Get your patient on Fensolvi - Leuprolide Acetate (Leuprolide Acetate)
Fensolvi - Leuprolide Acetate prescribing information
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
FENSOLVI is indicated for the treatment of pediatric patients 2 years of age and older with central precocious puberty (CPP).
DOSAGE AND ADMINISTRATION
Dosing Information
FENSOLVI must be administered by a healthcare professional.
The dose of FENSOLVI is 45 mg administered by subcutaneous injection once every six months.
Discontinue FENSOLVI treatment at the appropriate age of onset of puberty.
Monitoring
Monitor response to FENSOLVI with a GnRH agonist stimulation test, basal serum luteinizing hormone (LH) levels or serum concentration of sex steroid levels at 1 to 2 months following initiation of therapy and as needed to confirm adequate suppression of pituitary gonadotropins, sex steroids, and progression of secondary sexual characteristics. Measure height (for calculation of growth velocity) every 3 to 6 months and monitor bone age periodically.
Noncompliance with drug regimen or inadequate dosing may lead to gonadotropins and/or sex steroids increasing above prepubertal levels resulting in inadequate control of the pubertal process. If the dose of FENSOLVI is not adequate, switching to an alternative GnRH agonist for the treatment of CPP with the ability for dose adjustment may be necessary.
Reconstitution Instructions
Use aseptic technique including gloves for reconstitution and administration. Allow the product to reach room temperature before reconstitution to allow for easier administration. Once reconstituted, the concentration is 45 mg/0.375 mL. Administer the product within 30 minutes or discard.
FENSOLVI is packaged in a carton containing two trays and this package insert:
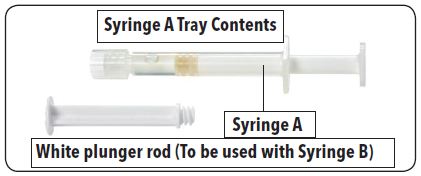
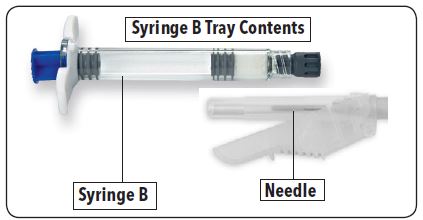
Table 1: Contents of the Two Trays in the FENSOLVI Carton
Syringe A Tray | Syringe B Tray |
Syringe A is prefilled with diluent for reconstitution (ATRIGEL Delivery System) | Syringe B is prefilled with 45 mg lyophilized leuprolide acetate powder |
White plunger rod (To be used with Syringe B) | Safety needle (18G x 5/8") |
Desiccant pack | Desiccant pack |
Follow the instructions below to prepare FENSOLVI:
  | 1. On a clean field, open both trays by tearing off the foil from the corners and removing the contents. Discard the desiccant pack(s). Open the safety needle package by peeling back the paper tab. |
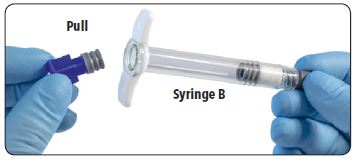 | 2. Pull out (do not unscrew) the short blue plunger rod with attached gray stopper from Syringe B and discard. |
 | 3. Gently screw the white plunger rod into the remaining gray stopper in Syringe B. |
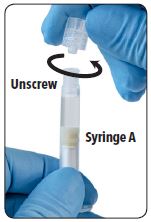 | 4. Unscrew and discard the clear cap from Syringe A. |
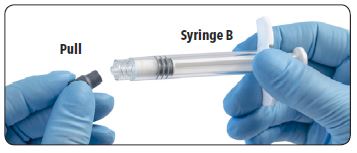 | 5. Remove and discard the gray rubber cap from Syringe B. |
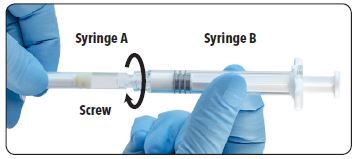 | 6. Join the two syringes together by pushing and gently screwing until secure. |
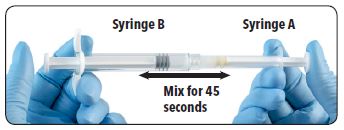 | 7. Inject the liquid contents of Syringe A into the leuprolide acetate powder contained in Syringe B. Thoroughly mix the product for approximately 45 seconds by pushing the contents back and forth between both syringes to obtain a uniform suspension. When thoroughly mixed, the suspension will appear pale yellow. Note: Product must be mixed as described; shaking will not provide adequate mixing. |
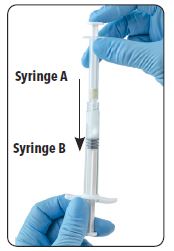 | 8. After mixing, hold the syringes vertically (upright) with Syringe B (wide syringe) on the bottom. The syringes should remain securely coupled. Draw all of the mixed product into Syringe B by depressing the Syringe A plunger and slightly withdrawing the Syringe B plunger. |
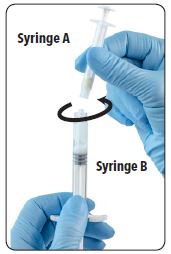 | 9. Unscrew Syringe A to decouple the syringes while continuing to withdraw the Syringe B plunger. Note: Small air bubbles will remain in the formulation – this is acceptable. |
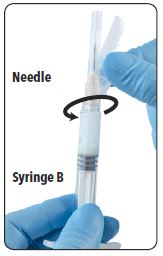 | 10. Continue to hold Syringe B upright with the open end at the top. Hold back the white plunger on Syringe B to prevent loss of the product and attach the safety needle cartridge. Gently screw clockwise with approximately a three-quarter turn until the needle is secure. Do not overtighten, as the hub may become damaged resulting in leakage of the product during injection. The safety sheath may also be damaged if the needle is screwed with too much force. |
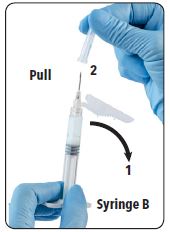 | 11. (1) Move the safety sheath away from the needle and towards the syringe and (2) pull off the clear needle cartridge cover immediately prior to administration. |
Note: Should the needle hub appear to be damaged, or leak, do not use the product. If the needle hub is damaged or leakage is observed, use a new FENSOLVI carton.
Administration Instructions
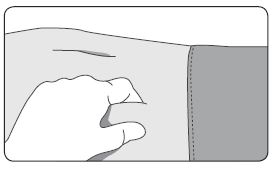 | 1. Select a subcutaneous injection site on the abdomen, upper buttocks, or another location with adequate amounts of subcutaneous tissue that does not have excessive pigment, nodules, lesions, or hair. Avoid areas with brawny or fibrous subcutaneous tissue or locations that could be rubbed or compressed (i.e., by a belt or clothing waistband). Rotate injection sites with each injection. 2. Cleanse the injection-site area with an alcohol swab (not enclosed). 3. Using the thumb and forefinger, grab and bunch the area of skin around the injection site. |
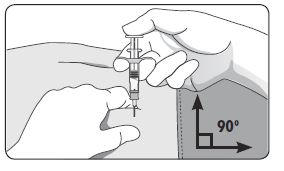 | 4. Using your dominant hand, insert the needle quickly at a 90° angle to the skin surface. The depth of penetration will depend on the amount and fullness of the subcutaneous tissue and the length of the needle. After the needle is inserted, release the skin. 5. Inject the drug using a slow, steady push and press down on the plunger until the syringe is empty. 6. Withdraw the needle quickly at the same 90° angle used for insertion. |
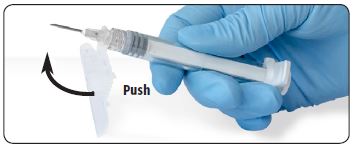 | 7. Immediately following the withdrawal of the needle, activate the safety shield using a finger/thumb or flat surface and push until it completely covers the needle tip and locks into place. |
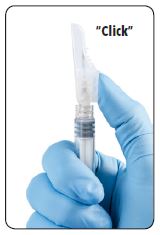 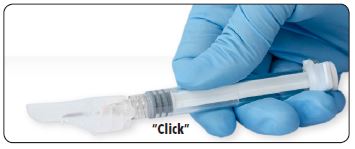 | 8. An audible and tactile “click” verifies a locked position. 9. Check to confirm the safety sheath is fully engaged. Discard all components safely in an appropriate biohazard container. |
DOSAGE FORMS AND STRENGTHS
For injectable suspension, 45 mg of leuprolide acetate is supplied in a kit containing:
- Syringe A contains diluent for reconstitution (ATRIGEL Delivery System) in a prefilled syringe.
- Syringe B contains 45 mg lyophilized leuprolide acetate powder in a single-dose prefilled syringe.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
FENSOLVI is contraindicated in pregnancy [see Contraindications (4 )] .
FENSOLVI may cause fetal harm based on findings from animal studies and the drug’s mechanism of action [see Clinical Pharmacology (12.1 )]. The available data from published clinical studies and case reports and from the pharmacovigilance database on exposure to leuprolide acetate during pregnancy are insufficient to assess the risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Based on animal reproduction studies, leuprolide acetate may be associated with an increased risk of pregnancy complications, including early pregnancy loss and fetal harm. In animal reproduction studies, subcutaneous administration of leuprolide acetate to rabbits during the period of organogenesis caused embryo-fetal toxicity, decreased fetal weights and a dose-dependent increase in major fetal abnormalities in animals at doses less than the recommended human dose based on body surface area using an estimated daily dose. A similar rat study also showed increased fetal mortality and decreased fetal weights but no major fetal abnormalities at doses less than the recommended human dose based on body surface area using an estimated daily dose (see Data) .
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
When administered on day 6 of pregnancy at test dosages of 0.00024 mg/kg, 0.0024 mg/kg, and 0.024 mg/kg (1/3255 to 1/33 of the human dose) to rabbits, leuprolide acetate produced a dose-related increase in major fetal abnormalities. Similar studies in rats failed to demonstrate an increase in fetal malformations. There was increased fetal mortality and decreased fetal weights with the two higher doses of leuprolide acetate in rabbits and with the highest dose (0.024 mg/kg) in rats.
Lactation
Risk Summary
There are no data on the presence of leuprolide acetate in either animal or human milk, the effects on the breastfed infants, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for FENSOLVI and any potential adverse effects on the breastfed infant from FENSOLVI or from the underlying maternal condition.
Females and Males of Reproductive Potential
Pregnancy Testing
Exclude pregnancy in women of reproductive potential prior to initiating FENSOLVI if clinically indicated [see Use in Specific Populations (8.1 ) ] .
Contraception
Females
FENSOLVI may cause embryo-fetal harm when administered during pregnancy. FENSOLVI is not a contraceptive. If contraception is indicated, advise females of reproductive potential to use a non-hormonal method of contraception during treatment with FENSOLVI [see Use in Specific Populations (8.1 )] .
Infertility
Based on its pharmacodynamic effects of decreasing secretion of gonadal steroids, fertility is expected to be decreased while on treatment with FENSOLVI. Clinical and pharmacologic studies in adults (>18 years) with leuprolide acetate and similar analogs have shown reversibility of fertility suppression when the drug is discontinued after continuous administration for periods of up to 24 weeks [see Clinical Pharmacology (12.1 )] .
There is no evidence that pregnancy rates are affected following discontinuation of FENSOLVI.
Animal studies (prepubertal and adult rats and monkeys) with leuprolide acetate and other GnRH analogs have shown functional recovery of fertility suppression.
Pediatric Use
The safety and effectiveness of FENSOLVI for the treatment of CPP has been established in pediatric patients 2 years of age and older. Use of FENSOLVI for this indication is supported by evidence from an adequate and uncontrolled open-label, single-arm study of 64 pediatric patients with CPP with an age range of 4 to 9 years [see Clinical Studies (14 )] . The safety and effectiveness of FENSOLVI have not been established in pediatric patients less than 2 years old.
WARNINGS AND PRECAUTIONS
Initial Rise of Gonadotropins and Sex Steroid Levels
During the early phase of therapy, gonadotropins and sex steroids rise above baseline because of the initial stimulatory effect of the drug [see Clinical Pharmacology (12.2 )] . Therefore, an increase in clinical signs and symptoms of puberty including vaginal bleeding may be observed during the first weeks of therapy or after subsequent doses [see Adverse Reactions (6) ] . Instruct patients and caregivers to notify the physician if these symptoms continue beyond the second month after FENSOLVI administration.
Psychiatric Events
Psychiatric events have been reported in patients taking GnRH agonists, including leuprolide acetate. Postmarketing reports with this class of drugs include symptoms of emotional lability, such as crying, irritability, impatience, anger, and aggression. Monitor for development or worsening of psychiatric symptoms during treatment with FENSOLVI [see Adverse Reactions (6.1 , 6.2) ] .
Convulsions
Postmarketing reports of convulsions have been observed in patients receiving GnRH agonists, including leuprolide acetate. These included patients with a history of seizures, epilepsy, cerebrovascular disorders, central nervous system anomalies or tumors, and patients on concomitant medications that have been associated with convulsions such as bupropion and SSRIs. Convulsions have also been reported in patients in the absence of any of the conditions mentioned above [see Adverse Reactions (6.2) ] .
Pseudotumor Cerebri (Idiopathic Intracranial Hypertension)
Pseudotumor cerebri (idiopathic intracranial hypertension) have been reported in pediatric patients receiving GnRH agonists, including leuprolide acetate. Monitor patients for signs and symptoms of pseudotumor cerebri, including headache, papilledema, blurred vision, diplopia, loss of vision, pain behind the eye or pain with eye movement, tinnitus, dizziness, and nausea.
ADVERSE REACTIONS
The following serious adverse reactions are described here and elsewhere in the label:
- Initial rise in gonadotropin and sex steroid levels [see Warnings and Precautions (5.1 )] .
- Psychiatric Events [see Warnings and Precautions (5.2 )] .
- Convulsions [see Warnings and Precautions (5.3 )] .
- Pseudotumor Cerebri (Idiopathic Intracranial Hypertension) [ see Warnings and Precautions (5.4 )]
Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
FENSOLVI was evaluated in an uncontrolled, open-label, single-arm clinical trial in which 64 pediatric patients with CPP received at least one dose of FENSOLVI. The age ranged from 4 to 9 years at start of treatment; 62 patients were female and 2 were male; 53% White; 23% Black; 8% American Indian or Alaska Native; 5% Asian; 2% Native Hawaiian or Other Pacific Islander. 56% of the subjects self-identified as Hispanic or Latino ethnicity. Adverse reactions that occurred in ≥ 5% of patients are shown in Table 2.
Table 2: Adverse Reactions Occurring in ≥ 5% of Patients Treated with FENSOLVI in an Open-Label, Single-Arm Trial
| Adverse Reactions | % of Patients (N=64) |
| Injection site pain | 31 |
| Nasopharyngitis | 22 |
| Pyrexia | 17 |
| Headache | 16 |
| Cough | 13 |
| Abdominal pain | 9 |
| Injection site erythema | 9 |
| Nausea | 8 |
| Constipation | 6 |
| Vomiting | 6 |
| Upper respiratory tract infection | 6 |
| Bronchospasm | 6 |
| Productive cough | 6 |
| Hot flush | 5 |
Other Adverse Reactions:
Psychiatric
Emotional disorder (2%) and irritability (2%)
Postmarketing Experience
The following adverse reactions have been identifed during postapproval use of leuprolide acetate or FENSOLVI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Allergic Reactions: anaphylactic, rash, urticaria, and photosensitivity reactions.
General : chest pain, weight increase, weight decrease, decreased appetite, fatigue.
Laboratory Abnormalities: decreased WBC.
Metabolic : diabetes mellitus.
Musculoskeletal and Connective Tissue : arthralgia, epiphysiolysis, muscle spasms, myalgia.
Neurologic: neuropathy peripheral, convulsion, paralysis, insomnia, pseudotumor cerebri (idiopathic intracranial hypertension).
Psychiatric: emotional lability, such as crying, irritability, impatience, anger and aggression. Depression, including rare reports of suicidal ideation and attempt. Many, but not all, of these patients had a history of psychiatric illness or other comorbidities with an increased risk of depression.
Skin and Subcutaneous Tissue : injection site reactions including induration and abscess, flushing, hyperhidrosis.
Reproductive System: vaginal bleeding, breast enlargement.
Vascular : hypertension, hypotension.
Respiratory: dyspnea.
DRUG INTERACTIONS
No pharmacokinetic drug-drug interaction studies have been conducted with FENSOLVI.
DESCRIPTION
FENSOLVI for injectable suspension is a sterile polymeric matrix formulation of leuprolide acetate, a GnRH agonist, for subcutaneous use. It is designed to deliver leuprolide acetate at a controlled rate over a six-month therapeutic period.
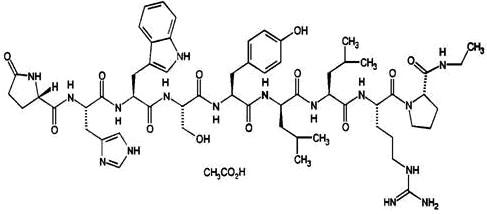
Leuprolide acetate is a synthetic nonapeptide analog of naturally occurring gonadotropin releasing hormone. The analog possesses greater potency than the natural hormone. The chemical name is 5-oxo-L-prolyl-L-histidyl-L-tryptophyl-L-seryl-L-tyrosyl-D-leucyl-L-leucyl-L-arginyl-N-ethyl-L-prolinamide acetate (salt) with the following structural formula:
FENSOLVI is prefilled and supplied in two separate, sterile syringes whose contents are mixed immediately prior to administration. The two syringes are joined and the single dose product is mixed until it is homogenous. FENSOLVI is administered subcutaneously, where it forms a solid drug delivery depot.
One syringe contains the ATRIGEL Delivery System and the other contains the leuprolide acetate. ATRIGEL is a polymeric (non-gelatin containing) delivery system consisting of a biodegradable poly(DL-lactide-co-glycolide) (PLG) polymer formulation dissolved in the biocompatible solvent, N-methyl-2-pyrrolidone (NMP).
Refer to Table 3 for the delivery system composition and reconstituted product formulation for FENSOLVI product.
Table 3: FENSOLVI Delivery System Composition and Reconstituted Product Formulation
| ATRIGEL Delivery System Syringe | Polymer | PLG |
| Polymer description | Copolymer with hexanediol | |
| Polymer DL-lactide to glycolide molar ratio | 85:15 | |
Reconstituted Product | Polymer delivered | 165 mg |
| NMP delivered | 165 mg | |
| Leuprolide acetate delivered | 45 mg | |
| Approximate leuprolide free base equivalent | 42 mg | |
| Approximate administered formulation weight | 375 mg | |
| Approximate injection volume | 0.375 mL |
CLINICAL PHARMACOLOGY
Mechanism of Action
Leuprolide acetate, a GnRH agonist, acts as a potent inhibitor of gonadotropin secretion (LH and follicle stimulating hormone (FSH)) when given continuously in therapeutic doses. Following an initial stimulation of GnRH receptors, chronic administration of leuprolide acetate results in downregulation of GnRH receptors, reduction in release of LH, FSH and consequent suppression of ovarian and testicular production of estradiol and testosterone respectively. This inhibitory effect is reversible upon discontinuation of drug therapy.
Pharmacodynamics
In the clinical trial evaluating FENSOLVI in pediatric patients with CPP, there was a transient surge in circulating levels of LH, FSH, estradiol and testosterone following the first administration. A decrease in basal and GnRH agonist-stimulated LH and FSH levels along with reductions in basal estradiol and testosterone were observed after repeat administration.
Pharmacokinetics
Absorption
After an initial subcutaneous injection of FENSOLVI 45 mg in pediatric patients 4 to 9 years of age with CPP, leuprolide levels peaked 4 hours postdose with a mean C max of 212.3 ng/mL. Absorption occurred in two phases, a burst phase followed by a plateau phase. The mean plateau serum leuprolide level from 4 to 48 weeks was approximately 0.37 ng/mL with a range of 0.18 to 0.63 ng/mL. There was no accumulation of leuprolide after the second dose.
Distribution
The distribution of leuprolide following FENSOLVI administration was not evaluated in pediatric patients. The mean steady-state volume of distribution of leuprolide following intravenous bolus administration to healthy male volunteers was 27 L.
In vitro binding of leuprolide to human plasma proteins ranged from 43% to 49%.
Elimination
Metabolism
In rats and dogs, administration of 14 C-labeled leuprolide was shown to be metabolized to smaller inactive peptides: a pentapeptide (Metabolite I), tripeptides (Metabolites II and III), and a dipeptide (Metabolite IV). These fragments may be further catabolized.
In healthy male volunteers, a 1 mg bolus of leuprolide administered intravenously revealed that the mean systemic clearance was 8.34 L/h, with a terminal elimination half-life of approximately 3 hours based on a two-compartment model.
Drug metabolism studies were not conducted with FENSOLVI. Upon administration with different leuprolide acetate formulations, the major metabolite of leuprolide acetate is a pentapeptide (M-1) metabolite.
Excretion
Drug excretion studies were not conducted with FENSOLVI.
Specific Populations
The pharmacokinetics of FENSOLVI in hepatically and renally impaired pediatric patients have not been determined.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted with leuprolide acetate in rats and mice. In rats, a dose-related increase of benign pituitary hyperplasia and benign pituitary adenomas was noted at 24 months when the drug was administered subcutaneously at high daily doses (0.6 to 4 mg/kg). There was a significant but not dose-related increase of pancreatic islet-cell adenomas in females and of testicular interstitial cell adenomas in males (highest incidence in the low dose group). In mice, no leuprolide acetate-induced tumors or pituitary abnormalities were observed at a dose as high as 60 mg/kg for two years. Patients have been treated with leuprolide acetate for up to three years with doses as high as 10 mg/day and for two years with doses as high as 20 mg/day without demonstrable pituitary abnormalities. No carcinogenicity studies have been conducted with FENSOLVI.
Mutagenicity studies have been performed with leuprolide acetate using bacterial and mammalian systems. These studies provided no evidence of a mutagenic potential.
CLINICAL STUDIES
The efficacy of FENSOLVI was evaluated in an uncontrolled, open-label, single arm clinical trial in which 64 pediatric patients (62 females and 2 males, naïve to previous GnRH agonist treatment) with CPP received at least one dose of FENSOLVI at a dosing interval of 24 weeks and were observed for 12 months. The mean age was 7.5 years (range 4 to 9 years) at the start of treatment. In pediatric patients with CPP, FENSOLVI reduced stimulated and basal gonadotropins to prepubertal levels. Suppression of peak stimulated LH concentrations to <4 IU/L was achieved in 87% of pediatric patients by month 6 and in 86% of patients by month 12. Suppression of estradiol or testosterone concentration to prepubertal levels at the 6-month assessment was achieved in 97% and 100% of patients, respectively. Suppression of estradiol or testosterone was maintained at the 12-month assessment with 98% (55/56 females) and 50% (1/2 males) maintaining suppression. FENSOLVI arrested or reversed progression of clinical signs of puberty with reductions in growth velocity and bone age. Mean growth velocity decreased from 8.9 ± 13.1 cm/yr at 1 month to 6.9 ± 3.1 cm/yr at 6 months and to 6.4 ± 1.9 cm/yr at 12 months.
Table 4: Reproductive Hormone Levels in Pediatric Patients with CPP Treated with FENSOLVI 45 mg Every 6 Months a
| % (n/N) of Patients Achieving Endpoints | ||||
| Endpoint b | Month 3 | Month 6 | Month 9 | Month 12 |
| LH levels < 4 IU/L | 85 (51/60) | 87 (54/62) c | 85 (50/59) | 86 (50/58) |
| Estradiol levels < 73.4 pmol/L (< 20 pg/mL) | 98 (56/57) | 97 (58/60) | 98 (56/57) | 98 (55/56) |
| Testosterone levels < 1 nmol/L (< 28.4 ng/dL) | 100 (2/2) | 100 (2/2) | 100 (2/2) | 50 (1/2) |
| FSH levels < 2.5 IU/L | 62 (37/60) | 66 (41/62) | 44 (26/59) | 55(32/58) |
| a Intent-to-treat Population (N=62) b Post GnRH agonist stimulation c Primary Efficacy Endpoint | ||||
Eight female patients out of 62 did not meet the primary efficacy criteria for LH <4 IU/L at 6 months. In four of the eight patients, the LH level at 6 months was between 4.2 and 4.8 IU/L. The remaining four patients had LH levels >5 IU/L. However, post stimulation estradiol was suppressed to prepubertal levels (<20 pg/mL) in seven of the eight patients at month 6 and was maintained through month 12.
HOW SUPPLIED/STORAGE AND HANDLING
For injectable suspension, 45 mg of leuprolide acetate supplied in a kit (NDC 62935-153-50) containing:
Syringe A Tray | Syringe B Tray |
Syringe A is prefilled with diluent for reconstitution (ATRIGEL Delivery System) | Syringe B is prefilled with 45 mg lyophilized leuprolide acetate powder |
White plunger rod (To be used with Syringe B) | Safety needle (18G x 5/8") |
Desiccant pack | Desiccant pack |
Store refrigerated at 2 - 8 °C (35.6 – 46.4 °F).
Once outside the refrigerator, this product may be stored in its original packaging at room temperature 15 – 30 °C (59 – 86 °F) for up to eight weeks prior to reconstitution and administration.
Mechanism of Action
Leuprolide acetate, a GnRH agonist, acts as a potent inhibitor of gonadotropin secretion (LH and follicle stimulating hormone (FSH)) when given continuously in therapeutic doses. Following an initial stimulation of GnRH receptors, chronic administration of leuprolide acetate results in downregulation of GnRH receptors, reduction in release of LH, FSH and consequent suppression of ovarian and testicular production of estradiol and testosterone respectively. This inhibitory effect is reversible upon discontinuation of drug therapy.