Get your patient on Etoposide - Etoposide injection, Solution, Concentrate (Etoposide)
Etoposide - Etoposide injection, Solution, Concentrate prescribing information
WARNINGS
Etoposide injection should be administered under the supervision of a qualified physician experienced in the use of cancer chemotherapeutic agents. Severe myelosuppression with resulting infection or bleeding may occur.
INDICATIONS AND USAGE
Etoposide Injection is indicated in the management of the following neoplasms:
Refractory Testicular Tumors
Etoposide Injection in combination therapy with other approved chemotherapeutic agents in patients with refractory testicular tumors who have already received appropriate surgical, chemotherapeutic, and radiotherapeutic therapy.
Small Cell Lung Cancer
Etoposide Injection and/or capsules in combination with other approved chemotherapeutic agents as first line treatment in patients with small cell lung cancer.
DOSAGE AND ADMINISTRATION
Note: Plastic devices made of acrylic or ABS (a polymer composed of acrylonitrile, butadiene, and styrene) have been reported to crack and leak when used with undiluted etoposide injection.
Etoposide Injection
The usual dose of etoposide injection in testicular cancer in combination with other approved chemotherapeutic agents ranges from 50 to 100 mg/m 2 /day on days 1 through 5 to 100 mg/m 2 /day on days 1, 3, and 5.
In small cell lung cancer, the etoposide injection dose in combination with other approved chemotherapeutic drugs ranges from 35 mg/m 2 /day for 4 days to 50 mg/m 2 /day for 5 days.
For recommended dosing adjustments in patients with renal impairment see PRECAUTIONS section.
Chemotherapy courses are repeated at 3- to 4-week intervals after adequate recovery from any toxicity.
The dosage should be modified to take into account the myelosuppressive effects of other drugs in the combination or the effects of prior x-ray therapy or chemotherapy which may have compromised bone marrow reserve.
Administration Precautions
As with other potentially toxic compounds, caution should be exercised in handling and preparing the solution of etoposide injection. Skin reactions associated with accidental exposure to etoposide injection may occur. The use of gloves is recommended. If etoposide injection solution contacts the skin or mucosa, immediately and thoroughly wash the skin with soap and water and flush the mucosa with water.
Preparation for Intravenous Administration
Etoposide injection must be diluted prior to use with either 5% Dextrose Injection, USP, or 0.9% Sodium Chloride Injection, USP, to give a final concentration of 0.2 to 0.4 mg/mL. If solutions are prepared at concentrations above 0.4 mg/mL, precipitation may occur. Hypotension following rapid intravenous administration has been reported, hence, it is recommended that the etoposide injection solution be administered over a 30- to 60-minute period. A longer duration of administration may be used if the volume of fluid to be infused is a concern. Etoposide injection should not be given by rapid intravenous injection.
Parenteral drug products should be inspected visually for particulate matter and discoloration (see DESCRIPTION section) prior to administration whenever solution and container permit.
Stability
Unopened vials of etoposide injection are stable until the date indicated on the package at room temperature (25°C). Vials diluted as recommended to a concentration of 0.2 to 0.4 mg/mL are stable for 96 and 24 hours, respectively, at room temperature (25°C) under normal room fluorescent light in both glass and plastic containers.
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published. 1-8 There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate.
CONTRAINDICATIONS
Etoposide injection is contraindicated in patients who have demonstrated a previous hypersensitivity to etoposide or any component of the formulation.
ADVERSE REACTIONS
The following data on adverse reactions are based on both oral and intravenous administration of etoposide injection as a single agent, using several different dose schedules for treatment of a wide variety of malignancies.
Hematologic Toxicity
Myelosuppression is dose related and dose limiting, with granulocyte nadirs occurring 7 to 14 days after drug administration and platelet nadirs occurring 9 to 16 days after drug administration. Bone marrow recovery is usually complete by day 20, and no cumulative toxicity has been reported. Fever and infection have also been reported in patients with neutropenia. Death associated with myelosuppression has been reported.
The occurrence of acute leukemia with or without a preleukemic phase has been reported rarely in patients treated with etoposide injection in association with other antineoplastic agents (see WARNINGS section).
Gastrointestinal Toxicity
Nausea and vomiting are the major gastrointestinal toxicities. The severity of such nausea and vomiting is generally mild to moderate with treatment discontinuation required in 1% of patients. Nausea and vomiting can usually be controlled with standard antiemetic therapy. Mild to severe mucositis/esophagitis may occur. Gastrointestinal toxicities are slightly more frequent after oral administration than after intravenous infusion.
Hypotension
Transient hypotension following rapid intravenous administration has been reported in 1% to 2% of patients. It has not been associated with cardiac toxicity or electrocardiographic changes. No delayed hypotension has been noted. To prevent this rare occurrence, it is recommended that etoposide injection be administered by slow intravenous infusion over a 30- to 60-minute period. If hypotension occurs, it usually responds to cessation of the infusion and administration of fluids or other supportive therapy as appropriate. When restarting the infusion, a slower administration rate should be used.
Allergic Reactions
Anaphylactic-like reactions characterized by chills, fever, tachycardia, bronchospasm, dyspnea, and/or hypotension have been reported to occur in 0.7% to 2% of patients receiving intravenous etoposide injection and in less than 1% of the patients treated with the oral capsules. These reactions have usually responded promptly to the cessation of the infusion and administration of pressor agents, corticosteroids, antihistamines, or volume expanders as appropriate; however, the reactions can be fatal. Hypertension and/or flushing have also been reported. Blood pressure usually normalizes within a few hours after cessation of the infusion. Anaphylactic-like reactions have occurred during the initial infusion of etoposide injection.
Facial/tongue swelling, coughing, diaphoresis, cyanosis, tightness in throat, laryngospasm, back pain, and/or loss of consciousness have sometimes occurred in association with the above reactions. In addition, an apparent hypersensitivity-associated apnea has been reported rarely.
Rash, urticaria, and/or pruritus have infrequently been reported at recommended doses. At investigational doses, a generalized pruritic erythematous maculopapular rash, consistent with perivasculitis, has been reported.
Alopecia
Reversible alopecia, sometimes progressing to total baldness, was observed in up to 66% of patients.
Other Toxicities
The following adverse reactions have been infrequently reported: abdominal pain, aftertaste, constipation, dysphagia, asthenia, fatigue, malaise, somnolence, transient cortical blindness, optic neuritis, interstitial pneumonitis/pulmonary fibrosis, fever, seizure (occasionally associated with allergic reactions), Stevens-Johnson syndrome, and toxic epidermal necrolysis, pigmentation, and a single report of radiation recall dermatitis.
Hepatic toxicity, generally in patients receiving higher doses of the drug than those recommended, has been reported with etoposide injection. Metabolic acidosis has also been reported in patients receiving higher doses.
Reports of extravasation with swelling have been received postmarketing. Rarely extravasation has been associated with necrosis and venous induration.
The incidences of adverse reactions in the table that follows are derived from multiple data bases from studies in 2,081 patients when etoposide injection was used either orally or by injection as a single agent.
ADVERSE DRUG EFFECT | PERCENT RANGE OF REPORTED INCIDENCE |
Hematologic toxicity | |
Leukopenia (less than 1,000 WBC/mm 3 ) | 3 to 17 |
Leukopenia (less than 4,000 WBC/mm 3 ) | 60 to 91 |
Thrombocytopenia (less than 50,000 platelets/mm 3 ) | 1 to 20 |
Thrombocytopenia (less than 100,000 platelets/mm 3 ) | 22 to 41 |
Anemia | 0 to 33 |
Gastrointestinal toxicity | |
Nausea and vomiting | 31 to 43 |
Abdominal pain | 0 to 2 |
Anorexia | 10 to 13 |
Diarrhea | 1 to 13 |
Stomatitis | 1 to 6 |
Hepatic | 0 to 3 |
Alopecia | 8 to 66 |
Peripheral neurotoxicity | 1 to 2 |
Hypotension | 1 to 2 |
Allergic reaction | 1 to 2 |
To report SUSPECTED ADVERSE REACTIONS, contact Meitheal Pharmaceuticals, Inc. at 1-844-824-8426 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
High-dose cyclosporin A resulting in concentrations above 2000 ng/mL administered with oral etoposide has led to an 80% increase in etoposide exposure with a 38% decrease in total body clearance of etoposide compared to etoposide alone.
DESCRIPTION
Etoposide Injection, USP (also commonly known as VP-16) is a semisynthetic derivative of podophyllotoxin used in the treatment of certain neoplastic diseases. It is 4'-demethylepipodophyllotoxin 9-[4,6-0-(R)-ethylidene-β-D-glucopyranoside]. It is very soluble in methanol and chloroform, slightly soluble in ethanol, and sparingly soluble in water and ether. It is made more miscible with water by means of organic solvents.
Etoposide Injection, USP is available for intravenous use as a sterile 20 mg per mL solution in 100 mg (5 mL), 500 mg (25 mL), or 1 g (50 mL) sterile, multiple dose vials. The pH of the clear, colorless to yellow solution is 3.0 to 4.0.
Each mL contains: 20 mg etoposide, USP, 2 mg citric acid anhydrous, 80 mg polysorbate 80, 650 mg polyethylene glycol 300 (57.5% v/v and 65.0% w/v), and 262 mg dehydrated alcohol (33.2% v/v and 26.2% w/v).
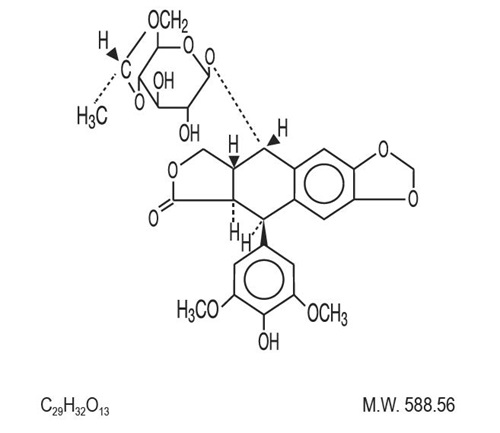
The structural formula is:
CLINICAL PHARMACOLOGY
Etoposide Injection has been shown to cause metaphase arrest in chick fibroblasts. Its main effect, however, appears to be at the G 2 portion of the cell cycle in mammalian cells. Two different dose-dependent responses are seen. At high concentrations (10 mcg/mL or more), lysis of cells entering mitosis is observed. At low concentrations (0.3 to 10 mcg/mL), cells are inhibited from entering prophase. It does not interfere with microtubular assembly. The predominant macromolecular effect of etoposide appears to be the induction of DNA strand breaks by an interaction with DNA topoisomerase II or the formation of free radicals.
Pharmacokinetics
On intravenous administration, the disposition of etoposide is best described as a biphasic process with a distribution half-life of about 1.5 hours and terminal elimination half-life ranging from 4 to 11 hours. Total body clearance values range from 33 to 48 mL/min or 16 to 36 mL/min/m 2 and, like the terminal elimination half-life, are independent of dose over a range 100 to 600 mg/m 2 . Over the same dose range, the areas under the plasma concentration vs time curves (AUC) and the maximum plasma concentration (C max ) values increase linearly with dose. Etoposide does not accumulate in the plasma following daily administration of 100 mg/m 2 for 4 to 5 days.
The mean volumes of distribution at steady state fall in the range of 18 to 29 liters or 7 to 17 L/m 2 . Etoposide enters the CSF poorly. Although it is detectable in CSF and intracerebral tumors, the concentrations are lower than in extracerebral tumors and in plasma. Etoposide concentrations are higher in normal lung than in lung metastases and are similar in primary tumors and normal tissues of the myometrium. In vitro , etoposide is highly protein bound (97%) to human plasma proteins. An inverse relationship between plasma albumin levels and etoposide renal clearance is found in children. In a study determining the effect of other therapeutic agents on the in vitro binding of carbon-14 labeled etoposide to human serum proteins, only phenylbutazone, sodium salicylate, and aspirin displaced protein-bound etoposide at concentrations achieved in vivo .
Etoposide binding ratio correlates directly with serum albumin in patients with cancer and in normal volunteers. The unbound fraction of etoposide significantly correlated with bilirubin in a population of cancer patients. Data have suggested a significant inverse correlation between serum albumin concentration and free fraction of etoposide (see PRECAUTIONS section).
After intravenous administration of 14 C-etoposide (100 to 124 mg/m 2 ), mean recovery of radioactivity in the urine was 56% of the dose at 120 hours, 45% of which was excreted as etoposide; fecal recovery of radioactivity was 44% of the dose at 120 hours.
In children, approximately 55% of the dose is excreted in the urine as etoposide in 24 hours. The mean renal clearance of etoposide is 7 to 10 mL/min/m 2 or about 35% of the total body clearance over a dose range of 80 to 600 mg/m 2 . Etoposide, therefore, is cleared by both renal and nonrenal processes, i.e., metabolism and biliary excretion. The effect of renal disease on plasma etoposide clearance is not known.
Biliary excretion of unchanged drug and/or metabolites is an important route of etoposide elimination as fecal recovery of radioactivity is 44% of the intravenous dose. The hydroxy acid metabolite [4'-demethylepipodophyllic acid-9-(4,6-O-(R)-ethylidene-β-D-glucopyranoside)], formed by opening of the lactone ring, is found in the urine of adults and children. It is also present in human plasma, presumably as the trans isomer. Glucuronide and/or sulfate conjugates of etoposide are also excreted in human urine. Only 8% or less of an intravenous dose is excreted in the urine as radiolabeled metabolites of 14 C-etoposide. In addition, O-demethylation of the dimethoxyphenol ring occurs through the CYP450 3A4 isoenzyme pathway to produce the corresponding catechol.
After either intravenous infusion or oral capsule administration, the C max and AUC values exhibit marked intra- and inter-subject variability.
In adults, the total body clearance of etoposide is correlated with creatinine clearance, serum albumin concentration, and nonrenal clearance. Patients with impaired renal function receiving etoposide have exhibited reduced total body clearance, increased AUC and a lower volume of distribution at steady state (see PRECAUTIONS section). Use of cisplatin therapy is associated with reduced total body clearance. In children, elevated serum SGPT levels are associated with reduced drug total body clearance. Prior use of cisplatin may also result in a decrease of etoposide total body clearance in children.
Although some minor differences in pharmacokinetic parameters between age and gender have been observed, these differences were not considered clinically significant.
HOW SUPPLIED
Etoposide Injection, USP, 20 mg per mL is supplied as follows:
| NDC | Etoposide Injection, USP (20 mg per mL) | Package Factor |
|---|---|---|
| 71288- 175 -05 | 100 mg per 5 mL Multi-Dose Vial | 1 vial per carton |
| 71288- 176 -25 | 500 mg per 25 mL Multi-Dose Vial | 1 vial per carton |
| 71288- 177 -50 | 1 gram per 50 mL Multi-Dose Vial | 1 vial per carton |
All are available individually packaged.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
DO NOT FREEZE.
Protect from light.
Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
Etoposide - Etoposide injection, Solution, Concentrate PubMed™ news
- Journal Article • 2026 MayAtezolizumab + Chemotherapy in Older Patients With Lung Cancer in Japan.
- Journal Article • 2026 MayAtezolizumab plus platinum-based chemotherapy and etoposide as first-line treatment for metastatic small cell lung cancer: A retrospective multicenter observational study.
- Journal Article • 2026 MayContemporary management of adrenocortical carcinoma: A narrative review.
- Journal Article • 2026 MayTargeting the GSK-3β/mTOR axis to overcome chemoresistance in gestational choriocarcinoma: molecular mechanisms and therapeutic opportunities.
- Journal Article • 2026 MayThe long-term efficacy and safety of high-dose chemotherapy with autologous stem cell transplantation in the treatment of primary central nervous system lymphoma: A real-world report from the Polish Lymphoma Research Group.