Get your patient on Everolimus
Everolimus prescribing information
INDICATIONS AND USAGE
Everolimus tablets for oral suspension are kinase inhibitor indicated for the treatment of adult and pediatric patients aged 1 year and older with TSC who have subependymal giant cell astrocytoma (SEGA) that requires therapeutic intervention but cannot be curatively resected. (1.5 )
Everolimus tablets for oral suspension is a kinase inhibitor indicated for the adjunctive treatment of adult and pediatric patients aged 2 years and older with TSC-associated partial-onset seizures. (1.6 )
Tuberous Sclerosis Complex (TSC)-Associated Subependymal Giant Cell Astrocytoma (SEGA)
Everolimus tablets for oral suspension are indicated in adult and pediatric patients aged 1 year and older with TSC for the treatment of SEGA that requires therapeutic intervention but cannot be curatively resected.
Tuberous Sclerosis Complex (TSC)-Associated Partial-Onset Seizures
Everolimus tablets for oral suspension is indicated for the adjunctive treatment of adult and pediatric patients aged 2 years and older with TSC-associated partial-onset seizures.
DOSAGE AND ADMINISTRATION
Do not combine AFINITOR and everolimus tablets for oral suspension to achieve the total daily dose. (2.1 )
Modify the dose for patients with hepatic impairment or for patients taking drugs that inhibit or induce P-glycoprotein (P-gp) and CYP3A4. (2.1 )
TSC-Associated SEGA:
4.5 mg/m 2 orally once daily; adjust dose to attain trough concentrations of 5-15 ng/mL. (2.6 , 2.8 )
TSC-Associated Partial-Onset Seizures:
Important Dosage Information
- Do not combine AFINITOR and everolimus tablets for oral suspension to achieve the total dose.
- Modify the dosage for patients with hepatic impairment or for patients taking drugs that inhibit or induce P-glycoprotein (P-gp) and CYP3A4 [see Dosage and Administration (2.10 , 2.11 , 2.12) ].
Recommended Dosage for Tuberous Sclerosis Complex (TSC)-Associated Subependymal Giant Cell Astrocytoma (SEGA)
The recommended starting dosage of everolimus tablets for oral suspension is 4.5 mg/m 2 orally once daily until disease progression or unacceptable toxicity [see Dosage and Administration (2.8) ] .
Recommended Dosage for Tuberous Sclerosis Complex (TSC)-Associated Partial-Onset Seizures
The recommended starting dosage of everolimus tablets for oral suspension is 5 mg/m 2 orally once daily until disease progression or unacceptable toxicity [see Dosage and Administration (2.8) ] .
Therapeutic Drug Monitoring (TDM) and Dose Titration for Tuberous Sclerosis Complex (TSC)-Associated Subependymal Giant Cell Astrocytoma (SEGA) and TSC-Associated Partial-Onset Seizures
- Monitor everolimus whole blood trough concentrations at time points recommended in Table 1.
- Titrate the dose to attain trough concentrations of 5 ng/mL to 15 ng/mL.
- Adjust the dose using the following equation: New dose• = current dose x (target concentration divided by current concentration) •The maximum dose increment at any titration must not exceed 5 mg. Multiple dose titrations may be required to attain the target trough concentration.
- When possible, use the same assay and laboratory for TDM throughout treatment.
Event | When to Assess Trough Concentrations After Event |
Initiation of everolimus tablets for oral suspension | 1 to 2 weeks |
Modification of everolimus tablets for oral suspension dose | 1 to 2 weeks |
Switch between AFINITOR and everolimus tablets for oral suspension | 1 to 2 weeks |
Initiation or discontinuation of P-gp and moderate CYP3A4 inhibitor | 2 weeks |
Initiation or discontinuation of P-gp and strong CYP3A4 inducer | 2 weeks |
Change in hepatic function | 2 weeks |
Stable dose with changing body surface area (BSA) | Every 3 to 6 months |
Stable dose with stable BSA | Every 6 to 12 months |
| Abbreviation: P-gp, P-glycoprotein. | |
Dosage Modifications for Adverse Reactions
Table 2 summarizes recommendations for dosage modifications of everolimus tablets for oral suspension for the management of adverse reactions.
Adverse Reaction | Severity | Dosage Modification |
| Non-infectious pneumonitis [see Warnings and Precautions (5.1) ] | Grade 2 | Withhold until improvement to Grade 0 or 1. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. Permanently discontinue if toxicity does not resolve or improve to Grade 1 within 4 weeks. |
Grade 3 | Withhold until improvement to Grade 0 or 1. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. If toxicity recurs at Grade 3, permanently discontinue. | |
Grade 4 | Permanently discontinue. | |
Stomatitis | Grade 2 | Withhold until improvement to Grade 0 or 1. Resume at same dose. If recurs at Grade 2, withhold until improvement to Grade 0 or 1. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. |
Grade 3 | Withhold until improvement to Grade 0 or 1. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. | |
Grade 4 | Permanently discontinue. | |
| Metabolic events (e.g., hyperglycemia, dyslipidemia) [see Warnings and Precautions (5.9) ] | Grade 3 | Withhold until improvement to Grade 0, 1, or 2. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. |
Grade 4 | Permanently discontinue. | |
Other non-hematologic toxicities | Grade 2 | If toxicity becomes intolerable, withhold until improvement to Grade 0 or 1. Resume at same dose. If toxicity recurs at Grade 2, withhold until improvement to Grade 0 or 1. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. |
Grade 3 | Withhold until improvement to Grade 0 or 1. Consider resuming at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. If recurs at Grade 3, permanently discontinue. | |
Grade 4 | Permanently discontinue. | |
Thrombocytopenia | Grade 2 | Withhold until improvement to Grade 0 or 1. Resume at same dose. |
Grade 3 OR Grade 4 | Withhold until improvement to Grade 0 or 1. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. | |
Neutropenia | Grade 3 | Withhold until improvement to Grade 0, 1, or 2. Resume at same dose. |
Grade 4 | Withhold until improvement to Grade 0, 1, or 2. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. | |
Febrile neutropenia | Grade 3 | Withhold until improvement to Grade 0, 1, or 2, and no fever. Resume at 50% of previous dose; change to every other day dosing if the reduced dose is lower than the lowest available strength. |
Grade 4 | Permanently discontinue. |
Dosage Modifications for Hepatic Impairment
The recommended dosages of everolimus tablets for oral suspension for patients with hepatic impairment are described in Table 3 [see Use in Specific Populations (8.6) ] :
Indication | Dose Modification for Everolimus Tablets for Oral Suspension |
| TSC-Associated SEGA and TSC-Associated Partial-Onset Seizures |
|
| Abbreviations: SEGA, Subependymal Giant Cell Astrocytoma; TSC, Tuberous Sclerosis Complex. | |
Dosage Modifications for P-gp and CYP3A4 Inhibitors
- Avoid the concomitant use of P-gp and strong CYP3A4 inhibitors [see Drug Interactions (7.1) ] .
- Avoid ingesting grapefruit and grapefruit juice.
- Reduce the dose for patients taking everolimus tablets for oral suspension with a P-gp and moderate CYP3A4 inhibitor as recommended in Table 4 [see Drug Interactions (7.1) , Clinical Pharmacology (12.3) ] .
Indication | Dose Modification for Everolimus Tablets for Oral Suspension |
| TSC-Associated SEGA and TSC-Associated Partial-Onset Seizures |
|
Dosage Modifications for P-gp and CYP3A4 Inducers
Avoid concomitant use of St. John’s Wort (Hypericum perforatum) .
Increase the dose for patients taking everolimus tablets for oral suspension with a P-gp and strong CYP3A4 inducer as recommended in Table 5 [see Drug Interactions (7.1) , Clinical Pharmacology (12.3) ] .
Indication | Dose Modification for Everolimus Tablets for Oral Suspension |
TSC-Associated SEGA and TSC-Associated Partial-Onset Seizures |
|
Administration and Preparation
Administer everolimus tablets for oral suspension at the same time each day.
Administer everolimus tablets for oral suspension consistently either with or without food [see Clinical Pharmacology (12.3) ] .
If a dose of everolimus tablets for oral suspension is missed, it can be administered up to 6 hours after the time it is normally administered. After more than 6 hours, the dose should be skipped for that day. The next day, everolimus tablets for oral suspension should be administered at its usual time. Double doses should not be administered to make up for the dose that was missed.
Everolimus tablets for oral suspension
Wear gloves to avoid possible contact with everolimus when preparing suspensions of everolimus tablets for oral suspension for another person.
Administer as a suspension only.
Administer suspension immediately after preparation. Discard suspension if not administered within 60 minutes after preparation.
Prepare suspension in water only.
Using an Oral Syringe to Prepare Oral Suspension:
Place the prescribed dose into a 10-mL syringe. Do not exceed a total of 10 mg per syringe. If higher doses are required, prepare an additional syringe. Do not break or crush tablets.
Draw approximately 5 mL of water and 4 mL of air into the syringe.
Place the filled syringe into a container (tip up) for 3 minutes, until the tablets are in suspension.
Gently invert the syringe 5 times immediately prior to administration.
After administration of the prepared suspension, draw approximately 5 mL of water and 4 mL of air into the same syringe, and swirl the contents to suspend remaining particles. Administer the entire contents of the syringe.
Using a Small Drinking Glass to Prepare Oral Suspension:
Place the prescribed dose into a small drinking glass (maximum size 100 mL) containing approximately 25 mL of water. Do not exceed a total of 10 mg per glass. If higher doses are required, prepare an additional glass. Do not break or crush tablets.
Allow 3 minutes for suspension to occur.
Stir the contents gently with a spoon, immediately prior to drinking.
After administration of the prepared suspension, add 25 mL of water and stir with the same spoon to re-suspend remaining particles. Administer the entire contents of the glass.
DOSAGE FORMS AND STRENGTHS
Everolimus tablets for oral suspension
2 mg tablets: White to off white, round shaped, flat faced bevelled edged tablets debossed with "E2" on one side and plain on other side and free from physical defects.
3 mg tablets: White to off white, round shaped, flat faced bevelled edged tablets debossed with "E3" on one side and plain on other side and free from physical defects.
5 mg tablets: White to off white, round shaped, flat faced bevelled edged tablets debossed with "E5" on one side and plain on other side and free from physical defects.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Based on animal studies and the mechanism of action [see Clinical Pharmacology (12.1) ] , everolimus tablets for oral suspension can cause fetal harm when administered to a pregnant woman. There are limited case reports of AFINITOR use in pregnant women; however, these reports are not sufficient to inform about risks of birth defects or miscarriage. In animal studies, everolimus caused embryo-fetal toxicities in rats when administered during the period of organogenesis at maternal exposures that were lower than human exposures at the recommended dose of AFINITOR 10 mg orally once daily (see Data ) . Advise pregnant women of the potential risk to the fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage is 2% to 4% and 15% to 20% of clinically recognized pregnancies, respectively.
Data
Animal Data
In animal reproductive studies, oral administration of everolimus to female rats before mating and through organogenesis induced embryo-fetal toxicities, including increased resorption, pre-implantation and post-implantation loss, decreased numbers of live fetuses, malformation (e.g., sternal cleft), and retarded skeletal development. These effects occurred in the absence of maternal toxicities. Embryo-fetal toxicities in rats occurred at doses ≥ 0.1 mg/kg (0.6 mg/m 2 ) with resulting exposures of approximately 4% of the human exposure at the recommended dose of AFINITOR 10 mg orally once daily based on area under the curve (AUC). In rabbits, embryo-toxicity evident as an increase in resorptions occurred at an oral dose of 0.8 mg/kg (9.6 mg/m 2 ), approximately 1.6 times the recommended dose of AFINITOR 10 mg orally once daily or the median dose administered to patients with tuberous sclerosis complex (TSC)-associated subependymal giant cell astrocytoma (SEGA), and 1.3 times the median dose administered to patients with TSC-associated partial-onset seizures based on BSA. The effect in rabbits occurred in the presence of maternal toxicities.
In a pre- and post-natal development study in rats, animals were dosed from implantation through lactation. At the dose of 0.1 mg/kg (0.6 mg/m 2 ), there were no adverse effects on delivery and lactation or signs of maternal toxicity; however, there were reductions in body weight (up to 9% reduction from the control) and in survival of offspring (~5% died or missing). There were no drug-related effects on the developmental parameters (morphological development, motor activity, learning, or fertility assessment) in the offspring.
Lactation
Risk Summary
There are no data on the presence of everolimus or its metabolites in human milk, the effects of everolimus on the breastfed infant or on milk production. Everolimus and its metabolites passed into the milk of lactating rats at a concentration 3.5 times higher than in maternal serum. Because of the potential for serious adverse reactions in breastfed infants from everolimus, advise women not to breastfeed during treatment with everolimus tablets for oral suspension and for 2 weeks after the last dose.
Females and Males of Reproductive Potential
Pregnancy Testing
Verify the pregnancy status of females of reproductive potential prior to starting everolimus tablets for oral suspension [see Use in Specific Populations (8.1) ] .
Contraception
Everolimus tablets for oral suspension can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1) ] .
Females: Advise female patients of reproductive potential to use effective contraception during treatment with everolimus tablets for oral suspension and for 8 weeks after the last dose.
Males: Advise male patients with female partners of reproductive potential to use effective contraception during treatment with everolimus tablets for oral suspension and for 4 weeks after the last dose.
Infertility
Females: Menstrual irregularities, secondary amenorrhea, and increases in luteinizing hormone (LH) and follicle stimulating hormone (FSH) occurred in female patients taking everolimus tablets for oral suspension. Based on these findings, everolimus tablets for oral suspension may impair fertility in female patients [see Adverse Reactions (6.1), Nonclinical Toxicology (13.1)] .
Males: Cases of reversible azoospermia have been reported in male patients taking AFINITOR. In male rats, sperm motility, sperm count, plasma testosterone levels and fertility were diminished at AUC similar to those of the clinical dose of AFINITOR 10 mg orally once daily. Based on these findings, everolimus tablets for oral suspension may impair fertility in male patients [see Nonclinical Toxicology (13.1)] .
Pediatric Use
TSC-Associated SEGA
The safety and effectiveness of everolimus tablets for oral suspension have been established in pediatric patients age 1 year and older with TSC-associated SEGA that requires therapeutic intervention but cannot be curatively resected. Use of everolimus tablets for oral suspension for this indication is supported by evidence from a randomized, double-blind, placebo-controlled trial in adult and pediatric patients (EXIST-1); an open-label, single-arm trial in adult and pediatric patients (Study 2485); and additional pharmacokinetic data in pediatric patients [see Adverse Reactions (6.1) , Clinical Pharmacology (12.3) , Clinical Studies (14.5) ] . The safety and effectiveness of everolimus tablets for oral suspension have not been established in pediatric patients less than 1 year of age with TSC-associated SEGA.
In EXIST-1, the incidence of infections and serious infections were reported at a higher frequency in patients < 6 years of age. Ninety-six percent of 23 AFINITOR-treated patients < 6 years had at least one infection compared to 67% of 55 AFINITOR-treated patients ≥ 6 years. Thirty-five percent of 23 AFINITOR-treated patients < 6 years of age had at least 1 serious infection compared to 7% of 55 AFINITOR-treated patients ≥ 6 years.
Although a conclusive determination cannot be made due to the limited number of patients and lack of a comparator arm in the open label follow-up periods of EXIST-1 and Study 2485, AFINITOR did not appear to adversely impact growth and pubertal development in the 115 pediatric patients treated with AFINITOR for a median duration of 4.1 years.
TSC-Associated Partial-Onset Seizures
The safety and effectiveness of everolimus tablets for oral suspension has been established for the adjunctive treatment of pediatric patients aged 2 years and older with TSC-associated partial-onset seizures. Use of everolimus tablets for oral suspension for this indication is supported by evidence from a randomized, double-blind, placebo-controlled trial in adult and pediatric patients (EXIST-3) with additional pharmacokinetic data in pediatric patients [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.6)] . The safety and effectiveness of everolimus tablets for oral suspension and AFINITOR have not been established for the adjunctive treatment of pediatric patients less than 2 years of age with TSC-associated partial-onset seizures.
The incidence of infections and serious infections were reported at a higher frequency in patients < 6 years of age compared to patients ≥ 6 years old. Seventy-seven percent of 70 everolimus tablets for oral suspension-treated patients < 6 years had at least one infection, compared to 53% of 177 everolimus tablets for oral suspension-treated patients ≥ 6 years. Sixteen percent of 70 everolimus tablets for oral suspension-treated patients < 6 years of age had at least 1 serious infection, compared to 4% of 177 everolimus tablets for oral suspension-treated patients ≥ 6 years of age. Two fatal cases due to infections were reported in pediatric patients.
Hepatic Impairment
Everolimus tablets for oral suspension exposure may increase in patients with hepatic impairment [see Clinical Pharmacology (12.3) ] .
For patients with TSC-associated SEGA and TSC-associated partial-onset seizures who have severe hepatic impairment (Child-Pugh class C), reduce the starting dose of everolimus tablets for oral suspension as recommended and adjust the dose based on everolimus trough concentrations [see Dosage and Administration (2.8 , 2.10) ] .
CONTRAINDICATIONS
Everolimus tablets for oral suspension are contraindicated in patients with clinically significant hypersensitivity to everolimus or to other rapamycin derivatives [see Warnings and Precautions (5.3) ] .
WARNINGS AND PRECAUTIONS
- Non-Infectious Pneumonitis: Monitor for clinical symptoms or radiological changes. Withhold or permanently discontinue based on severity. (2.9 , 5.1 )
- Infections: Monitor for signs and symptoms of infection. Withhold or permanently discontinue based on severity. (2.9 , 5.2 )
- Severe Hypersensitivity Reactions: Permanently discontinue for clinically significant hypersensitivity. (5.3 )
- Angioedema: Patients taking concomitant angiotensin-converting-enzyme (ACE) inhibitors may be at increased risk for angioedema. Permanently discontinue for angioedema. (5.4 , 7.2 )
- Stomatitis: Initiate dexamethasone alcohol-free mouthwash when starting treatment. (5.5, 6.1 )
- Renal Failure: Monitor renal function prior to treatment and periodically thereafter. (5.6 )
- Risk of Impaired Wound Healing: Withhold for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment after resolution of wound healing complications has not been established. (5.7 )
- Metabolic Disorders: Monitor serum glucose and lipids prior to treatment and periodically thereafter. Withhold or permanently discontinue based on severity. (2.9 , 5.9 )
- Myelosuppression: Monitor hematologic parameters prior to treatment and periodically thereafter. Withhold or permanently discontinue based on severity. (2.9 , 5.10 )
- Risk of Infection or Reduced Immune Response with Vaccination: Avoid live vaccines and close contact with those who have received live vaccines. Complete recommended childhood vaccinations prior to starting treatment. (5.11 )
- Radiation Sensitization and Radiation Recall: Severe radiation reactions may occur. (5.12 , 6.2 )
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.13 , 8.1 , 8.3 )
Non-infectious Pneumonitis
Non-infectious pneumonitis is a class effect of rapamycin derivatives. Non-infectious pneumonitis was reported in up to 19% of patients treated with everolimus tablets for oral suspension in clinical trials, some cases were reported with pulmonary hypertension (including pulmonary arterial hypertension) as a secondary event. The incidence of Grade 3 and 4 non-infectious pneumonitis was up to 4% and up to 0.2%, respectively [see Adverse Reactions (6.1) ] . Fatal outcomes have been observed.
Consider a diagnosis of non-infectious pneumonitis in patients presenting with non-specific respiratory signs and symptoms. Consider opportunistic infections, such as pneumocystis jiroveci pneumonia (PJP) in the differential diagnosis. Advise patients to report promptly any new or worsening respiratory symptoms.
Continue everolimus tablets for oral suspension without dose alteration in patients who develop radiological changes suggestive of non-infectious pneumonitis and have few or no symptoms. Imaging appears to overestimate the incidence of clinical pneumonitis.
For Grade 2 to 4 non-infectious pneumonitis, withhold or permanently discontinue everolimus tablets for oral suspension based on severity [see Dosage and Administration (2.9) ] . Corticosteroids may be indicated until clinical symptoms resolve. Administer prophylaxis for PJP when concomitant use of corticosteroids or other immunosuppressive agents are required. The development of pneumonitis has been reported even at a reduced dose.
Infections
Everolimus tablets for oral suspension has immunosuppressive properties and may predispose patients to bacterial, fungal, viral, or protozoal infections, including infections with opportunistic pathogens [see Adverse Reactions (6.1) ] . Localized and systemic infections, including pneumonia, mycobacterial infections, other bacterial infections, invasive fungal infections (e.g., aspergillosis, candidiasis, or PJP), and viral infections (e.g., reactivation of hepatitis B virus) have occurred. Some of these infections have been severe (e.g., sepsis, septic shock, or resulting in multisystem organ failure) or fatal. The incidence of Grade 3 and 4 infections was up to 10% and up to 3%, respectively. The incidence of serious infections was reported at a higher frequency in patients < 6 years of age [see Use in Specific Populations (8.4) ] .
Complete treatment of preexisting invasive fungal infections prior to starting treatment. Monitor for signs and symptoms of infection. Withhold or permanently discontinue everolimus tablets for oral suspension based on severity of infection [see Dosage and Administration (2.9) ].
Administer prophylaxis for PJP when concomitant use of corticosteroids or other immunosuppressive agents are required.
Severe Hypersensitivity Reactions
Hypersensitivity reactions to everolimus tablets for oral suspension have been observed and include anaphylaxis, dyspnea, flushing, chest pain, and angioedema (e.g., swelling of the airways or tongue, with or without respiratory impairment) [see Contraindications (4) ] . The incidence of Grade 3 hypersensitivity reactions was up to 1%. Permanently discontinue everolimus tablets for oral suspension for the development of clinically significant hypersensitivity.
Angioedema with Concomitant Use of Angiotensin-Converting Enzyme (ACE) Inhibitors
Patients taking concomitant ACE inhibitors with everolimus tablets for oral suspension may be at increased risk for angioedema (e.g., swelling of the airways or tongue, with or without respiratory impairment). In a pooled analysis of randomized double-blind oncology clinical trials, the incidence of angioedema in patients taking AFINITOR with an ACE inhibitor was 6.8% compared to 1.3% in the control arm with an ACE inhibitor. Permanently discontinue everolimus tablets for oral suspension for angioedema.
Stomatitis
Stomatitis, including mouth ulcers and oral mucositis, has occurred in patients treated with everolimus tablets for oral suspension at an incidence ranging from 44% to 78% across clinical trials. Grades 3-4 stomatitis was reported in 4% to 9% of patients. Stomatitis most often occurs within the first 8 weeks of treatment. When starting everolimus tablets for oral suspension, initiating dexamethasone alcohol-free oral solution as a swish and spit mouthwash reduces the incidence and severity of stomatitis. If stomatitis does occur, mouthwashes and/or other topical treatments are recommended. Avoid alcohol-, hydrogen peroxide-, iodine-, or thyme-containing products, as they may exacerbate the condition. Do not administer antifungal agents, unless fungal infection has been diagnosed.
Renal Failure
Cases of renal failure (including acute renal failure), some with a fatal outcome, have occurred in patients taking AFINITOR. Elevations of serum creatinine and proteinuria have been reported in patients taking everolimus tablets for oral suspension [see Adverse Reactions (6.1) ] . The incidence of Grade 3 and 4 elevations of serum creatinine was up to 2% and up to 1%, respectively. The incidence of Grade 3 and 4 proteinuria was up to 1% and up to 0.5%, respectively. Monitor renal function prior to starting everolimus tablets for oral suspension and annually thereafter. Monitor renal function at least every 6 months in patients who have additional risk factors for renal failure.
Risk of Impaired Wound Healing
Impaired wound healing can occur in patients who receive drugs that inhibit the VEGF signaling pathway. Therefore, everolimus tablets for oral suspension have the potential to adversely affect wound healing.
Withhold everolimus tablets for oral suspension for at least 1 week prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment upon resolution of wound healing complications has not been established.
Metabolic Disorders
Hyperglycemia, hypercholesterolemia, and hypertriglyceridemia have been reported in patients taking everolimus tablets for oral suspension at an incidence up to 75%, 86%, and 73%, respectively. The incidence of these Grade 3 and 4 laboratory abnormalities was up to 15% and up to 0.4%, respectively [see Adverse Reactions (6.1) ] . In non-diabetic patients, monitor fasting serum glucose prior to starting everolimus tablets for oral suspension and annually thereafter. In diabetic patients, monitor fasting serum glucose more frequently as clinically indicated. Monitor lipid profile prior to starting everolimus tablets for oral suspension and annually thereafter. When possible, achieve optimal glucose and lipid control prior to starting everolimus tablets for oral suspension. For Grade 3 to 4 metabolic events, withhold or permanently discontinue everolimus tablets for oral suspension based on severity [see Dosage and Administration (2.9) ] .
Myelosuppression
Anemia, lymphopenia, neutropenia, and thrombocytopenia have been reported in patients taking everolimus tablets for oral suspension. The incidence of these Grade 3 and 4 laboratory abnormalities was up to 16% and up to 2%, respectively [see Adverse Reactions (6.1) ] . Monitor complete blood count (CBC) prior to starting everolimus tablets for oral suspension every 6 months for the first year of treatment and annually thereafter. Withhold or permanently discontinue everolimus tablets for oral suspension based on severity [see Dosage and Administration (2.9) ] .
Risk of Infection or Reduced Immune Response With Vaccination
The safety of immunization with live vaccines during everolimus tablets for oral suspension therapy has not been studied. Due to the potential increased risk of infection, avoid the use of live vaccines and close contact with individuals who have received live vaccines during treatment with everolimus tablets for oral suspension. Due to the potential increased risk of infection or reduced immune response with vaccination, complete the recommended childhood series of vaccinations according to American Council on Immunization Practices (ACIP) guidelines prior to the start of therapy. An accelerated vaccination schedule may be appropriate.
Radiation Sensitization and Radiation Recall
Radiation sensitization and recall, in some cases severe, involving cutaneous and visceral organs (including radiation esophagitis and pneumonitis) have been reported in patients treated with radiation prior to, during, or subsequent to everolimus tablets for oral suspension treatment [see Adverse Reactions (6.2) ] .
Monitor patients closely when everolimus tablets for oral suspension is administered during or sequentially with radiation treatment.
Embryo-Fetal Toxicity
Based on animal studies and the mechanism of action, everolimus tablets for oral suspension can cause fetal harm when administered to a pregnant woman. In animal studies, everolimus caused embryo-fetal toxicities in rats when administered during the period of organogenesis at maternal exposures that were lower than human exposures at the clinical dose of 10 mg once daily. Advise pregnant women of the potential risk to a fetus. Advise female patients of reproductive potential to avoid becoming pregnant and to use effective contraception during treatment with everolimus tablets for oral suspension and for 8 weeks after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with everolimus tablets for oral suspension and for 4 weeks after the last dose [see Use in Specific Populations (8.1 , 8.3) ].
ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
- Non-Infectious Pneumonitis [see Warnings and Precautions (5.1) ]
- Infections [see Warnings and Precautions (5.2) ]
- Severe Hypersensitivity Reactions [see Warnings and Precautions (5.3) ]
- Angioedema with Concomitant Use of ACE inhibitors [see Warnings and Precautions (5.4) ]
- Stomatitis [see Warnings and Precautions (5.5) ]
- Renal Failure [see Warnings and Precautions (5.6) ]
- Impaired Wound Healing [see Warnings and Precautions (5.7) ]
- Metabolic Disorders [see Warnings and Precautions (5.9) ]
- Myelosuppression [see Warnings and Precautions (5.10) ]
- Radiation Sensitization and Radiation Recall [see Warnings and Precautions (5.12) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other trials and may not reflect the rates observed in clinical practice.
TSC-Associated Subependymal Giant Cell Astrocytoma (SEGA)
The data described below are based on a randomized (2:1), double-blind, placebo-controlled trial (EXIST-1) of AFINITOR in 117 patients with SEGA and TSC. The median age of patients was 9.5 years (0.8 to 26 years), 93% were white, and 57% were male. The median duration of blinded study treatment was 52 weeks (24 to 89 weeks) for patients receiving AFINITOR.
The most common adverse reactions reported for AFINITOR (incidence ≥ 30%) were stomatitis and respiratory tract infection. The most common Grade 3-4 adverse reactions (incidence ≥ 2%) were stomatitis, pyrexia, pneumonia, gastroenteritis, aggression, agitation, and amenorrhea. The most common laboratory abnormalities (incidence ≥ 50%) were hypercholesterolemia and elevated partial thromboplastin time. The most common Grade 3-4 laboratory abnormality (incidence ≥ 3%) was neutropenia.
There were no adverse reactions resulting in permanent discontinuation. Dose adjustments (interruptions or reductions) due to adverse reactions occurred in 55% of AFINITOR-treated patients. The most common adverse reaction leading to AFINITOR dose adjustment was stomatitis.
Adverse reactions reported with an incidence of ≥ 10% for patients receiving AFINITOR and occurring more frequently with AFINITOR than with placebo are reported in Table 16. Laboratory abnormalities are presented in Table 17.
Grading according to NCI CTCAE Version 3.0. a Includes mouth ulceration, stomatitis, and lip ulceration. b Includes respiratory tract infection, upper respiratory tract infection, and respiratory tract infection viral. c Includes gastroenteritis, gastroenteritis viral, and gastrointestinal infection. d Includes agitation, anxiety, panic attack, aggression, abnormal behavior, and obsessive compulsive disorder. e Includes rash, rash generalized, rash macular, rash maculo-papular, rash papular, dermatitis allergic, and urticaria. f No Grade 4 adverse reactions were reported. | ||||
AFINITOR N = 78 | Placebo N = 39 | |||
All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % | |
Gastrointestinal | ||||
Stomatitis a | 62 | 9 f | 26 | 3 f |
Vomiting | 22 | 1 f | 13 | 0 |
Diarrhea | 17 | 0 | 5 | 0 |
Constipation | 10 | 0 | 3 | 0 |
Infections | ||||
Respiratory tract infection b | 31 | 3 | 23 | 0 |
Gastroenteritis c | 10 | 5 | 3 | 0 |
Pharyngitis streptococcal | 10 | 0 | 3 | 0 |
General | ||||
Pyrexia | 23 | 6 f | 18 | 3 f |
Fatigue | 14 | 0 | 3 | 0 |
Psychiatric | ||||
Anxiety, aggression or other behavioral disturbance d | 21 | 5 f | 3 | 0 |
Skin and subcutaneous tissue | ||||
Rash e | 21 | 0 | 8 | 0 |
Acne | 10 | 0 | 5 | 0 |
Amenorrhea occurred in 17% of AFINITOR-treated females aged 10 to 55 years (3 of 18). For this same group of AFINITOR-treated females, the following menstrual abnormalities were reported: dysmenorrhea (6%), menorrhagia (6%), metrorrhagia (6%), and unspecified menstrual irregularity (6%).
The following additional adverse reactions occurred in less than 10% of AFINITOR-treated patients: nausea (8%), pain in extremity (8%), insomnia (6%), pneumonia (6%), epistaxis (5%), hypersensitivity (3%), increased blood luteinizing hormone (LH) levels (1%), and pneumonitis (1%).
Grading according to NCI CTCAE Version 3.0. a No Grade 4 laboratory abnormalities were reported. | ||||
AFINITOR N = 78 | Placebo N = 39 | |||
All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % | |
Hematology | ||||
Elevated partial thromboplastin time | 72 | 3 a | 44 | 5 a |
Neutropenia | 46 | 9 a | 41 | 3 a |
Anemia | 41 | 0 | 21 | 0 |
Chemistry | ||||
Hypercholesterolemia | 81 | 0 | 39 | 0 |
Elevated AST | 33 | 0 | 0 | 0 |
Hypertriglyceridemia | 27 | 0 | 15 | 0 |
Elevated ALT | 18 | 0 | 3 | 0 |
Hypophosphatemia | 9 | 1 a | 3 | 0 |
Updated safety information from 111 patients treated with AFINITOR for a median duration of 47 months identified the following additional notable adverse reactions and selected laboratory abnormalities: decreased appetite (14%), hyperglycemia (13%), hypertension (11%), urinary tract infection (9%), decreased fibrinogen (8%), cellulitis (6%), abdominal pain (5%), decreased weight (5%), elevated creatinine (5%), and azoospermia (1%).
TSC-Associated Partial-Onset Seizures
The data described below are based on the 18-week Core phase of a randomized, double-blind, multicenter, three-arm trial (EXIST-3) comparing two everolimus trough levels (3-7 ng/mL and 9-15 ng/mL) to placebo as adjunctive antiepileptic therapy in patients with TSC-associated partial-onset seizures. A total of 366 patients were randomized to everolimus tablets for oral suspension low trough (LT) (n = 117), everolimus tablets for oral suspension high trough (HT) (n = 130), or placebo (n = 119). The median age of patients was 10 years (2.2 to 56 years; 28% were < 6 years, 31% were 6 to < 12 years, 22% were 12 to < 18 years, and 18% were ≥ 18 years), 65% were white, and 52% were male. Patients received between one and three concomitant antiepileptic drugs.
The most common adverse reaction reported for everolimus tablets for oral suspension in both arms (incidence ≥ 30%) was stomatitis. The most common Grade 3-4 adverse reactions (incidence ≥ 2%) were stomatitis, pneumonia, and irregular menstruation. The most common laboratory abnormality (incidence ≥ 50%) was hypercholesterolemia. The most common Grade 3-4 laboratory abnormality (incidence ≥ 2%) was neutropenia.
Adverse reactions leading to study drug discontinuation occurred in 5% and 3% of patients in the LT and HT arms, respectively. The most common adverse reaction (incidence ≥ 1%) leading to discontinuation was stomatitis. Dose adjustments (interruptions or reductions) due to adverse reactions occurred in 24% and 35% of patients in the LT and HT arms, respectively. The most common adverse reactions (incidence ≥ 3%) leading to dose adjustments in the everolimus tablets for oral suspension arms were stomatitis, pneumonia, and pyrexia.
Adverse reactions reported with an incidence of ≥ 10% for patients receiving everolimus tablets for oral suspension are presented in Table 18. Laboratory abnormalities are presented in Table 19.
Table 18: Adverse Reactions Reported in ≥ 10% of Everolimus Tablets for Oral Suspension-Treated Patients With TSC-Associated Partial-Onset Seizures in EXIST-3
| Everolimus Tablets for Oral Suspension | Placebo | ||||||
Target of 3-7 ng/mL N = 117 | Target of 9-15 ng/mL N = 130 | N = 119 | |||||
All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % | ||
| Gastrointestinal | |||||||
| Stomatitis a | 55 | 3b | 64 | 4b | 9 | 0 | |
| Diarrhea | 17 | 0 | 22 | 0 | 5 | 0 | |
| Vomiting | 12 | 0 | 10 | 2b | 9 | 0 | |
| Infections | |||||||
| Nasopharyngitis | 14 | 0 | 16 | 0 | 16 | 0 | |
| Upper respiratory tract infection | 13 | 0 | 15 | 0 | 13 | 0.8 b | |
| General | |||||||
| Pyrexia | 20 | 0 | 14 | 0.8 b | 5 | 0 | |
| Respiratory, thoracic and mediastinal | |||||||
| Cough | 11 | 0 | 10 | 0 | 3 | 0 | |
| Skin and subcutaneous tissue | |||||||
| Rash | 6 | 0 | 10 | 0 | 3 | 0 | |
Grading according to NCI CTCAE Version 4.03. a Includes stomatitis, mouth ulceration, aphthous ulcer, lip ulceration, tongue ulceration, mucosal inflammation, gingival pain. b No Grade 4 adverse reactions were reported. | |||||||
The following additional adverse reactions occurred in < 10% of everolimus tablets for oral suspension treated patients (% everolimus tablets for oral suspension LT, % everolimus tablets for oral suspension HT): decreased appetite (9%, 7%), pneumonia (2%, 4%), aggression (2%, 0.8%), proteinuria (0%, 2%), menorrhagia (0.9%, 0.8%), and pneumonitis (0%, 0.8%).
Table 19: Selected Laboratory Abnormalities Reported in ≥ 10% Everolimus Tablets for Oral Suspension-Treated Patients With TSC-Associated Partial-Onset Seizures
| Everolimus Tablets for Oral Suspension | Placebo | |||||
Target of 3-7 ng/mL N = 117 | Target of 9-15 ng/mL N = 130 | N = 119 | ||||
All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % | All Grades % | Grade 3-4 % | |
| Hematology | ||||||
| Neutropenia | 25 | 4 a | 37 | 6 | 23 | 7 a |
| Anemia | 27 | 0.9 a | 30 | 0 | 21 | 0.8 a |
| Thrombocytopenia | 12 | 0 | 15 | 0 | 6 | 0 |
| Chemistry | ||||||
| Hypercholesterolemia | 86 | 0 | 85 | 0.8 a | 58 | 0 |
| Hypertriglyceridemia | 43 | 2 a | 39 | 2 | 22 | 0 |
| Increased ALT | 17 | 0 | 22 | 0 | 6 | 0 |
| Increased AST | 13 | 0 | 19 | 0 | 4 | 0 |
| Hyperglycemia | 19 | 0 | 18 | 0 | 17 | 0 |
| Increased alkaline phosphatase | 24 | 0 | 16 | 0 | 29 | 0 |
| Hypophosphatemia | 9 | 0.9 a | 16 | 2 | 3 | 0 |
| Grading according to NCI CTCAE version 4.03. | ||||||
| a No Grade 4 laboratory abnormalities were reported. | ||||||
Updated safety information from 357 patients treated with everolimus tablets for oral suspension for a median duration of 48 weeks identified the following additional notable adverse reactions: hypersensitivity (0.6%), angioedema (0.3%), and ovarian cyst (0.3%).
Postmarketing Experience
The following adverse reactions have been identified during postapproval use of everolimus tablets for oral suspension. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate frequency or establish a causal relationship to drug exposure:
Blood and lymphatic disorders: Thrombotic microangiopathy
Cardiac: Cardiac failure with some cases reported with pulmonary hypertension (including pulmonary arterial hypertension) as a secondary event
Gastrointestinal: Acute pancreatitis
Hepatobiliary: Cholecystitis and cholelithiasis
Infections: Sepsis and septic shock
Nervous system: Reflex sympathetic dystrophy
Vascular: Arterial thrombotic events, lymphedema
Injury, poisoning and procedural complications: Radiation Sensitization and Radiation Recall
DRUG INTERACTIONS
Effect of Other Drugs on Everolimus Tablets for Oral Suspension
Inhibitors
Avoid the concomitant use of P-gp and strong CYP3A4 inhibitors [see Dosage and Administration (2.11) , Clinical Pharmacology (12.3) ] .
Reduce the dose for patients taking everolimus tablets for oral suspension with a P-gp and moderate CYP3A4 inhibitor as recommended [see Dosage and Administration (2.11) , Clinical Pharmacology (12.3) ].
Inducers
Increase the dose for patients taking everolimus tablets for oral suspension with a P-gp and strong CYP3A4 inducer as recommended [see Dosage and Administration (2.12) , Clinical Pharmacology (12.3) ].
Effects of Combination Use of Angiotensin Converting Enzyme (ACE) Inhibitors
Patients taking concomitant ACE inhibitors with everolimus tablets for oral suspension may be at increased risk for angioedema. Avoid the concomitant use of ACE inhibitors with everolimus tablets for oral suspension [see Warnings and Precautions (5.4) ] .
DESCRIPTION
Everolimus tablets for oral suspension are kinase inhibitor.
The chemical name of everolimus USP is (1R,9S,12S,15R,16E,18R,19R,21R,23S,24E,26E,28E,30S,32S,35R)-1,18- dihydroxy-12-{(1R)-2-[(1S,3R,4R)-4-(2-hydroxyethoxy)-3-methoxycyclohexyl]-1-methylethyl}-19,30-dimethoxy-15,17,21,23,29,35-hexamethyl-11,36-dioxa-4-aza-tricyclo[30.3.1.0 4,9 ]hexatriaconta-16,24,26,28-tetraene-2,3,10,14,20-pentaone. The molecular formula is C 53 H 83 NO 14 and the molecular weight is 958.2 g/mol. The structural formula is:
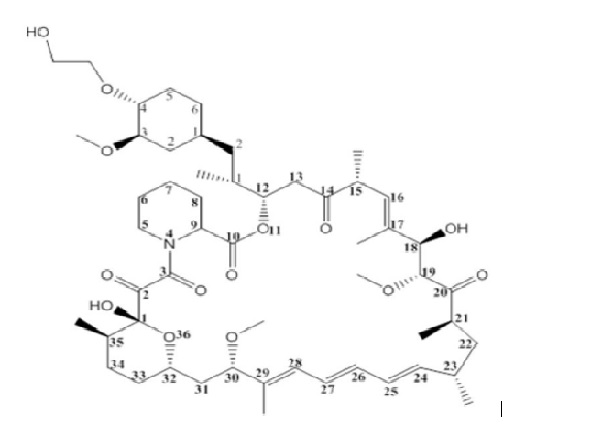
Everolimus tablets for oral suspension for oral administration contains 2 mg, 3 mg or 5 mg of everolimus, USP and the following inactive ingredients: butylated hydroxytoluene, colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, mannitol, and microcrystalline cellulose.
CLINICAL PHARMACOLOGY
Mechanism of Action
Everolimus is an inhibitor of mammalian target of rapamycin (mTOR), a serine-threonine kinase, downstream of the PI3K/AKT pathway. The mTOR pathway is dysregulated in several human cancers and in tuberous sclerosis complex (TSC). Everolimus binds to an intracellular protein, FKBP-12, resulting in an inhibitory complex formation with mTOR complex 1 (mTORC1) and thus inhibition of mTOR kinase activity. Everolimus reduced the activity of S6 ribosomal protein kinase (S6K1) and eukaryotic initiation factor 4E-binding protein (4E-BP1), downstream effectors of mTOR, involved in protein synthesis. S6K1 is a substrate of mTORC1 and phosphorylates the activation domain 1 of the estrogen receptor which results in ligand-independent activation of the receptor. In addition, everolimus inhibited the expression of hypoxia-inducible factor (e.g., HIF-1) and reduced the expression of vascular endothelial growth factor (VEGF). Inhibition of mTOR by everolimus has been shown to reduce cell proliferation, angiogenesis, and glucose uptake in in vitro and/or in vivo studies.
Two regulators of mTORC1 signaling are the oncogene suppressors tuberin-sclerosis complexes 1 and 2 ( TSC1, TSC2 ). Loss or inactivation of either TSC1 or TSC2 leads to activation of downstream signaling. In TSC, a genetic disorder, inactivating mutations in either the TSC1 or the TSC2 gene lead to hamartoma formation throughout the body as well as seizures and epileptogenesis. Overactivation of mTOR results in neuronal dysplasia, aberrant axonogenesis and dendrite formation, increased excitatory synaptic currents, reduced myelination, and disruption of the cortical laminar structure causing abnormalities in neuronal development and function. Treatment with an mTOR inhibitor in animal models of mTOR dysregulation in the brain resulted in seizure suppression, prevention of the development of new-onset seizures, and prevention of premature death.
Pharmacodynamics
Exposure-Response Relationship
In patients with TSC-associated subependymal giant cell astrocytoma (SEGA), the magnitude of the reduction in SEGA volume was correlated with the everolimus trough concentration.
In patients with TSC-associated partial-onset seizures, the magnitude of the reduction in absolute seizure frequency was correlated with the everolimus trough concentration.
Cardiac Electrophysiology
In a randomized, placebo-controlled, cross-over study, 59 healthy subjects were administered a single oral dose of AFINITOR (20 mg and 50 mg) and placebo. AFINITOR at single doses up to 50 mg did not prolong the QT/QTc interval.
Pharmacokinetics
Absorption
In patients with TSC-associated SEGA, everolimus C min was approximately dose-proportional within the dose range from 1.35 mg/m 2 to 14.4 mg/m 2 .
Effect of Food: In healthy subjects who received 9 mg of everolimus tablets for oral suspension, high-fat meals (containing approximately 1000 calories and 55 grams of fat) reduced everolimus AUC by 12% and C max by 60% and low-fat meals (containing approximately 500 calories and 20 grams of fat) reduced everolimus AUC by 30% and C max by 50%.
Relative Bioavailability : The AUC inf of everolimus was equivalent between everolimus tablets for oral suspension and AFINITOR; the C max of everolimus in the everolimus tablets for oral suspension dosage form was 20% to 36% lower than that of AFINITOR. The predicted trough concentrations at steady-state were similar after daily administration.
Distribution
The blood-to-plasma ratio of everolimus, which is concentration-dependent over the range of 5 to 5000 ng/mL, is 17% to 73%. The amount of everolimus confined to the plasma is approximately 20% at blood concentrations observed in cancer patients given AFINITOR 10 mg orally once daily. Plasma protein binding is approximately 74% both in healthy subjects and in patients with moderate hepatic impairment.
Elimination
The mean elimination half-life of everolimus is approximately 30 hours.
Metabolism : Everolimus is a substrate of CYP3A4. Following oral administration, everolimus is the main circulating component in human blood. Six main metabolites of everolimus have been detected in human blood, including three monohydroxylated metabolites, two hydrolytic ring-opened products, and a phosphatidylcholine conjugate of everolimus. These metabolites were also identified in animal species used in toxicity studies, and showed approximately 100-times less activity than everolimus itself.
Excretion : No specific elimination studies have been undertaken in cancer patients. Following the administration of a 3 mg single dose of radiolabeled everolimus in patients who were receiving cyclosporine, 80% of the radioactivity was recovered from the feces, while 5% was excreted in the urine. The parent substance was not detected in urine or feces.
Specific Populations
No relationship was apparent between oral clearance and age or sex in patients with cancer.
Patients with Renal Impairment: No significant influence of creatinine clearance (25 to 178 mL/min) was detected on oral clearance (CL/F) of everolimus.
Patients with Hepatic Impairment: Compared to normal subjects, there was a 1.8-fold, 3.2-fold, and 3.6-fold increase in AUC for subjects with mild (Child-Pugh class A), moderate (Child-Pugh class B), and severe (Child-Pugh class C) hepatic impairment, respectively. In another study, the average AUC of everolimus in subjects with moderate hepatic impairment (Child-Pugh class B) was twice that found in subjects with normal hepatic function [see Dosage and Administration (2.10), Use in Specific Populations (8.6)] .
Pediatric Patients: In patients with TSC-associated SEGA or TSC-associated partial-onset seizures, the mean C min values normalized to mg/m 2 dose in pediatric patients (< 18 years of age) were lower than those observed in adults, suggesting that everolimus clearance adjusted to BSA was higher in pediatric patients as compared to adults.
Race or Ethnicity: Based on a cross-study comparison, Japanese patients had on average exposures that were higher than non-Japanese patients receiving the same dose. Oral clearance (CL/F) is on average 20% higher in black patients than in white patients.
Drug Interaction Studies
Effect of CYP3A4 and P-glycoprotein (P-gp) Inhibitors on Everolimus : Everolimus exposure increased when AFINITOR was coadministered with:
ketoconazole (a P-gp and strong CYP3A4 inhibitor) - C max and AUC increased by 3.9- and 15-fold, respectively.
erythromycin (a P-gp and moderate CYP3A4 inhibitor) - C max and AUC increased by 2- and 4.4-fold, respectively.
verapamil (a P-gp and moderate CYP3A4 inhibitor) - C max and AUC increased by 2.3- and 3.5-fold, respectively.
Effect of CYP3A4 and P-gp Inducers on Everolimus : The coadministration of AFINITOR with rifampin, a P-gp and strong inducer of CYP3A4, decreased everolimus AUC by 63% and C max by 58% compared to AFINITOR alone [see Dosage and Administration (2.12)].
Effect of Everolimus on CYP3A4 Substrates : No clinically significant pharmacokinetic interactions were observed between AFINITOR and the HMG-CoA reductase inhibitors atorvastatin (a CYP3A4 substrate), pravastatin (a non CYP3A4-substrate), and simvastatin (a CYP3A4 substrate).
The coadministration of an oral dose of midazolam (sensitive CYP3A4 substrate) with AFINITOR resulted in a 25% increase in midazolam C max and a 30% increase in midazolam AUC 0-inf.
Effect of Everolimus on Antiepileptic Drugs (AEDs) : Everolimus increased pre-dose concentrations of the carbamazepine, clobazam, oxcarbazepine, and clobazam’s metabolite N-desmethylclobazam by about 10%. Everolimus had no impact on pre-dose concentrations of AEDs that are substrates of CYP3A4 (e.g., clonazepam and zonisamide) or other AEDs, including valproic acid, topiramate, phenobarbital, and phenytoin.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Administration of everolimus for up to 2 years did not indicate oncogenic potential in mice and rats up to the highest doses tested (0.9 mg/kg) corresponding, respectively to 3.9 and 0.2 times the estimated human exposure based on AUC at the recommended dose of AFINITOR 10 mg orally once daily.
Everolimus was not genotoxic in a battery of in vitro assays (Ames mutation test in Salmonella , mutation test in L5178Y mouse lymphoma cells, and chromosome aberration assay in V79 Chinese hamster cells). Everolimus was not genotoxic in an in vivo mouse bone marrow micronucleus test at doses up to 500 mg/kg/day (1500 mg/m 2 /day, approximately 255-fold the recommended dose of AFINITOR 10 mg orally once daily, and approximately 200-fold the median dose administered to patients with TSC-associated SEGA and TSC-associated partial-onset seizures, based on the BSA), administered as 2 doses, 24 hours apart.
Based on non-clinical findings, everolimus tablets for oral suspension may impair male fertility. In a 13-week male fertility study in rats, testicular morphology was affected at doses of 0.5 mg/kg and above. Sperm motility, sperm count, and plasma testosterone levels were diminished in rats treated with 5 mg/kg. The exposures at these doses (52 ng•hr/mL and 414 ng•hr/mL, respectively) were within the range of human exposure at the recommended dose of AFINITOR 10 mg orally once daily (560 ng•hr/mL) and resulted in infertility in the rats at 5 mg/kg. Effects on male fertility occurred at AUC 0-24h values 10% to 81% lower than human exposure at the recommended dose of AFINITOR 10 mg orally once daily. After a 10-13 week non-treatment period, the fertility index increased from zero (infertility) to 60%.
Oral doses of everolimus in female rats at doses ≥ 0.1 mg/kg (approximately 4% the human exposure based on AUC at the recommended dose of AFINITOR 10 mg orally once daily) resulted in increased incidence of pre-implantation loss, suggesting that the drug may reduce female fertility.
Animal Toxicology and/or Pharmacology
In juvenile rat toxicity studies, dose-related delayed attainment of developmental landmarks, including delayed eye-opening, delayed reproductive development in males and females and increased latency time during the learning and memory phases were observed at doses as low as 0.15 mg/kg/day.
CLINICAL STUDIES
Tuberous Sclerosis Complex (TSC)-Associated Subependymal Giant Cell Astrocytoma (SEGA)
EXIST-1
A randomized (2:1), double-blind, placebo-controlled trial (EXIST-1, NCT00789828) of AFINITOR was conducted in 117 pediatric and adult patients with SEGA and TSC. Eligible patients had at least one SEGA lesion ≥ 1 cm in longest diameter on MRI based on local radiology assessment and one or more of the following: serial radiological evidence of SEGA growth, a new SEGA lesion ≥ 1 cm in longest diameter, or new or worsening hydrocephalus. Patients randomized to the treatment arm received AFINITOR at a starting dose of 4.5 mg/m 2 daily, with subsequent dose adjustments as needed to achieve and maintain everolimus trough concentrations of 5 to 15 ng/mL as tolerated. AFINITOR or matched placebo continued until disease progression or unacceptable toxicity. MRI scans for disease assessment were obtained at baseline, 12, 24, and 48 weeks, and annually thereafter.
The main efficacy outcome measure was SEGA response rate based on independent central radiology review. SEGA response was defined as a ≥ 50% reduction in the sum of SEGA volume relative to baseline, in the absence of unequivocal worsening of non-target SEGA lesions, a new SEGA lesion ≥ 1 cm, and new or worsening hydrocephalus. The primary analysis of SEGA response rate was limited to the blinded treatment period and conducted 6 months after the last patient was randomized. The analysis of SEGA response rate was stratified by use of enzyme-inducing antiepileptic drugs (EIAEDs) at randomization (yes vs. no).
Of the 117 patients enrolled, 78 were randomized to AFINITOR and 39 to placebo. The median age was 9.5 years (0.8 to 26 years); a total of 20 patients were < 3 years, 54 patients were 3 to < 12 years, 27 patients were 12 to < 18 years, and 16 patients were ≥ 18 years; 57% were male, and 93% were white. At baseline, 18% of patients were receiving EIAEDs. Based on central radiology review at baseline, 98% of patients had at least one SEGA lesion ≥ 1.0 cm in longest diameter, 79% had bilateral SEGAs, 43% had ≥ 2 target SEGA lesions, 26% had growth in or into the inferior surface of the ventricle, 9% had evidence of growth beyond the subependymal tissue adjacent to the ventricle, and 7% had radiographic evidence of hydrocephalus. The median values for the sum of all target SEGA lesions at baseline were 1.63 cm 3 (0.18 to 25.15 cm 3 ) and 1.30 cm 3 (0.32 to 9.75 cm 3 ) in the AFINITOR and placebo arms, respectively. Eight (7%) patients had prior SEGA-related surgery. The median duration of follow-up was 8.4 months (4.6 to 17.2 months) at the time of primary analysis.
The SEGA response rate was statistically significantly higher in AFINITOR-treated patients (Table 25). At the time of the primary analysis, all SEGA responses were ongoing and the median duration of response was 5.3 months (2.1 to 8.4 months).
With a median follow-up of 8.4 months, SEGA progression was detected in 15.4% of the 39 patients randomized to receive placebo and none of the 78 patients randomized to receive AFINITOR. No patient in either treatment arm required surgical intervention.
AFINITOR N = 78 | Placebo N = 39 | p-value | |
Primary analysis | |||
SEGA response rate a - (%) | 35 | 0 | < 0.0001 |
95% CI | 24, 46 | 0, 9 | |
| a Per independent central radiology review. | |||
Patients randomized to placebo were permitted to receive AFINITOR at the time of SEGA progression or after the primary analysis, whichever occurred first. After the primary analysis, patients treated with AFINITOR underwent additional follow-up MRI scans to assess tumor status until discontinuation of treatment or completion of 4 years of follow-up after the last patient was randomized. A total of 111 patients (78 patients randomized to AFINITOR and 33 patients randomized to placebo) received at least one dose of AFINITOR. Median duration of AFINITOR treatment and follow-up was 3.9 years (0.2 to 4.9 years).
By four years after the last patient was enrolled, 58% of the 111 patients treated with AFINITOR had a ≥ 50% reduction in SEGA volume relative to baseline, including 27 patients identified at the time of the primary analysis and 37 patients with a SEGA response after the primary analysis. The median time to SEGA response was 5.3 months (2.5 to 33.1 months). Twelve percent of the 111 patients treated with AFINITOR had documented disease progression by the end of the follow-up period and no patient required surgical intervention for SEGA during the study.
Study 2485
Study 2485 (NCT00411619) was an open-label, single-arm trial conducted to evaluate the antitumor activity of AFINITOR 3 mg/m 2 /orally once daily in patients with SEGA and TSC. Serial radiological evidence of SEGA growth was required for entry. Tumor assessments were performed every 6 months for 60 months after the last patient was enrolled or disease progression, whichever occurred earlier. The major efficacy outcome measure was the reduction in volume of the largest SEGA lesion with 6 months of treatment, as assessed via independent central radiology review. Progression was defined as an increase in volume of the largest SEGA lesion over baseline that was ≥ 25% over the nadir observed on study.
A total of 28 patients received AFINITOR for a median duration of 5.7 years (5 months to 6.9 years); 82% of the 28 patients remained on AFINITOR for at least 5 years. The median age was 11 years (3 to 34 years), 61% male, 86% white.
At the primary analysis, 32% of the 28 patients (95% CI: 16%, 52%) had an objective response at 6 months, defined as at least a 50% decrease in volume of the largest SEGA lesion. At the completion of the study, the median duration of durable response was 12 months (3 months to 6.3 years).
By 60 months after the last patient was enrolled, 11% of the 28 patients had documented disease progression. No patient developed a new SEGA lesion while on AFINITOR. Nine additional patients were identified as having a ≥ 50% volumetric reduction in their largest SEGA lesion between 1 to 4 years after initiating AFINITOR, including 3 patients who had surgical resection with subsequent regrowth prior to receiving AFINITOR.
Tuberous Sclerosis Complex (TSC)-Associated Partial-Onset Seizures
The efficacy of everolimus tablets for oral suspension as an adjunctive anti-epileptic drug (AED) was evaluated in a randomized, double-blind, multicenter, placebo-controlled study conducted in patients with TSC-associated partial-onset seizures (EXIST-3, NCT01713946). Patients with a history of inadequate control of partial-onset seizures despite treatment with ≥ 2 sequential AED regimens were randomized to receive placebo or everolimus tablets for oral suspension once daily at a dose to achieve a low trough (LT) level (3-7 ng/mL) or a high trough (HT) level (9-15 ng/mL). Randomization was stratified by age group (1 to < 6, 6 to < 12, 12 to < 18, ≥ 18 years). The study consisted of 3 phases: an 8-week Baseline observation phase; an 18-week double-blind, placebo-controlled Core phase (6-week titration period and a 12-week maintenance period), and an Extension phase of ≥ 48 weeks. Patients were required to have a diagnosis of TSC per the modified Gomez criteria, and ≥ 16 partial-onset seizures during the Baseline phase while receiving a stable dose of 1 to 3 concomitant AEDs. The starting doses for everolimus tablets for oral suspension in the Core phase ranged from 3 to 6 mg/m 2 orally once daily, depending on age, in patients not receiving concomitant CYP3A4/P-gp inducers and from 5 to 9 mg/m 2 orally once daily, depending on age, in patients receiving concomitant CYP3A4/P-gp inducers. During the 6-week titration period, everolimus trough levels were assessed every 2 weeks and up to 3 dose adjustments were allowed to attempt to reach the targeted everolimus trough concentration range.
The major efficacy outcome measure was the percentage reduction in seizure frequency from the Baseline phase, during the maintenance period of the Core phase. Additional efficacy outcome measures included response rate, defined as at least a 50% reduction in seizure frequency from the Baseline phase during the maintenance period of the Core phase, and seizure freedom rate during the maintenance period of the Core phase.
A total of 366 patients were randomized to everolimus tablets for oral suspension LT (n = 117), everolimus tablets for oral suspension HT (n = 130) or placebo (n = 119). Median age was 10.1 years (2.2 to 56 years); 28% of patients were < 6 years, 31% were 6 to < 12 years, 22% were 12 to < 18 years, and 18% were ≥ 18 years). The majority were white (65%) and male (52%). The most common major features of TSC were cortical tubers (92%), hypomelanotic macules (84%), and subependymal nodules (83%). While 17% of the patients had SEGA, 42% had renal angiomyolipoma, and 9% had both SEGA and renal angiomyolipoma; no patients were receiving treatment with everolimus tablets for oral suspension for these manifestations of TSC. During the Baseline phase, 65% of patients had complex partial seizures, 52% had secondarily generalized seizures, 19% had simple partial seizures, and 2% had generalized onset seizures. The median seizure frequency per week during the Baseline phase was 9.4 for all patients and 47% of patients were receiving 3 AEDs during the Baseline phase. The efficacy results are summarized in Table 26.
Table 26: Percentage Reduction in Seizure Frequency and Response Rate in TSC-Associated Partial-Onset Seizures in EXIST-3
| Everolimus Tablets for Oral Suspension | Placebo | ||
Target of 3-7 ng/mL N = 117 | Target of 9-15 ng/mL N = 130 | ||
| N = 119 | |||
| Seizures per week | |||
| Median at Baseline (Min, Max) | 8.6 (1.4, 192.9) | 9.5 (0.3, 218.4) | 10.5 (1.3, 231.7) |
| Median at Core phase a (Min, Max) | 6.8 (0.0, 193.5) | 4.9 (0.0, 133.7) | 8.5 (0.0, 217.7) |
| Percentage reduction from Baseline to Core phase (Maintenance a ) | |||
| Median | 29.3 | 39.6 | 14.9 |
| 95% CI b | 18.8, 41.9 | 35.0, 48.7 | 0.1, 21.7 |
| p-value c | 0.003 | < 0.001 | |
| Response rate | |||
| Responders, n (%) | 28.2 | 40 | 15.1 |
| 95% CI d | 20.3, 37.3 | 31.5, 49.0 | 9.2, 22.8 |
a If patient discontinued before starting the Maintenance period, then the Titration period is used. b 95% CI of the median based on bootstrap percentiles. c p-values were for superiority vs. placebo, and obtained from rank ANCOVA with Baseline seizure frequency as covariate, stratified by age subgroup. d Exact 95% CI obtained using Clopper-Pearson method. | |||
HOW SUPPLIED/STORAGE AND HANDLING
Everolimus tablets for oral suspension
2 mg tablets: White to off white, round shaped, flat faced bevelled edged tablets debossed with "E2" on one side and plain on other side and free from physical defects.
HDPE Bottles of 28’s Count ---------------------------------- NDC 70377-090-11
Blisters of 28 tablets (Desiccant Embedded Alu-Alu Blister pack) -- NDC 70377-090-23
Each carton contains 4 blister cards of 7 tablets each
3 mg tablets: White to off white, round shaped, flat faced bevelled edged tablets debossed with "E3" on one side and plain on other side and free from physical defects.:
HDPE Bottles of 28’s Count ---------------------------------NDC 70377-091-11
Blisters of 28 tablets (Desiccant Embedded Alu-Alu Blister pack) -- NDC 70377-091-23
Each carton contains 4 blister cards of 7 tablets each
5 mg tablets: White to off white, round shaped, flat faced bevelled edged tablets debossed with "E5" on one side and plain on other side and free from physical defects.:
HDPE Bottles of 28’s Count ---------------------------------NDC 70377-092-11
Blisters of 28 tablets (Desiccant Embedded Alu-Alu Blister pack) -- NDC 70377-092-23
Each carton contains 4 blister cards of 7 tablets each
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature.
Store in the original container, protect from light and moisture.
Follow special handling and disposal procedures for anti-cancer pharmaceuticals. 1
Mechanism of Action
Everolimus is an inhibitor of mammalian target of rapamycin (mTOR), a serine-threonine kinase, downstream of the PI3K/AKT pathway. The mTOR pathway is dysregulated in several human cancers and in tuberous sclerosis complex (TSC). Everolimus binds to an intracellular protein, FKBP-12, resulting in an inhibitory complex formation with mTOR complex 1 (mTORC1) and thus inhibition of mTOR kinase activity. Everolimus reduced the activity of S6 ribosomal protein kinase (S6K1) and eukaryotic initiation factor 4E-binding protein (4E-BP1), downstream effectors of mTOR, involved in protein synthesis. S6K1 is a substrate of mTORC1 and phosphorylates the activation domain 1 of the estrogen receptor which results in ligand-independent activation of the receptor. In addition, everolimus inhibited the expression of hypoxia-inducible factor (e.g., HIF-1) and reduced the expression of vascular endothelial growth factor (VEGF). Inhibition of mTOR by everolimus has been shown to reduce cell proliferation, angiogenesis, and glucose uptake in in vitro and/or in vivo studies.
Two regulators of mTORC1 signaling are the oncogene suppressors tuberin-sclerosis complexes 1 and 2 ( TSC1, TSC2 ). Loss or inactivation of either TSC1 or TSC2 leads to activation of downstream signaling. In TSC, a genetic disorder, inactivating mutations in either the TSC1 or the TSC2 gene lead to hamartoma formation throughout the body as well as seizures and epileptogenesis. Overactivation of mTOR results in neuronal dysplasia, aberrant axonogenesis and dendrite formation, increased excitatory synaptic currents, reduced myelination, and disruption of the cortical laminar structure causing abnormalities in neuronal development and function. Treatment with an mTOR inhibitor in animal models of mTOR dysregulation in the brain resulted in seizure suppression, prevention of the development of new-onset seizures, and prevention of premature death.