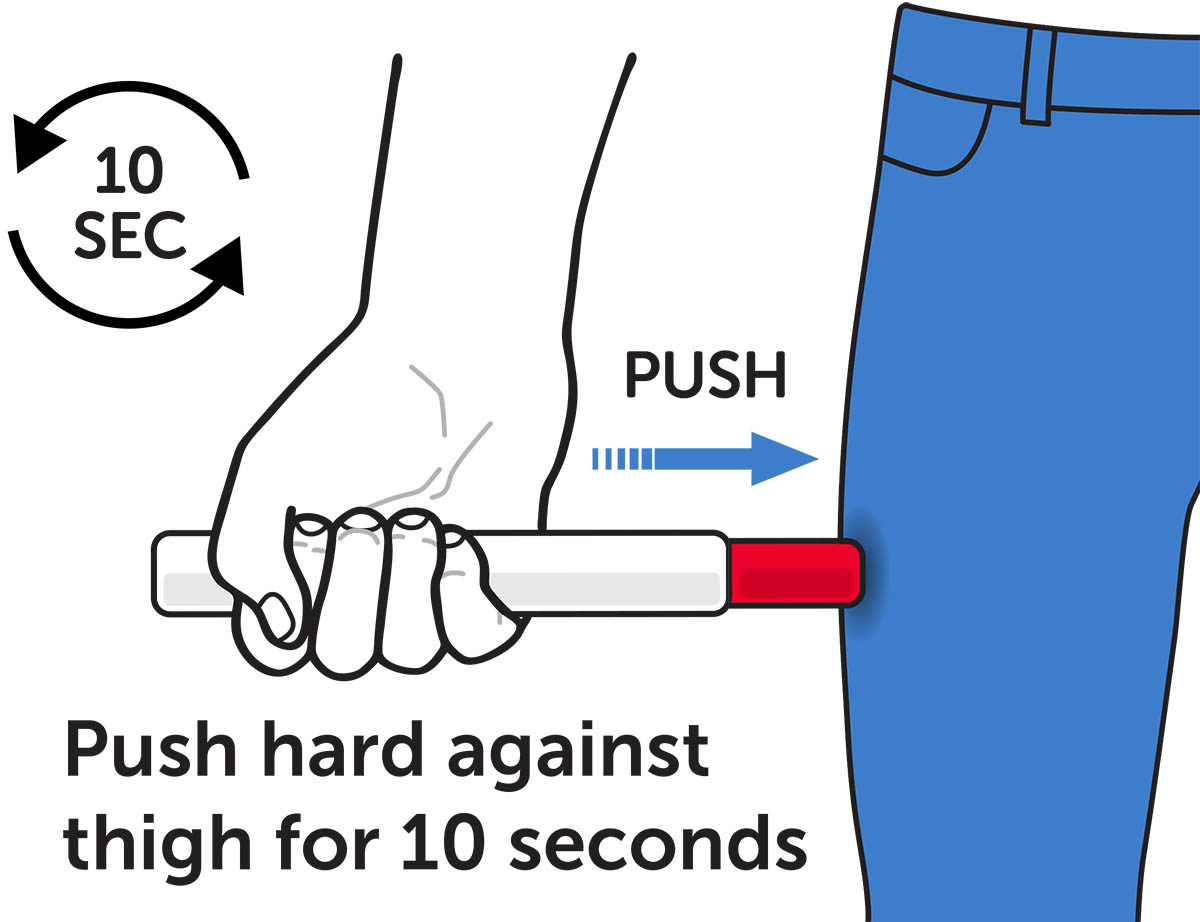
Figure H – Self Administration
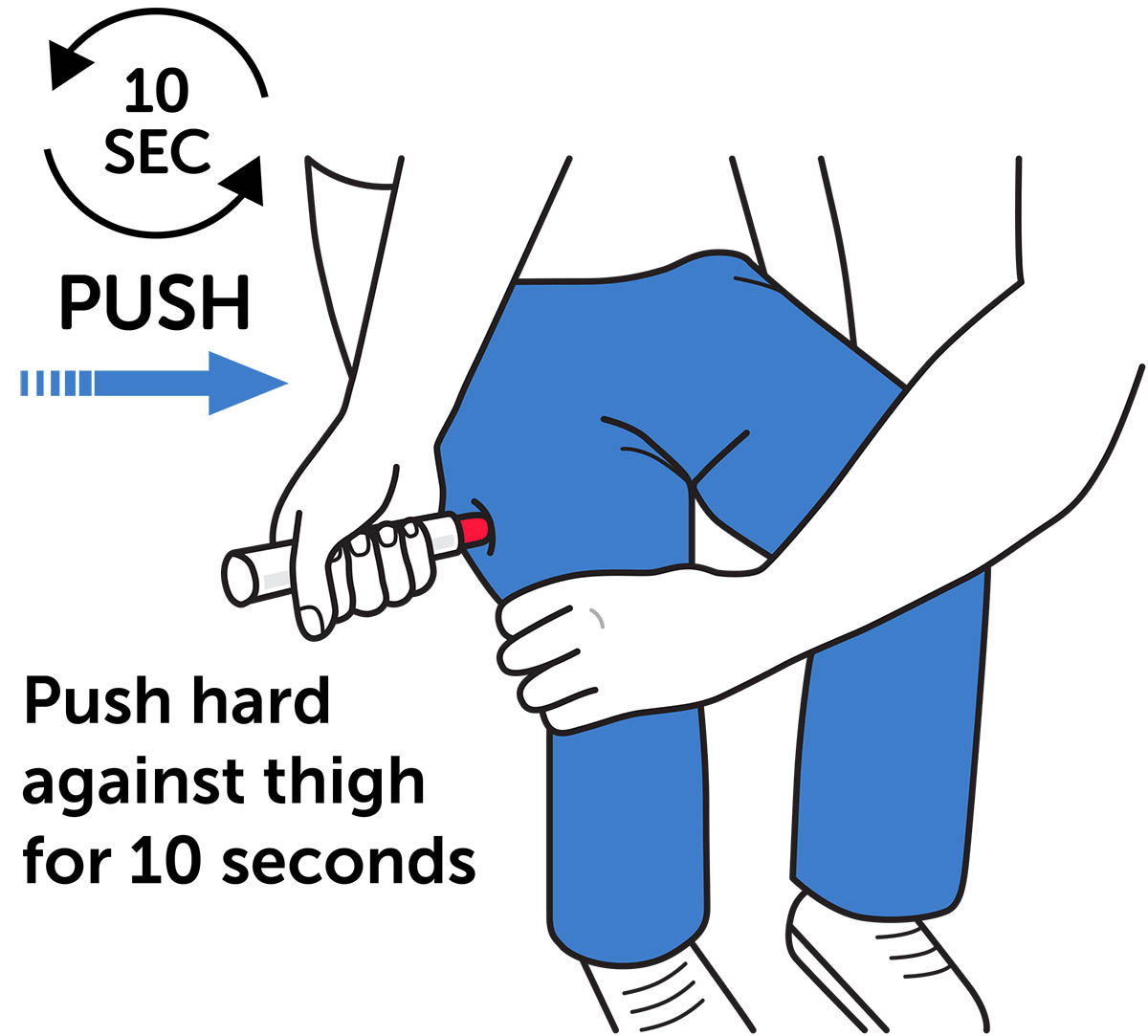
Figure I – Caregiver Administration
- Only inject into the middle of the outer thigh (upper leg). Do not inject into any other part of the body.
Step 4. Remove the Auto-Injector
Pull Epinephrine Injection straight out at a 90° angle (perpendicular) from the outer thigh (upper leg).
- Massage the injection area for 10 seconds.
- Check the red needle end. The injection is complete and the correct dose has been delivered if you see the needle sticking out of the red needle end.
- If you do not see the needle, use a new Epinephrine Injection and repeat Step 3.
- Do not reuse the same Epinephrine Injection.
Step 5. Get emergency medical help if needed.
Get emergency medical help for further treatment of the allergic emergency (anaphylaxis), if needed, after using Epinephrine Injection. Before you receive Epinephrine Injection, your healthcare provider should talk to you about when to get emergency medical help.
Step 6. After use
Warning: After Epinephrine Injection is activated, the needle does not retract back into the device.
After use, carefully place the used Epinephrine Injection back into the carrier tube to cover the needle.
- Place the labeled half of the carrier tube on a flat surface, with the opening facing upward.
- Using 1 hand, carefully slide the red needle end of Epinephrine Injection into the labeled half of the carrier tube until the needle is fully covered. See Figure J.
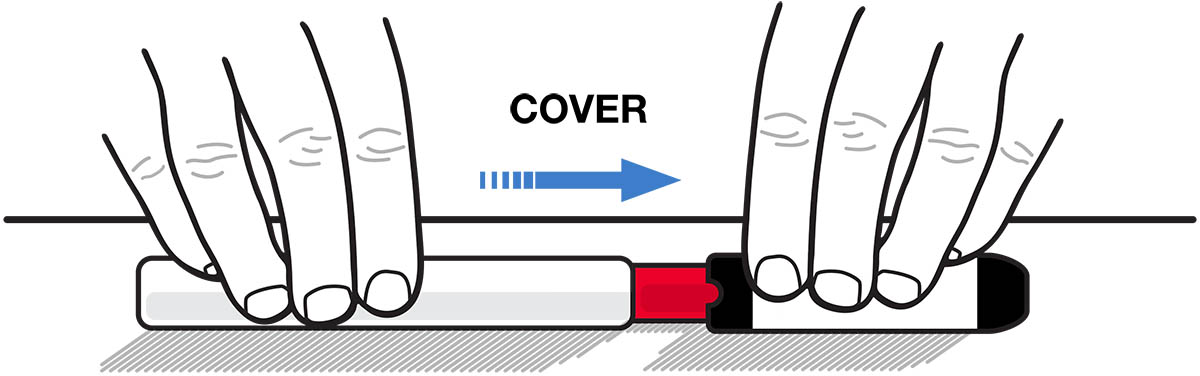
Figure J
- After the needle is inside the labeled half of the carrier tube, firmly push the unlabeled half of the carrier tube over the non-needle end of Epinephrine Injection until it is securely closed. See Figure K.
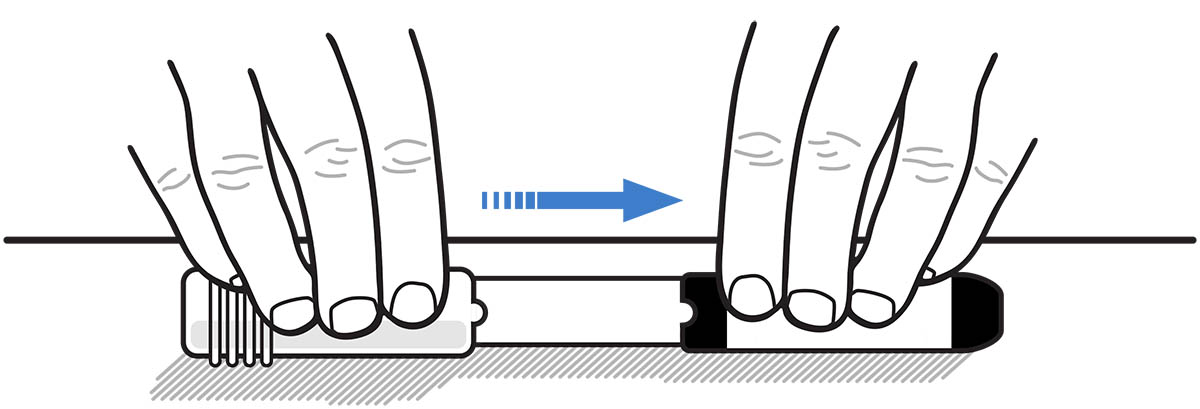
Figure K
- Take your used Epinephrine Injection with you when you go to see a healthcare provider.
- Tell the healthcare provider that you have received an injection of epinephrine. Show the healthcare provider where you received the injection.
- Give the used Epinephrine Injection to the healthcare provider for inspection and proper disposal.
- Ask your healthcare provider if you need a new prescription.
Storing Epinephrine Injection
- Store Epinephrine Injection at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep Epinephrine Injection in the carrier tube to protect it from light and damage. The carrier tube is not waterproof.
- Do not expose Epinephrine Injection to extreme heat or cold. For example, do not store in your vehicle’s glove box and do not store it in the refrigerator or freezer.
- Keep Epinephrine Injection and all medicines out of the reach of children .
Disposing of Epinephrine Injection
Important: The blue needle shield cap and blue end cap are small parts and may pose a choking hazard for children. Throw away the blue needle shield cap and blue end cap immediately after using Epinephrine Injection.
- Epinephrine Injection has an expiration date. Replace it before the expiration date.
- For instructions on how to throw away (dispose of) expired, unused, or used Epinephrine Injection, see the section titled “Disposing of an Expired, Unused or Used Epinephrine Injection” on the Patient Information side of this leaflet.
For more information and video instructions on the use of Epinephrine Injection, visit www.epinephrineautoinject.com or call 1-877-835-5472.
© 2025 Amneal Pharmaceuticals LLC. All rights reserved.
Manufactured for and Distributed by:
Amneal Pharmaceuticals LLC
Bridgewater, NJ 08807
1872-06
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised 03/2026
Mechanism of Action
Epinephrine acts on both alpha- and beta-adrenergic receptors.
Through its action on alpha-adrenergic receptors, epinephrine lessens the vasodilation and increased vascular permeability that occurs during anaphylaxis, which can lead to loss of intravascular fluid volume and hypotension.
Through its action on beta-adrenergic receptors, epinephrine causes bronchial smooth muscle relaxation and helps alleviate bronchospasm, wheezing, and dyspnea that may occur during anaphylaxis.
Epinephrine also alleviates pruritus, urticaria, and angioedema, and may relieve gastrointestinal and genitourinary symptoms associated with anaphylaxis because of its relaxer effects on the smooth muscle of the stomach, intestine, uterus and urinary bladder.