Get your patient on Ertaczo - Sertaconazole Nitrate cream (Sertaconazole Nitrate)
Ertaczo - Sertaconazole Nitrate cream prescribing information
INDICATIONS AND USAGE
ERTACZO ® cream, 2%, is indicated for the topical treatment of interdigital tinea pedis in immunocompetent adult and pediatric patients 12 years of age and older caused by Trichophyton rubrum , Trichophyton mentagrophytes, and Epidermophyton floccosum .
DOSAGE AND ADMINISTRATION
- Apply ERTACZO cream, 2% twice daily for 4 weeks. Apply a sufficient amount of ERTACZO cream, 2% to cover both the affected areas between the toes and the immediately surrounding healthy skin.
- Use ERTACZO cream, 2% for the full treatment time recommended by the physician, even though symptoms may have improved.
- Dry the affected area(s) thoroughly before application, if using ERTACZO cream, 2% after bathing.
- Wash hands after use.
- Avoid the use of occlusive dressings or wrappings.
- For topical use.
- Not for ophthalmic, oral, or intravaginal use.
DOSAGE FORMS AND STRENGTHS
Cream, 2%. Each gram of ERTACZO cream, 2%, contains 17.5 mg of sertaconazole (as sertaconazole nitrate, 20 mg) in a white cream base.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no available data on ERTACZO cream, 2% use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, there were no adverse developmental effects observed with oral administration of sertaconazole nitrate to pregnant rats and rabbits during organogenesis at doses 40 and 80 times, respectively, the maximum recommended human dose (MRHD) based on body surface area (BSA) comparison. In rats, when maternal dosing was continued until weaning, a reduction in live birth indices and an increase in the number of still-born pups was observed at doses 20 and 40 times the MRHD based on BSA comparison ( see Data ).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of major birth defects, loss and other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Animal embryofetal development studies have not been conducted with ERTACZO cream, 2%. Embryofetal development studies performed in pregnant rats and rabbits administered oral doses of sertaconzaole nitrate up to 160 mg/kg/day (40 times [rats] and 80 times [rabbits] the MRHD based on a BSA comparison) during the period of organogenesis revealed no malformations or embryofetal developmental toxicity. In a pre- and postnatal development study, pregnant rats were administered oral doses of sertaconazole nitrate from pregnancy day 6 to lactation day 20. A reduction in live birth indices and an increase in the number of still-born pups were seen at doses 20 and 40 times the MRHD based on BSA comparison.
Lactation
Risk Summary
There are no data available on the presence of sertaconazole in human or animal milk, its effects on the breastfed infant, or its effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ERTACZO cream, 2% and any potential adverse effects on the breastfed infant from ERTACZO cream, 2% or from the underlying maternal condition.
Pediatric Use
The safety and effectiveness of ERTACZO cream, 2%, have not been established in pediatric patients younger than 12 years of age.
Geriatric Use
Clinical trials of ERTACZO cream, 2%, did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
Local Adverse Reactions
If irritation develops, discontinue treatment and institute appropriate therapy.
Physicians should exercise caution when prescribing ERTACZO cream, 2%, to patients known to be sensitive to azole antifungals since cross-reactivity may occur.
ADVERSE REACTIONS
Most common adverse reactions observed in clinical trials (incidence >2%) were contact dermatitis, dry skin, burning skin, application site skin tenderness. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Lacer Pharma, LLC at 1-888-403-8874 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In clinical trials, cutaneous adverse events occurred in 7 of 297 (2%) subjects (2 of them severe) receiving ERTACZO cream, 2%, and in 7 of 291 (2%) subjects (2 of them severe) receiving vehicle. These reported cutaneous adverse events included contact dermatitis, dry skin, burning skin, and application site skin tenderness.
In a dermal sensitization trial, 8 of 202 evaluable subjects tested with ERTACZO cream, 2%, and 4 of 202 evaluable subjects tested with vehicle exhibited a erythematous reaction in the challenge phase. There was no evidence of cumulative irritation or contact sensitization in a repeated insult patch test involving 202 healthy volunteers.
Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ERTACZO cream, 2%. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cutaneous adverse events: erythema, pruritus, vesiculation, desquamation, and hyperpigmentation.
DESCRIPTION
ERTACZO (sertaconazole nitrate) cream, 2%, is for topical application. It contains the azole antifungal, sertaconazole nitrate. Sertaconazole nitrate contains one asymmetric carbon atom and exists as a racemic mixture of equal amounts of R and S enantiomers.
Sertaconazole nitrate is designated chemically as (±)-1-[2,4-dichloro-β-[(7-chlorobenzo-[ b ]thien-3-yl)methoxy]phenethyl]imidazole nitrate. It has a molecular weight of 500.8. The molecular formula is C 20 H 15 Cl 3 N 2 OS ● HNO 3 , and the structural formula is as follows:
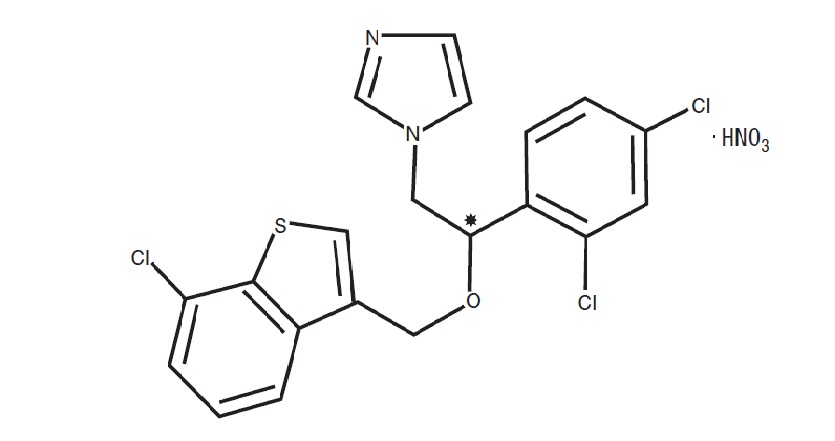
Sertaconazole nitrate is a white or almost white powder. It is practically insoluble in water, soluble in methanol, and sparingly soluble in alcohol and in methylene chloride. Each gram of ERTACZO cream, 2%, contains 17.5 mg of sertaconazole (as sertaconazole nitrate, 20 mg) in a white cream base of ethylene glycol, glyceryl isostearate, glycolized saturated glycerides, light mineral oil, methylparaben, polyethylene glycol palmitostearate, polyoxyethylened saturated glycerides, purified water, and sorbic acid.
CLINICAL PHARMACOLOGY
Mechanism of Action
Sertaconazole nitrate is an azole antifungal [see Clinical Pharmacology (12.4) ] .
Pharmacokinetics
In a multiple-dose pharmacokinetic trial that included 5 male subjects with interdigital tinea pedis (range of diseased area, 42 - 140 cm 2 ; mean, 93 cm 2 ), ERTACZO cream, 2%, was topically applied every 12 hours for a total of 13 doses to the diseased skin (0.5 g sertaconazole nitrate per 100 cm 2 ). Sertaconazole concentrations in plasma measured by serial blood sampling for 72 hours after the thirteenth dose were below the limit of quantitation (2.5 ng/mL) of the analytical method used.
Microbiology
Mechanism of Action
Sertaconazole, an azole antifungal agent, inhibits fungal cytochrome P-450-mediated 14 alpha-lanosterol demethylase enzyme. This enzyme functions to convert lanosterol to ergosterol. Ergosterol is a key component of fungal cell membranes and lack of this component leads to fungal cell injury by leakage of key constituents in the cytoplasm from the cell.
Activity In Vitro and in Clinical Infections
Sertaconazole nitrate has been shown to be active against isolates of the following microorganisms in clinical infections [see Indications and Usage (1) ] :
- Trichophyton rubrum
- Trichophyton mentagrophytes
- Epidermophyton floccosum
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year rat dermal carcinogenicity study, topical administration of sertaconazole nitrate cream did not increase the number of neoplastic lesions compared to control animals, at sertaconazole nitrate doses of up to 800 mg/kg/day (200 times the MRHD based on a BSA comparison).
No clastogenic potential was observed in a mouse micronucleus test. Sertaconazole nitrate was nonclastogenic in the in vivo mouse sister chromatid exchange assay. There was no evidence that sertaconazole nitrate induced unscheduled DNA synthesis in primary rat hepatocyte cultures.
Rats treated orally with up to 160 mg/kg/day of sertaconazole nitrate (40 times the MRHD based on a BSA comparison), exhibited no toxicity or adverse effects on reproductive performance or fertility in male or female rats.
CLINICAL STUDIES
In two randomized, double-blind clinical trials, subjects 12 years and older with interdigital tinea pedis applied either ERTACZO cream, 2%, or vehicle, twice daily for 4 weeks. Subjects with moccasin-type (plantar) tinea pedis and/or onychomycosis were excluded from the trial. Two weeks after completion of therapy (6 weeks after beginning therapy), subjects were evaluated for signs and symptoms related to interdigital tinea pedis.
Treatment outcomes are summarized in the table below.
| Treatment Outcomes as Percent (%) of Total Subjects with Interdigital Tinea Pedis | ||||
|---|---|---|---|---|
| Trial 1 | Trial 2 | |||
| Sertaconazole | Vehicle | Sertaconazole | Vehicle | |
Complete Cure• (Primary Efficacy Variable) | 13/99 (13.1%) | 3/92 (3.3%) | 28/103 (27.2%) | 5/103 (4.9%) |
Effective Treatment•• | 32/99 (32.3%) | 11/92 (12.0%) | 52/103 (50.5%) | 16/103 (15.5%) |
Mycological Cure••• | 49/99 (49.5%) | 18/92 (19.6%) | 71/103 (68.9%) | 20/103 (19.4%) |
• Complete Cure – Patients who had complete clearing of signs and symptoms and Mycological Cure. •• Effective Treatment – Patients who had minimal residual signs and symptoms of interdigital tinea pedis and Mycological Cure. ••• Mycological Cure – Patients who had both negative microscopic KOH preparation and negative fungal culture.
In clinical trials, complete cure in ERTACZO cream, 2%-treated subjects was achieved in 32 of 160 (20%) subjects with Trichophyton rubrum , in 7 of 28 (25%) subjects with Trichophyton mentagrophytes , and in 1 of 13 (15%) subjects with Epidermophyton floccosum .
HOW SUPPLIED/STORAGE AND HANDLING
ERTACZO cream, 2%, is white in color and supplied in tubes in the following size:
60-gram tube NDC 73159-004-60
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Mechanism of Action
Sertaconazole nitrate is an azole antifungal [see Clinical Pharmacology (12.4) ] .