Get your patient on Erythromycin Lactobionate - Erythromycin Lactobionate injection, Powder, Lyophilized, For Solution (Erythromycin Lactobionate)
Erythromycin Lactobionate - Erythromycin Lactobionate injection, Powder, Lyophilized, For Solution prescribing information
INDICATIONS AND USAGE
Erythromycin lactobionate for injection, USP is indicated in the treatment of infections caused by susceptible strains of the designated organisms in the diseases listed below when oral administration is not possible or when the severity of the infection requires immediate high serum levels of erythromycin. Intravenous therapy should be replaced by oral administration at the appropriate time.
Upper respiratory tract infections of mild to moderate degree caused by Streptococcus pyogenes (Group A beta-hemolytic streptococci); Streptococcus pneumoniae (Diplococcus pneumoniae); Haemophilus influenzae (when used concomitantly with adequate doses of sulfonamides, since many strains of H. influenzae are not susceptible to the erythromycin concentrations ordinarily achieved). (See appropriate sulfonamide labeling for prescribing information).
Lower respiratory tract infections of mild to moderate severity caused by Streptococcus pyogenes (Group A beta-hemolytic streptococci); Streptococcus pneumoniae (Diplococcus pneumoniae).
Respiratory tract infections due to Mycoplasma pneumoniae.
Skin and skin structure infections of mild to moderate severity caused by Streptococcus pyogenes and Staphylococcus aureus (resistant staphylococci may emerge during treatment).
Diphtheria: As an adjunct to antitoxin infections due to Corynebacterium diphtheriae to prevent establishment of carriers and to eradicate the organism in carriers.
Erythrasma: In the treatment of infections due to Corynebacterium minutissimum .
Acute pelvic inflammatory disease caused by Neisseria gonorrhoeae : Erythromycin lactobionate for injection, USP, followed by erythromycin stearate or erythromycin base orally, as an alternative drug in treatment of acute pelvic inflammatory disease caused by N. gonorrhoeae in female patients with a history of sensitivity to penicillin.
Before treatment of gonorrhea, patients who are suspected of also having syphilis should have a microscopic examination for T. pallidum (by immunofluorescence or darkfield) before receiving erythromycin and monthly serologic tests for a minimum of 4 months thereafter.
Legionnaires' Disease caused by Legionella pneumophila . Although no controlled clinical efficacy studies have been conducted, in vitro and limited preliminary clinical data suggest that erythromycin may be effective in treating Legionnaires' Disease.
Prevention of Initial Attacks of Rheumatic Fever
Penicillin is considered by the American Heart Association to be the drug of choice in the prevention of initial attacks of rheumatic fever (treatment of Group A betahemolytic streptococcal infections of the upper respiratory tract e.g., tonsillitis, or pharyngitis). 1 Erythromycin is indicated for the treatment of penicillin-allergic patients. The therapeutic dose should be administered for ten days.
Prevention of Recurrent Attacks of Rheumatic Fever
Penicillin or sulfonamides are considered by the American Heart Association to be the drugs of choice in the prevention of recurrent attacks of rheumatic fever. In patients who are allergic to penicillin and sulfonamides, oral erythromycin is recommended by the American Heart Association in the long-term prophylaxis of streptococcal pharyngitis (for the prevention of recurrent attacks of rheumatic fever). 1
Prevention of Bacterial Endocarditis
Although no controlled clinical efficacy trials have been conducted, oral erythromycin has been recommended by the American Heart Association for prevention of bacterial endocarditis in penicillin-allergic patients with prosthetic cardiac valves, most congenital cardiac malformations, surgically constructed systemic pulmonary shunts, rheumatic or other acquired valvular dysfunction, idiopathic hypertrophic subaortic stenosis (IHSS), previous history of bacterial endocarditis and mitral valve prolapse with insufficiency when they undergo dental procedures and surgical procedures of the upper respiratory tract. 2
To reduce the development of drug-resistant bacteria and maintain the effectiveness of erythromycin and other antibacterial drugs, erythromycin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
For the treatment of severe infections in adults and pediatric patients, the recommended intravenous dose of erythromycin lactobionate is 15 to 20 mg/kg/day. Higher doses, up to 4 g/day, may be given for severe infections.
Administration of doses of ≥4 g/day may increase the risk for the development of erythromycin-induced hearing loss in elderly patients, particularly those with reduced renal or hepatic function. Erythromycin Lactobionate for Injection, USP, must be administered by continuous or intermittent intravenous infusion only. Due to the irritative properties of erythromycin, IV push is an unacceptable route of administration.
Continuous infusion of erythromycin lactobionate is preferable due to the slower infusion rate and lower concentration of erythromycin; however, intermittent infusion at six hour intervals is also effective. Intravenous erythromycin should be replaced by oral erythromycin as soon as possible.
For slow continuous infusion: The final diluted solution of erythromycin lactobionate is prepared to give a concentration of 1 g per liter (1 mg/mL).
For intermittent infusion: Administer one-fourth the total daily dose of erythromycin lactobionate by intravenous infusion in 20 to 60 minutes at intervals not greater than every six hours. The final diluted solution of erythromycin lactobionate is prepared to give a concentration of 1 to 5 mg/mL. No less than 100 mL of IV diluent should be used. Infusion should be sufficiently slow to minimize pain along the vein.
For treatment of acute pelvic inflammatory disease caused by N. Gonorrhoeae , in female patients hypersensitive to penicillins, administer 500 mg erythromycin lactobionate every six hours for three days, followed by oral administration of 250 mg erythromycin stearate or base every six hours for seven days.
For treatment of Legionnaires' Disease: Although optimal doses have not been established, doses utilized in reported clinical data were 1 to 4 grams daily in divided doses.
Administration of doses of ≥ 4 g/day may increase the risk for the development of erythromycin-induced hearing loss in elderly patients, particularly those with reduced renal or hepatic function.
In the treatment of Group A beta-hemolytic streptococcal infections of the upper respiratory tract (e.g., tonsillitis or pharyngitis), the therapeutic dosage of erythromycin should be administered for ten days. The American Heart Association suggests a dosage of 250 mg of erythromycin orally, twice a day in long-term prophylaxis of streptococcal upper respiratory tract infections for the prevention of recurring attacks of rheumatic fever in patients allergic to penicillin and sulfonamides. 1
In prophylaxis against bacterial endocarditis (See INDICATIONS AND USAGE ) the oral regimen for penicillin allergic patients is erythromycin 1 gram, 1 hour before the procedure followed by 500 mg six hours later. 2 Preparation of Solution:
Erythromycin lactobionate for Injection, USP is a lyophilized powder, which requires reconstitution and further dilution before administration according to the steps below:
- Reconstitute Erythromycin lactobionate for Injection, USP by adding 10 mL of Sterile Water for Injection, USP to the 500 mg single-dose vial to give a concentration of 50 mg of erythromycin activity per milliliter (50 mg/mL). Do not use other diluents in this step because other diluents may cause precipitation during reconstitution. Do not use diluents containing preservatives or inorganic salts. The reconstituted solution is stable at refrigerator temperature for 2 weeks, or for 24 hours at room temperature.
- Add the reconstituted solution to one of the following diluents before administration to give a concentration of 1 g of erythromycin activity per liter (1 mg/mL) for continuous infusion or 1 to 5 mg/mL for intermittent infusion:
- Do not use other diluents in this step 2. Acidic solutions of Erythromycin lactobionate for Injection, USP (solutions with a pH < 5.5) are unstable and lose their potency rapidly.
Do not add any drug or chemical agent to an erythromycin lactobionate for injection solution unless its effect on the chemical and physical stability of the solution has first been determined.
Stability
The final diluted solution of erythromycin lactobionate should be completely administered within 8 hours, since it is not suitable for storage. Discard unused portion.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
Erythromycin is contraindicated in patients with known hypersensitivity to this antibiotic. Erythromycin is contraindicated in patients taking terfenadine or astemizole, cisapride, pimozide, ergotamine, or dihydroergotamine (See WARNINGS and PRECAUTIONS – Drug Interactions ).
Do not use erythromycin concomitantly with 3-hydroxy-3-methylglutaryl-coenzyme A (HMG CoA) reductase inhibitors (statins) that are extensively metabolized by cytochrome P450 isoform 3A4 (lovastatin or simvastatin), due to the increased risk of myopathy, including rhabdomyolysis (See WARNINGS and PRECAUTIONS – Drug Interactions ).
ADVERSE REACTIONS
Erythromycin has been associated with QT prolongation and ventricular arrhythmias, including ventricular tachycardia and torsades de pointes (See WARNINGS ).
Side effects following the use of intravenous erythromycin are rare. Occasional venous irritation has been encountered, but if the infusion is given slowly, in dilute solution, preferably by continuous intravenous infusion or intermittent infusion in no less than 20 to 60 minutes, pain and vessel trauma are minimized.
Allergic reactions ranging from urticaria to anaphylaxis have occurred. Skin reactions ranging from mild eruptions to erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis have been reported rarely.
There have been isolated reports of reversible hearing loss occurring chiefly in patients with renal insufficiency and in patients receiving high doses of erythromycin.
Elderly patients, particularly those with reduced renal or hepatic function, may also be at increased risk for developing this effect when erythromycin doses of 4 grams/day or higher are given (See DOSAGE AND ADMINISTRATION ).
To report SUSPECTED ADVERSE REACTIONS, contact safety.nexuspharma@lambda-cro.com or 1-855-642-2594 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Serious adverse reactions have been reported in patients taking erythromycin concomitantly with CYP3A4 substrates. These include colchicine toxicity with colchicine; rhabdomyolysis with simvastatin, lovastatin, and atorvastatin; and hypotension with calcium channel blockers metabolized by CYP3A4 (e.g. verapamil, amlodipine, diltiazem, vasospasm and ischemia with ergotamine/dihydroergotamine) (See PRECAUTIONS – Drug Interactions ).
DESCRIPTION
Erythromycin is produced by a strain of Streptomyces erythraeus and belongs to the macrolide group of antibiotics. It is basic and readily forms salts with acids.
Erythromycin lactobionate for injection, USP is a soluble salt of erythromycin suitable for intravenous administration. The lactobionic acid content is 244 mg per vial. Lactobionic acid and/or erythromycin are used to adjust the pH during the manufacture of the product. When reconstituted as directed, each mL contains 50 mg of erythromycin activity. The pH of the reconstituted solution is 6.5 -7.5.
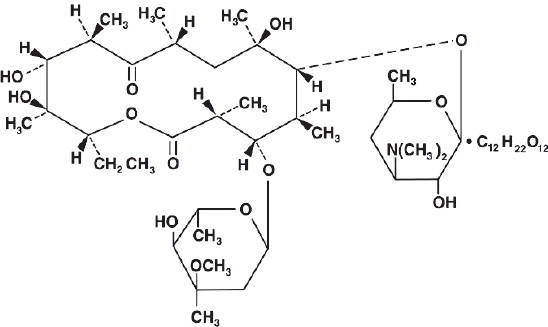
Erythromycin lactobionate is chemically known as erythromycin mono (4-0-β-Dgalactopyranosyl-D-gluconate) (salt). The structural formula is:
CLINICAL PHARMACOLOGY
Erythromycin diffuses readily into most body fluids. In the absence of meningeal inflammation, low concentrations are normally achieved in the spinal fluid but the passage of the drug across the blood-brain barrier increases in meningitis. Erythromycin crosses the placental barrier and is excreted in breast milk. Erythromycin is not removed by peritoneal dialysis or hemodialysis.
In the presence of normal hepatic function, erythromycin is concentrated in the liver and is excreted in the bile; the effect of hepatic dysfunction on biliary excretion of erythromycin is not known. From 12 to 15 percent of intravenously administered erythromycin is excreted in active form in the urine.
Intravenous infusion of 500 mg of erythromycin lactobionate at a constant rate over 1 hour in fasting adults produced a mean serum erythromycin level of approximately 7 mcg/mL at 20 minutes, 10 mcg/mL at 1 hour, 2.6 mcg/mL at 2.5 hours, and 1 mcg/mL at 6 hours.
Microbiology
Mechanism of Action
Erythromycin acts by inhibition of protein synthesis by binding 50 S ribosomal subunits of susceptible organisms. It does not affect nucleic acid synthesis.
Resistance
Resistance to erythromycin in S. aureus may emerge during therapy. Many isolates of Haemophilus influenzae are resistant to erythromycin but are susceptible to erythromycin and sulfonamides when used concomitantly.
Interactions With Other Antimicrobials
Antagonism has been demonstrated in vitro between erythromycin and clindamycin, lincomycin and chloramphenicol.
Antimicrobial Activity
Erythromycin has been shown to be active against most isolates of the following microorganisms, both in vitro and in clinical infections (see INDICATIONS AND USAGE ):
Aerobic bacteria
Gram-positive bacteria
Corynebacterium diphtheriae
Corynebacterium minutissimum
Staphylococcus aureus
Streptococcus pneumoniae
Streptococcus pyogenes
Gram-negative bacteria
Legionella pneumophila
Neisseria gonorrhoeae
Other Microorganisms
Mycoplasma pneumoniae
The following in vitro data are available, but their clinical significance is unknown. At least 90 percent of the following bacteria exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for erythromycin against isolates of similar genus or organism group. However, the efficacy of erythromycin in treating clinical infections caused by these bacteria has not been established in adequate and well-controlled clinical trials.
Gram-negative bacteria
Moraxella catarrhalis
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
HOW SUPPLIED
Erythromycin lactobionate for injection, USP, is supplied as a sterile, lyophilized powder in packages of five vials, each vial containing the equivalent of 500 mg of erythromycin.
| Item | NDC Number |
| 5 Vials per Carton | 14789-116-05 |
| Single-dose vial | 14789-116-07 |
Store at 20 to 25ºC (68 to 77ºF). [See USP Controlled Room Temperature].
Mechanism of Action
Erythromycin acts by inhibition of protein synthesis by binding 50 S ribosomal subunits of susceptible organisms. It does not affect nucleic acid synthesis.