Get your patient on Etodolac
Etodolac prescribing information
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions ].
- Etodolac tablets 400 mg and 500 mg are contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications and Warnings ].
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal (GI) events. (See WARNINGS ).
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of etodolac tablets, and other treatment options before deciding to use etodolac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
Etodolac tablets are indicated:
• For acute and long-term use in the management of signs and symptoms of the following:
1. Osteoarthritis
2. Rheumatoid arthritis
• For the management of acute pain
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of etodolac tablets and other treatment options before deciding to use etodolac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
After observing the response to initial therapy with etodolac tablets, the dose and frequency should be adjusted to suit an individual patient's needs.
Dosage adjustment of etodolac tablets is generally not required in patients with mild to moderate renal impairment. Etodolac should be used with caution in such patients, because, as with other NSAIDs, it may further decrease renal function in some patients with impaired renal function (see WARNINGS, Renal Effects ).
Analgesia
The recommended total daily dose of etodolac for acute pain is up to 1000 mg, given as 200-400 mg every 6 to 8 hours. Doses of etodolac greater than 1000 mg/day have not been adequately evaluated in well-controlled trials.
Osteoarthritis and Rheumatoid Arthritis
The recommended starting dose of etodolac for the management of the signs and symptoms of osteoarthritis or rheumatoid arthritis is: 300 mg b.i.d., t.i.d., or 400 mg b.i.d., or 500 mg b.i.d. A lower dose of 600 mg/day may suffice for long-term administration. Physicians should be aware that doses above 1000 mg/day have not been adequately evaluated in well-controlled clinical trials.
In chronic conditions, a therapeutic response to therapy with etodolac is sometimes seen within one week of therapy, but most often is observed by two weeks. After a satisfactory response has been achieved, the patient's dose should be reviewed and adjusted as required.
CLINICAL TRIALS
Analgesia
Controlled clinical trials in analgesia were single-dose, randomized, double-blind, parallel studies in three pain models, including dental extractions. The analgesic effective dose for etodolac established in these acute pain models was 200 to 400 mg. The onset of analgesia occurred approximately 30 minutes after oral administration. Etodolac 200 mg provided efficacy comparable to that obtained with aspirin (650 mg). Etodolac 400 mg provided efficacy comparable to that obtained with acetaminophen with codeine (600 mg + 60 mg). The peak analgesic effect was between 1 to 2 hours. Duration of relief averaged 4 to 5 hours for 200 mg of etodolac and 5 to 6 hours for 400 mg of etodolac as measured by when approximately half of the patients required remedication.
Osteoarthritis
The use of etodolac in managing the signs and symptoms of osteoarthritis of the hip or knee was assessed in double-blind, randomized, controlled clinical trials in 341 patients. In patients with osteoarthritis of the knee, etodolac, in doses of 600 to 1000 mg/day, was better than placebo in two studies. The clinical trials in osteoarthritis used b.i.d. dosage regimens.
Rheumatoid Arthritis
In a 3-month study with 426 patients, etodolac 300 mg b.i.d. was effective in management of rheumatoid arthritis and comparable in efficacy to piroxicam 20 mg/day. In a long-term study with 1,446 patients in which 60% of patients completed 6 months of therapy and 20% completed 3 years of therapy, etodolac in a dose of 500 mg b.i.d. provided efficacy comparable to that obtained with ibuprofen 600 mg q.i.d. In clinical trials of rheumatoid arthritis patients, etodolac has been used in combination with gold, d-penicillamine, chloroquine, corticosteroids, and methotrexate.
CONTRAINDICATIONS
Etodolac tablets are contraindicated in patients with known hypersensitivity to etodolac or other ingredients in etodolac.
Etodolac tablets should not be given to patients who have experienced asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS, Anaphylactoid Reactions and PRECAUTIONS, Pre-existing Asthma ).
- In the setting of coronary artery bypass graft (CABG) surgery [see Warnings ]
ADVERSE REACTIONS
In patients taking etodolac tablets or other NSAIDs, the most frequently reported adverse experiences occurring in approximately 1-10% of patients are:
Gastrointestinal experiences including: abdominal pain, constipation, diarrhea, dyspepsia, flatulence, gross bleeding/perforation, heartburn, nausea, GI ulcers (gastric/duodenal), vomiting.
Other events including: abnormal renal function, anemia, dizziness, edema, elevated liver enzymes, headaches, increased bleeding time, pruritis, rashes, tinnitus.
Adverse-reaction information for etodolac was derived from 2,629 arthritic patients treated with etodolac tablets in double-blind and open-label clinical trials of 4 to 320 weeks in duration and worldwide postmarketing surveillance studies. In clinical trials, most adverse reactions were mild and transient. The discontinuation rate in controlled clinical trials, because of adverse events, was up to 10% for patients treated with etodolac.
New patient complaints (with an incidence greater than or equal to 1%) are listed below by body system. The incidences were determined from clinical trials involving 465 patients with osteoarthritis treated with 300 to 500 mg of etodolac b.i.d. (i.e., 600 to 1000 mg/day).
Incidence Greater Than Or Equal To 1% - Probably Causally Related
Body as a whole - Chills and fever.
Digestive system - Dyspepsia (10%), abdominal pain Drug-related patient complaints occurring in 3 to 9% of patients treated with etodolac ., diarrhea, flatulence, nausea, abdominal distension, epigastric pain,abnormal stools, constipation, gastritis, melena, vomiting.
Nervous system - Asthenia/malaise, dizziness, depression, nervousness, fatigue.
Skin and appendages - Pruritus, rash.
Special senses - Blurred vision, tinnitus.
Urogenital system - Dysuria, urinary frequency.
Musculoskeletal - Arthralgia.
Drug-related patient complaints occurring in fewer than 3%, but more than 1%, are unmarked.
Drug Interactions
ACE-inhibitors
Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors (see WARNINGS ).
Antacids
The concomitant administration of antacids has no apparent effect on the extent of absorption of etodolac tablets. However, antacids can decrease the peak concentration reached by 15% to 20% but have no detectable effect on the time-to-peak.
Aspirin
When etodolac is administered with aspirin, its protein binding is reduced, although the clearance of free etodolac is not altered. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of etodolac and aspirin is not generally recommended because of the potential of increased adverse effects.
Cyclosporine, Digoxin, Methotrexate
Etodolac, like other NSAIDs, through effects on renal prostaglandins, may cause changes in the elimination of these drugs leading to elevated serum levels of cyclosporine, digoxin, methotrexate, and increased toxicity. Nephrotoxicity associated with cyclosporine may also be enhanced. Patients receiving these drugs who are given etodolac, or any other NSAID, and particularly those patients with altered renal function, should be observed for the development of the specific toxicities of these drugs. NSAIDs, such as etodolac, should not be administered prior to or concomitantly with high doses of methotrexate. NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. In general, caution should be used when NSAIDs are administered concomitantly with methotrexate.
Diuretics
Etodolac has no apparent pharmacokinetic interaction when administered with furosemide or hydrochlorothiazide. Nevertheless, clinical studies, as well as postmarketing observations have shown that etodolac can reduce the natriuretic effect of furosemide and thiazides in some patients with possible loss of blood pressure control. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal insufficiency or failure (see WARNINGS, Renal Effects ), as well as to assure diuretic efficacy.
Glyburide
Etodolac has no apparent pharmacokinetic interaction when administered with glyburide.
Lithium
NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity. Careful monitoring of lithium levels is advised in the event NSAID dosage adjustments are required.
Phenylbutazone
Phenylbutazone causes increase (by about 80%) in the free fraction of etodolac. Although in vivo studies have not been done to see if etodolac clearance is changed by coadministration of phenylbutazone, it is not recommended that they be coadministered.
Phenytoin
Etodolac has no apparent pharmacokinetic interaction when administered with phenytoin.
Warfarin
The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than that of users of either drug alone. Short-term pharmacokinetic studies have demonstrated that concomitant administration of warfarin and etodolac tablets results in reduced protein binding of warfarin, but there was no change in the clearance of free warfarin. There was no significant difference in the pharmacodynamic effect of warfarin administered alone and warfarin administered with etodolac tablets as measured by prothrombin time. Thus, concomitant therapy with warfarin and etodolac should not require dosage adjustment of either drug. However, caution should be exercised because there have been a few spontaneous reports of prolonged prothrombin times, with or without bleeding, in etodolac-treated patients receiving concomitant warfarin therapy. Close monitoring of such patients is therefore recommended.
DESCRIPTION
Etodolac or etodolac tablets, USP is member of the pyranocarboxylic acid group of nonsteroidal anti-inflammatory drugs (NSAIDs). Each tablet contains etodolac for oral administration. Etodolac is a racemic mixture of [+]S and [-]R-enantiomers. Etodolac is a white or almost white crystalline powder, practically insoluble in water, freely soluble in acetone and ethanol [96%].
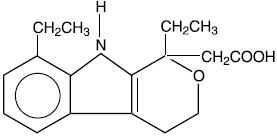
The chemical name is (±) 1,8-diethyl-1,3,4,9-tetrahydropyrano-[3,4-b]indole-1-acetic acid. The molecular weight of the base is 287.36. It has a pKa of 4.65 and an n-octanol: water partition coefficient of 11.4 at pH 7.4. The molecular formula for etodolac is C 17 H 21 NO 3 , and it has the following structural formula:
Each film coated tablet, for oral administration, contains 400 mg or 500 mg of etodolac, USP. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, crospovidone, hypromellose, iron oxide yellow, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone, sodium lauryl sulphate, talc, and titanium dioxide. The 400 mg tablets also contain iron oxide red.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Etodolac is a nonsteroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic, and antipyretic activities in animal models. The mechanism of action of etodolac, like that of other NSAIDs, is not completely understood, but may be related to the prostaglandin synthetase inhibition.
Etodolac is a racemic mixture of [-]R- and [+]S-etodolac. As with other NSAIDs, it has been demonstrated in animals that the [+]S-form is biologically active. Both enantiomers are stable and there is no [-]R to [+]S conversion in vivo.
Pharmacokinetics
Absorption
The systemic bioavailability of etodolac from etodolac tablets is 100% as compared to solution and at least 80% as determined from mass balance studies. Etodolac is well absorbed and had a relative bioavailability of 100% when 200 mg capsules were compared with a solution of etodolac. Based on mass balance studies, the systemic availability of etodolac from either the tablet or capsule formulation is at least 80%. Etodolac does not undergo significant first-pass metabolism following oral administration. Mean (± 1 SD) peak plasma concentrations (C max ) range from approximately 14 ± 4 to 37 ± 9 mcg/mL after 200 to 600 mg single doses and are reached in 80 ± 30 minutes (see Table 1 for summary of pharmacokinetic parameters). The dose-proportionality based on the area under the plasma concentration-time curve (AUC) is linear following doses up to 600 mg every 12 hours. Peak concentrations are dose proportional for both total and free etodolac following doses up to 400 mg every 12 hours, but following a 600 mg dose, the peak is about 20% higher than predicted on the basis of lower doses. The extent of absorption of etodolac is not affected when etodolac tablets are administered after a meal. Food intake, however, reduces the peak concentration reached by approximately one-half and increases the time to peak concentration by 1.4 to 3.8 hours.
NA = not available | ||||||||
• % Coefficient of variation | ||||||||
† Age Range (years) | ||||||||
| PK Parameters | Normal Healthy Adults ( 18 - 65 ) † ( n = 179 ) | Healthy Males ( 18 - 65 ) ( n = 176 ) | Healthy Females ( 27 - 65 ) ( n = 3 ) | Elderly (> 65 ) ( 70 - 84 ) | Hemodialysis ( 24 - 65 ) ( n = 9 ) | Renal Impairment ( 46 - 73 ) ( n = 10 ) | Hepatic Impairment ( 34 - 60 ) ( n = 9 ) | |
| Dialysis On | Dialysis Off | |||||||
| Tmax, h | 1.4 (61%)• | 1.4 (60%) | 1.7 (60%) | 1.2 (43%) | 1.7 (88%) | 0.9 (67%) | 2.1 (46%) | 1.1 (15%) |
| Oral Clearance, mL/ h/kg (CL/F) | 49.1 (33%) | 49.4 (33%) | 35.7 (28%) | 45.7 (27%) | NA | NA | 58.3 (19%) | 42.0 (43%) |
| Apparent Volume of Distribution, mL/kg (Vd/F) | 393 (29%) | 394 (29%) | 300 (8%) | 414 (38%) | NA | NA | NA | NA |
| Terminal Half- Life, h | 6.4 (22%) | 6.4 (22%) | 7.9 (35%) | 6.5 (24%) | 5.1 (22%) | 7.5 (34%) | NA | 5.7 (24%) |
Distribution
The mean apparent volume of distribution (Vd/F) of etodolac is approximately 390 mL/kg. Etodolac is more than 99% bound to plasma proteins, primarily to albumin. The free fraction is less than 1% and is independent of etodolac total concentration over the dose range studied. It is not known whether etodolac is excreted in human milk; however, based on its physical-chemical properties, excretion into breast milk is expected. Data from in vitro studies, using peak serum concentrations at reported therapeutic doses in humans, show that the etodolac free fraction is not significantly altered by acetaminophen, ibuprofen, indomethacin, naproxen, piroxicam, chlorpropamide, glipizide, glyburide, phenytoin, and probenecid.
Metabolism
Etodolac is extensively metabolized in the liver. The role, if any, of a specific cytochrome P450 system in the metabolism of etodolac is unknown. Several etodolac metabolites have been identified in human plasma and urine. Other metabolites remain to be identified. The metabolites include 6-, 7-, and 8- hydroxylated-etodolac and etodolac glucuronide. After a single dose of 14 C-etodolac, hydroxylated metabolites accounted for less than 10% of total drug in serum. On chronic dosing, hydroxylated-etodolac metabolite does not accumulate in the plasma of patients with normal renal function. The extent of accumulation of hydroxylated-etodolac metabolites in patients with renal dysfunction has not been studied. The hydroxylated-etodolac metabolites undergo further glucuronidation followed by renal excretion and partial elimination in the feces.
Excretion
The mean oral clearance of etodolac following oral dosing is 49 (± 16) mL/h/kg. Approximately 1% of an etodolac dose is excreted unchanged in the urine with 72% of the dose excreted into urine as parent drug plus metabolite:
| -etodolac, unchanged -etodolac glucuronide -hydroxylated metabolites (6-, 7-, and 8-OH) -hydroxylated metabolite glucuronides -unidentified metabolites | 1% 13% 5% 20% 33% |
Although renal elimination is a significant pathway of excretion for etodolac metabolites, no dosing adjustment in patients with mild to moderate renal dysfunction is generally necessary. The terminal half-life (t 1/2 ) of etodolac is 6.4 hours (22% CV). In patients with severe renal dysfunction or undergoing hemodialysis, dosing adjustment is not generally necessary.
Fecal excretion accounted for 16% of the dose.
Special Populations
Geriatric
In etodolac clinical studies, no overall differences in safety or effectiveness were observed between these patients and younger patients. In pharmacokinetic studies, age was shown not to have any effect on etodolac half-life or protein binding, and there was no change in expected drug accumulation. Therefore no dosage adjustment is generally necessary in the elderly on the basis of pharmacokinetics (see PRECAUTIONS, Geriatric Use ).
Etodolac is eliminated primarily by the kidney. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see WARNINGS, Renal Effects ).
Pediatric
Safety and effectiveness in pediatric patients below the age of 18 years have not been established.
Race
Pharmacokinetic differences due to race have not been identified. Clinical studies included patients of many races, all of whom responded in a similar fashion.
Hepatic Insufficiency
Etodolac is predominantly metabolized by the liver. In patients with compensated hepatic cirrhosis, the disposition of total and free etodolac is not altered. Patients with acute and chronic hepatic diseases do not generally require reduced doses of etodolac compared to patients with normal hepatic function. However, etodolac clearance is dependent on liver function and could be reduced in patients with severe hepatic failure. Etodolac plasma protein binding did not change in patients with compensated hepatic cirrhosis given etodolac.
Renal Insufficiency
Etodolac pharmacokinetics have been investigated in subjects with renal insufficiency. Etodolac renal clearance was unchanged in the presence of mild-to-moderate renal failure (creatinine clearance 37 to 88 mL/min). Furthermore, there were no significant differences in the disposition of total and free etodolac in these patients. However, etodolac should be used with caution in such patients because, as with other NSAIDs, it may further decrease renal function in some patients. In patients undergoing hemodialysis, there was a 50% greater apparent clearance of total etodolac, due to a 50% greater unbound fraction. Free etodolac clearance was not altered, indicating the importance of protein binding in etodolac's disposition. Etodolac is not significantly removed from the blood in patients undergoing hemodialysis.
HOW SUPPLIED
Etodolac tablets, USP are available as:
Etodolac tablets, USP 400 mg are brown colored oval shaped film coated tablets debossed with '49' on one side and 'I' on other side
NDC 83980-029-13 Bottle of 30's
NDC 83980-029-01 Bottle of 100's
NDC 83980-029-10 Bottle of 1,000's
Etodolac tablets, USP 500 mg are yellow colored oval shaped film coated tablets debossed with '48' on one side and 'I' on other side
NDC 83980-030-13 Bottle of 30's
NDC 83980-030-01 Bottle of 100's
NDC 83980-030-05 Bottle of 500's
Store at 20ºC to 25ºC (68ºF to 77ºF) [See USP Controlled Room Temperature].Store tablets inoriginal container until ready to use. Dispense in light-resistant container.
You may report side eff ects to Ipca at 1-888-472-2651 or FDA at 1-800-FDA-1088.
Additional medication guides can be obtained by calling Ipca at 1-888-472-2651.
Distributed by:
Unichem Pharmaceuticals (USA), Inc.
East Brunswick,
NJ 08816, USA
January 2025
IEPON1